 ABOUT NPASS
ABOUT NPASS
Modern drug discovery and today's pharmacopeia is largely benefited from nature. More than 50% of approved drugs are natural products or natural product derivatives. It is estimated that there are about a million natural products have been isolated and many of them have been subjected to experimental assays to evaluate quantitative biological activities. By manually collecting and integrating these valuable data from individual literatures into a centralized database, the Natural Product Activity and Species Source Database (NPASS) has serverd as one of a leading data source for the NP research community since its first version release in 2017. For example, Athina Gavriilidou et al. (Nature Microbiology, 7:726–735, 2022) analyzed the of biosynthetic diversity and NP discovery hotspots to guide NP research trends, based on NPASS active NP dataset. Michael A. Skinnider et al. developed artifial intelligence alogrithm for molecule AI design (Nature Machine Intelligence, 3:759–770, 2021). Laura Holzmeyer et al. revealed the hidden relationship between phylogenetic, spatial and bioactivity of plant-derived NPs (PNAS, 117(22):12444–12451, 2020).
Since 2018, a large number of new NPs have been found and biologically evaluated against a broad of biological targets by the research community, to better benefit the NP research community, we updated NPASS database to deposit more data points and more information layers of NP in the new version (v2.0). The NPASS database in the current version (V2.0) provides 94,413 unique natural products isolated from 32,287 source organisms and together with 958,866 activity records on 7,753 targets. Major changes in NPASS-v2.0 were summarized in the below table:
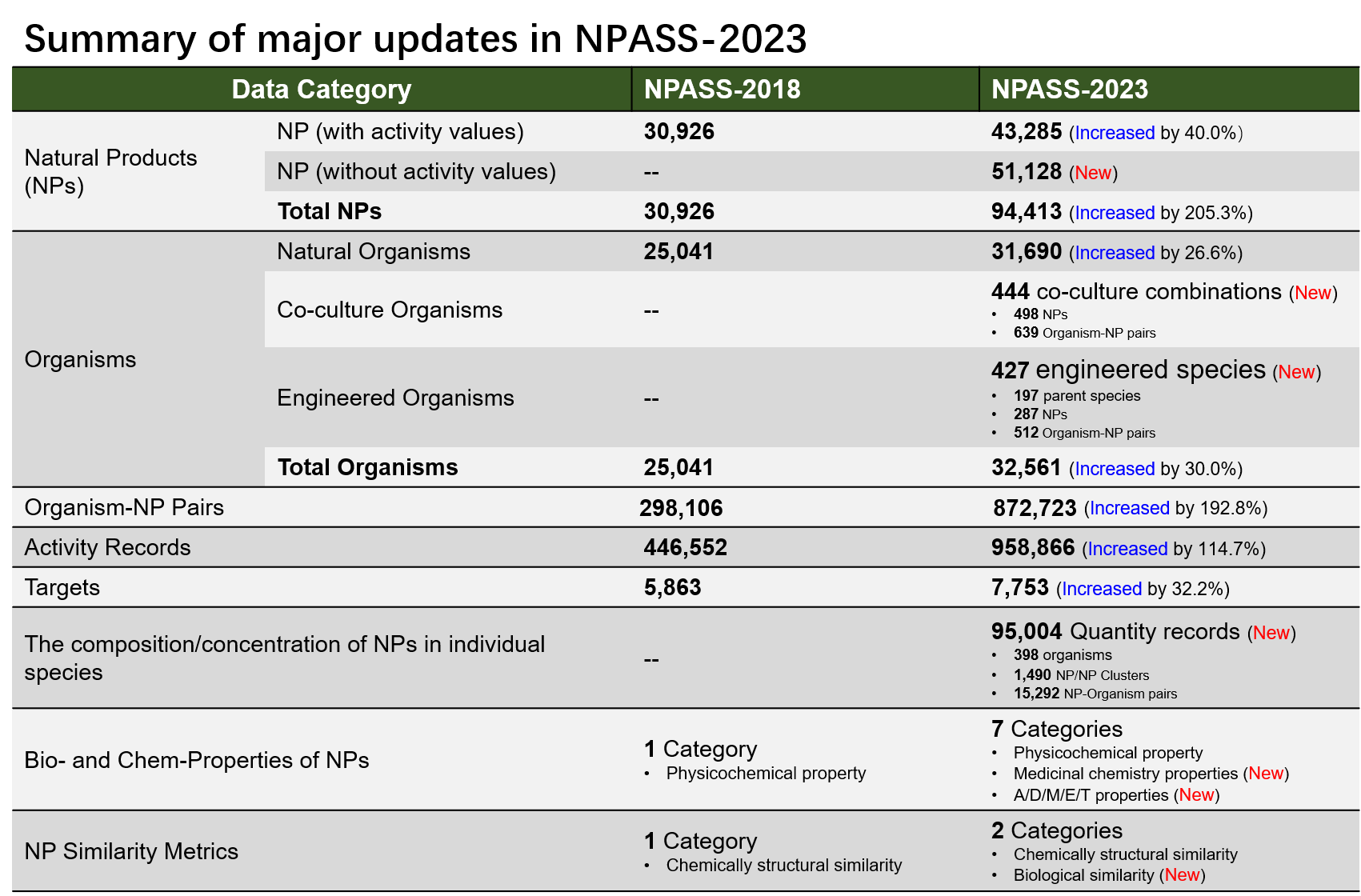
❖ Co-culture microorganisms and engineered microorganisms for optimized production of high-value NPs
Recently years, deeper understanding in biosynthesis mechanisms, advances in synthetic biology and metabolic/genetic reprogramming strategies are evolving the NP research paradigm. In the nature, microorganisms merely grow and function (e.g., producing secondary metabolites) independently, but cooperate with other species in their surrounding living system. In addition, many synthetic gene clusters for biosynthesis of high-value NPs were usually silenced in natural organisms.
