Collective Molecular Activities of the Plant: Erythrina Suberosa
Plant ID: NPO8301
Plant Latin Name: Erythrina Suberosa
Taxonomy Genus: Erythrina
Taxonomy Family: Fabaceae
Plant External Links:
NCBI TaxonomyDB:
1288013
Plant-of-the-World-Online:
n.a.
Overview of Ingredients
18 All known Ingredients in Total
Unique ingredients have been isolated from this plant.Plant-Ingredients Associations were manually curated from publications or collected from other databases.
9 Ingredients with Acceptable Bioavailablity
Unique ingredients exhibit acceptable human oral bioavailablity, according to the criteria of SwissADME [PMID: 28256516] and HobPre [PMID: 34991690]. The criteria details:SwissADME: six descriptors are used by SwissADME to evaluate the oral bioavailability of a natural product:
☑ LIPO(Lipophility): -0.7 < XLOGP3 < +5.0
☑ SIZE: 150g/mol < MW < 500g/mol
☑ POLAR(Polarity): 20Ų < TPSA < 130Ų
☑ INSOLU(Insolubility): -6 < Log S (ESOL) < 0
☑ INSATU(Insaturation): 0.25 < Fraction Csp3 < 1
☑ FLEX(Flexibility): 0 < Num. rotatable bonds < 9
If 6 descriptors of a natural plant satisfy the above rules, it will be labeled high HOB.
HobPre: A natural plant ingredient with HobPre score >0.5 is labeled high human oral availability (HOB)
9 Ingredients with experimental-derived Activity
Unique ingredients have activity data available.Ingredient Structrual Cards
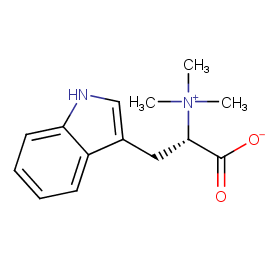
Ingredient ID: NPC84827
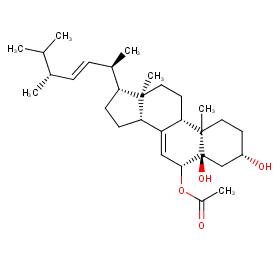
Ingredient ID: NPC77531
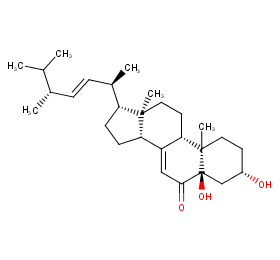
Ingredient ID: NPC66172
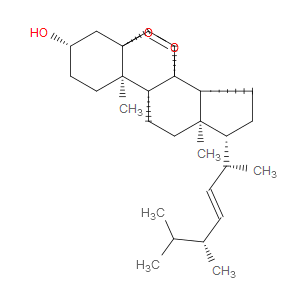
Ingredient ID: NPC4574
Ingredient ID: NPC266746
Ingredient ID: NPC247957
Ingredient ID: NPC231834
Ingredient ID: NPC224103
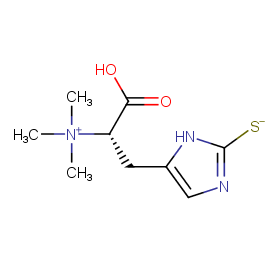
Ingredient ID: NPC218262
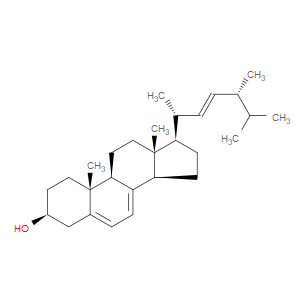
Ingredient ID: NPC18603
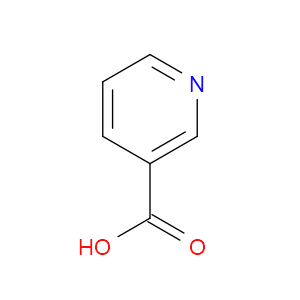
Ingredient ID: NPC167400
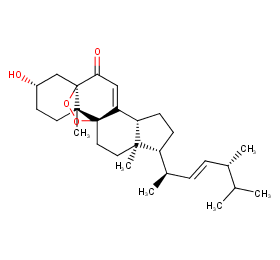
Ingredient ID: NPC155477
Ingredient ID: NPC150480
Ingredient ID: NPC140052
Ingredient ID: NPC133011
Ingredient ID: NPC130926
Ingredient ID: NPC126136
Ingredient ID: NPC121654
Classification of Human Proteins Collectively Targeted by the Plant
Detailed Information of Target Proteins
| Target Type | Protein Class | Gene ID | Protein Name | Uniprot ID | Target ChEMBL ID |
|---|---|---|---|---|---|
| Therapeutic Target | Small molecule receptor (family A GPCR) | HCAR2 | Hydroxycarboxylic acid receptor 2 | Q8TDS4 | CHEMBL3785 |
Clinical trials associated with plant from natural product (NP) & plant level:
| Clinical trials type | Number of clinical trials | |
|---|---|---|
| 76 | ||
| NCT ID | Title | Condition | Form in clinical use | Associated by plant or compound |
|---|---|---|---|---|
| NCT00000482 | Coronary Drug Project | Myocardial Ischemia;myocardial infarction | Niacin (NPC167400) | |
| NCT00300365 | Pioglitazone vs Placebo in Combination With Niacin Extended Release on Low HDL | metabolic syndrome | Niacin (NPC167400) | |
| NCT02702492 | PAK4 and NAMPT in Patients With Solid Malignancies or NHL (PANAMA) | non-Hodgkins lymphoma | Niacin (NPC167400) | |
| NCT00378833 | Tolerability of MK0524A Versus Niacin Extended-Release (0524A-054) | Hypercholesterolemia;hyperlipidemia | Niacin (NPC167400) | |