By manipulating the cultivation system or engineering metabolic pathways/synthetic gene clusters, NPs can be produced by organisms in more efficient and programmable manners. Co-culture and engineering natural species are two important synthetic biology strategies to achieve such purposes. By activating silenced synthetic pathways or introducing synthetic enzymes to model species, or by recovering microbe-microbe interactions, organism engineering and co-culture systems (also termed microbial consortia) are emerging strategies to produce novel NPs or increase yield of high-value NPs.
A comprehensive dataset of co-culture and engineered organisms is of importance for the NP research community, such as to deeper understand the potentials and diversity of biosynthesis space of organisms for NPs and drug discovery. To cater this need, we extensively searched PubMed and manually curated 444 co-culture combinations and 427 engineered organisms from literature. This dataset can be browsed via the below link.
BROWSE All Co-culture Organism Systems and Engineered Organisms for Optimized NP Producing [Click here]
❖ The workflow of developing NPASS is illustrated in the below figure

 DATA SOURCES
DATA SOURCES
I. Species source of natural products
Species source information is mainly from manually inspection of publications. Besides, we surveyed existing natural product-related databases to find species source annotations.
♦ Manually annotated from publications
Multiple keywords/keywords conbinations are used to search literatures that may revelant to isolation, total synthesis, activity evaluation of NPs through PubMed. These keywords include natural product, NP, nature, marine, plant, microbe, microbial, bacterium, bacteria, bacterial, fungus, fungi, fungal, species, traditional medicine, medicinal, indigenous, folk, herb, herbal, herbalism, Chinese medicine, TCM, Ayurveda, activity, active, bioactive, potent, potency, IC50, Ki, EC50, GI50, and MIC. Searched publications are subjected to first-step manually check the title to confirm if the literature is really revelant. Then, full articles of these relavent publications are downloaded for manually checking the species source (including if the NP is novel structure claimed by authors, species collection location and time, species part used for isolation, etc.) of corresponding natural products.