| NCT04677049 | Study of Niacin in Glioblastoma | glioblastoma multiforme | Niacin (NPC167400) | |
| NCT00508989 | Niacin to Improve Blood Flow in People With Sickle Cell Disease | sickle cell anemia | Niacin (NPC167400) | |
| NCT00000553 | HDL-Atherosclerosis Treatment Study (HATS) | Myocardial Ischemia | Niacin (NPC167400) | |
| NCT00111891 | An Investigational Drug Study Evaluating a Novel Approach to Treat Dyslipidemia (0524A-032) | Disorder of lipid metabolism | Niacin (NPC167400) | |
| NCT00618995 | A Study to Evaluate the Effects of ER Niacin/Laropiprant, Laropiprant, ER Niacin, and Placebo on Urinary Prostanoid Metabolites (0524A-079)(COMPLETED) | type 2 diabetes mellitus | Niacin (NPC167400) | |
| NCT00203476 | A Prospective, Open Label Comparison of Ezetimibe, Niacin, and Colestipol as Adjunct Therapy in Lipid Reduction | Hypercholesterolemia;hyperlipidemia | Niacin (NPC167400) | |
| NCT00289900 | Lipid Efficacy and Safety in Participants With Mixed Hyperlipidemia (MK-0524B-024) | Combined hyperlipidemia | Niacin (NPC167400) | |
| NCT00485758 | Extended Niacin/Laropiprant in Patients With Type 2 Diabetes (0524A-069) | type 2 diabetes mellitus | Niacin (NPC167400) | |
| NCT00533611 | Effect of MK0524A on Flushing Caused by Niacin (0524A-056) | Flushing | Niacin (NPC167400) | |
| NCT03685773 | The Role of Hepatic Denervation in the Dysregulation of Glucose Metabolism in Liver Transplant Recipients | type 2 diabetes mellitus | Niacin (NPC167400) | |
| NCT00080275 | Evaluation of the Safety & Efficacy of a Combination of Niacin ER & Simvastatin in Patients With Dyslipidemia (OCEANS) | Hypercholesterolemia | Niacin (NPC167400) | |
| NCT00626392 | Study to Evaluate the EFFECTS of Acetylsalicylic Acid (ASA) on Niaspan®-Induced Flushing in Subjects With Dyslipidemia | Disorder of lipid metabolism | Niacin (NPC167400) | |
| NCT00957359 | Psilocybin Cancer Anxiety Study | cancer | Niacin (NPC167400) | |
| NCT00533312 | MK0524A Clinical Efficacy Study (0524A-026)(COMPLETED) | Hypercholesterolemia | Niacin (NPC167400) | |
| NCT00536237 | MK0524A Phase IIb Study (0524A-011)(COMPLETED) | Flushing | Niacin (NPC167400) | |
| NCT01321034 | Effect of Niacin in the Lipoprotein (a) Concentration | Hypercholesterolemia | Niacin (NPC167400) | |
| NCT00150722 | High-Density Lipoprotein (HDL) Modulation and Endothelial Function | atherosclerosis | Niacin (NPC167400) | |
| NCT00903617 | Study to Test GSK256073 in Patients With Dyslipidemia | Disorder of lipid metabolism | Niacin (NPC167400) | |
| NCT01426438 | Endothelial Function, Lipoproteins, and Inflammation With Low HDL Cholesterol in HIV: ER Niacin Versus Fenofibrate | HIV-1 infection | Niacin (NPC167400) | |
| NCT04703361 | Effects of Ketones and Niacin in Heart Failure Patients | systolic heart failure | Niacin (NPC167400) | |
| NCT00120289 | Niacin Plus Statin to Prevent Vascular Events | stroke;atherosclerosis;myocardial infarction | Niacin (NPC167400) | |
| NCT01071525 | The Effect of Niacin Administration on Oxidative Stress in Patients With Hypercholesterolmia, as Measured by the Use of a Novel Biomarker | Hypercholesterolemia | Niacin (NPC167400) | |
| NCT00376584 | Effect of an Investigational Compound on Tolerability of Extended Release Niacin (0524A-023)(COMPLETED) | Disorder of lipid metabolism;hyperlipidemia | Niacin (NPC167400) | |