♦ Collected from existing databases
Few existing databases include a part of species source information of natural products. These databases includes: TCM-ID, TCMID, TCM@TaiWan, TCMSP, UNPD, TM-MC, StreptomeDB, TTD, TarNet, ChEBI, and HerDing. Therefore, NP names/structures are searched against these databases to extract species source information.
II. Biological activities of natural products
Quantitative activity data of NPs against specific targets (including: target information, activity type and values, compound dose etc.) are integrated from ChEMBL database (ver-30) and manually curated from literatures searched described in previous paragraph. Collected activity types includes inhibition concentration/dose like IC50/IC90/ID50, activity concentrations like AC/AC40/AC50/Potency, microbial inhibitory or lethal concentrations like MIC/MFC/MBC/FC, growth inhibitory concentrations like GI/GI50/TGI, percentage inhibition at fixed concentrations like inhibition rate, efective contrations/doses like EC50/EC90/ED50/ED90, equilibrium inhibition constant Ki, lethal concentrations/doses like LC/LC50/LC90/LD50/LD90, inhibition zone IZ, equilibrium binding constant Kd, ratio IC50/ratio EC50/ratio/Ki, cytotoxic concentrations like CC25/CC50/CC90/CC100, and toxic concentration/dose like TC50/TD50. More than half of activity values are stored as the unit of nM, other units include ug/ml, mg/kg, %, mm, and so on.
 NATURAL PRODUCT PROFILE
NATURAL PRODUCT PROFILE
I. Natural products
♦ Natural products chemical representation
Common name, synonyms, IUPAC Name, Standard InCHI, Standard InCHI Key, Canonical SMILES, and MOL file.
♦ Natural products physical & chemical properties
Molecular formula, molecular weight, AlogP, # hydrogen bond donor, # hydrogen bond acceptor, polar surface area, # rotatable bond, # aromatic rings, # heavy atoms.
II. Clinical/approved drugs
Clinical trial and approved drugs are collected from TTD (Therapeutical Target Database), Drug Bank, and ChEMBL database.
III. Similarity between molecules
Structure similarity between molecules is defined by Tanimoto coefficient (Tc). Tc is calculated by using PubChem 881-bit substructure fingerprints according to below equation:

Where 'X' and 'X' are fingerprints of two molecules, and 'xi' and 'yi' are the ith bits in each fingerprint. "∧" and "∨" represent the bitwise "and" and bitwise "or", respectively. Ts(xi,yi) is the value of Tanimoto coefficient which is equal to the total number of common substructure features divided by the total number of unique substructures existing in both molecules.
Tc lies between [0,1] where '1' represents the highest similarity between molecule 'X' and 'Y'.
Tc scores between NPs are pre-calculated and stored in NPASS database. While Tc score between user-query-molecule and NPs is calculated realtime by using functions of chemfp toolkit.
 SOURCE ORGANISM PROFILE
SOURCE ORGANISM PROFILE
I. Organism taxonomy information
All organism names extracted from orginal files are firstly matched to scientific names from NCBI taxonomy database, then unmatched names are matched to synonyms from NCBI Taxonomy Database and transformed to scientific name when matched. Finally, those organism names that can not match to any scientific names or synonyms are kept in orginal format. After matching to NCBI TaxonomyDB, taxonomy IDs are recorded to generate external links of organisms.
II. Organism external links
About 60% and 93% source species can be matched to NCBI Taxonomy database at species level and genus level, respectively. For these species, Taxonomy IDs are annotated so that users can review taxonomic details from NCBI Taxonomy database. For the remaining about 7% species which can not match to any entries in NCBI Taxonomy database, we will further check the accuracy of species name and annotate taxonomic information from original literatures. Apart from NCBI Taxonomy database, species organisms are also links to other databases such as World Register of Marine Species (WoRMS) when data available.
 TARGET PROFILE
TARGET PROFILE
Targets are classified into several categories according to classification of ChEMBL database, including 'Individual protein', 'Protein family', 'Protein complex', 'Protein-protein interaction', 'Cell line', 'Organism', and so on. Targets are corsslinked to TTD, Uniprot, ChEMBL, IUPHAR/BPS when possible.