| NCT01368328 | Effect of the Chromium Nicotinate on Type 2 Diabetes | type 2 diabetes mellitus | Niacin (NPC167400) | |
| NCT02109614 | Early Aortic Valve Lipoprotein(a) Lowering Trial | aortic stenosis | Niacin (NPC167400) | |
| NCT00000461 | Harvard Atherosclerosis Reversibility Project (HARP) | Myocardial Ischemia | Niacin (NPC167400) | |
| NCT00500045 | Retrospective Study of the Effectiveness and Safety of Niacin and Steroid Eye Drops for Retinal Vein Occlusions | retinal vein occlusion | Niacin (NPC167400) | |
| NCT00082251 | Safety & Efficacy of a Combination Niacin ER/Simvastatin in Patients With Dyslipidemia: A Dose-Ranging Study - SEACOAST | Hypercholesterolemia | Niacin (NPC167400) | |
| NCT00397657 | Comparative Study of the Effect of Ezetimibe Versus Extended-Release Niacin on Atherosclerosis | atherosclerosis | Niacin (NPC167400) | |
| NCT00630877 | Evaluation of a Flushing ASsessment Tool (FAST) in Subjects Receiving Niacin Extended-release Plus Aspirin | Disorder of lipid metabolism | Niacin (NPC167400) | |
| NCT00269204 | Lipid Efficacy/Tolerability Study (0524A-020) | Combined hyperlipidemia | Niacin (NPC167400) | |
| NCT01583647 | A Study of Extended-release (ER) Niacin/Laropiprant in Adolescents With Heterozygous Familial Hypercholesterolemia (MK-0524A-158) | familial hypercholesterolemia | Niacin (NPC167400) | |
| NCT00796887 | Randomized, Controlled Trial of Extended-Release Niacin (Niaspan®) to Augment Subacute Ischemic Stroke Recovery | Ischemic stroke | Niacin (NPC167400) | |
| NCT01054508 | Effect of Tredaptive on Serum Lipoproteins and Inflammatory Markers | Hypercholesterolemia | Niacin (NPC167400) | |
| NCT01104519 | A Study of Niaspan on Endothelium-Dependent and Endothelium-Independent Vascular Reactivity (0000-093) | Disorder of lipid metabolism | Niacin (NPC167400) | |
| NCT03540758 | Regulation of Endogenous Glucose Production by Central KATP Channels | type 2 diabetes mellitus | Niacin (NPC167400) | |
| NCT01005303 | Micronutrient Supplementation in Patients With Heart Failure | heart failure | Niacin (NPC167400) | |
| NCT03752918 | The Effects of MDMA on Prefrontal and Amygdala Activation in PTSD. | post-traumatic stress disorder | Niacin (NPC167400) | |
| NCT00652431 | Assessment of Interaction Between Vytorin and Niaspan in Healthy Subjects (P04955AM2) | Hypercholesterolemia;hyperlipidemia | Niacin (NPC167400) | |
| NCT01071291 | Effects of Niaspan™ on High-density Lipoprotein (HDL) in Healthy Male Subjects (0000-069) | Disorder of lipid metabolism | Niacin (NPC167400) | |
| NCT00384293 | Carotid IMT (Intima Media Thickening) Study (0524A-041)(TERMINATED) | familial hypercholesterolemia | Niacin (NPC167400) | |
| NCT00269217 | Lipid Efficacy Study (0524B-022)(COMPLETED) | Combined hyperlipidemia;Hypercholesterolemia | Niacin (NPC167400) | |
| NCT00769132 | A Study to Evaluate the Effects of Extended Release (ER) Niacin/Laropiprant, Laropiprant, ER Niacin, and Placebo on Urinary Prostanoid Metabolites in Subjects With High Cholesterol (0524A-075)(COMPLETED) | Hypercholesterolemia | Niacin (NPC167400) | |
| NCT01984073 | Effects of Niacin On Fatty Acid Trapping | Disorder of lipid metabolism | Niacin (NPC167400) | |
| NCT00000599 | Cholesterol-Lowering Atherosclerosis Study (CLAS) | carotid artery disease;cerebral atherosclerosis;Myocardial Ischemia | Niacin (NPC167400) | |
| NCT01720095 | Niapsan as Add -on Therapy to Antipsychotic Treatment in First Episode Psychosis | psychosis | Niacin (NPC167400) | |
| NCT00536510 | Effect of MK0524A on Cholesterol Levels (0524A-048) | Hypercholesterolemia;hyperlipidemia | Niacin (NPC167400) | |
| NCT01083329 | Effect of Nicotinic Acid on Adipose Tissue Inflammation in Obese Subjects | obesity | Niacin (NPC167400) | |
| NCT00127218 | High-Density Lipoprotein (HDL) Cholesterol Increased Plaque Stabilization in the Elderly | atherosclerosis | Niacin (NPC167400) | |
| NCT00152893 | To Determine if Chromium Nicotinate Supplementation Will Improve Insulin Resistance in HIV Patients With Metabolic Abnormalities | HIV infection | Niacin (NPC167400) | |
| NCT00493064 | To Study the Effectiveness and Safety of Niacin and a Topical Steroid Eye Drop to Treat Retinal Vein Occlusions | retinal vein occlusion | Niacin (NPC167400) | |
| NCT03510715 | An Efficacy and Safety Study of Alirocumab in Children and Adolescents With Homozygous Familial Hypercholesterolemia | Hypercholesterolemia | Niacin (NPC167400) | |
| NCT00664287 | Extended Release (ER) Niacin/Laropiprant Add on Study (0524A-082) | Disorder of lipid metabolism | Niacin (NPC167400) | |
| NCT02018965 | Niacin on Immune Activation : a Proof-of-concept Study | HIV infection | Niacin (NPC167400) | |
| NCT00170404 | TB Nutrition, Immunology and Epidemiology | tuberculosis | Niacin (NPC167400) | |
| NCT03356483 | Efficacy of Psilocybin in OCD: a Double-Blind, Placebo-Controlled Study. | obsessive-compulsive disorder | Niacin (NPC167400) | |
| NCT00944645 | MK0524A Bioequivalence Study (0524A-059) | Disorder of lipid metabolism | Niacin (NPC167400) | |
| NCT03510884 | An Efficacy and Safety Study of Alirocumab in Children and Adolescents With Heterozygous Familial Hypercholesterolemia | Disorder of lipid metabolism | Niacin (NPC167400) | |
| NCT01221402 | Effect of Extended-Release Niacin on Saphenous Vein Graft Atherosclerosis | atherosclerosis | Niacin (NPC167400) | |
| NCT00961636 | A Long-term Study of ERN/LRPT (Extended Release Niacin/Laropiprant [MK0524A]) in Patients With Dyslipidemia (0524A-102) | Disorder of lipid metabolism | Niacin (NPC167400) | |
| NCT00855257 | Assessment of Endothelial Vasomotricity After Treatment by Nicotinic Acid in Acute Coronary Syndrome | acute coronary syndrome | Niacin (NPC167400) | |
| NCT00943124 | MK0524B Bioequivalence Study (0524B-070) | Disorder of lipid metabolism | Niacin (NPC167400) | |
| NCT05452772 | 5-HT2A Agonist Psilocybin in the Treatment of Tobacco Use Disorder | nicotine dependence | Niacin (NPC167400) | |
| NCT00000512 | Familial Atherosclerosis Treatment Study | Myocardial Ischemia | Niacin (NPC167400) | |
| NCT00728910 | Combined Effects of Non-statin Treatments on Apolipoprotein A-I Up-Regulation (CENTAUR): A Feasibility Study | Disorder of lipid metabolism | Niacin (NPC167400) | |
| NCT00804843 | Merck Carotid Atherosclerosis Trial (MK-0000-111)(COMPLETED) | carotid atherosclerosis | Niacin (NPC167400) | |
| NCT04630964 | The Effect of Psilocybin on MDD Symptom Severity and Synaptic Density | major depressive disorder | Niacin (NPC167400) | |
| NCT02890992 | An 8-Week Dose-Finding Study to Evaluate the Efficacy and Safety of Alirocumab in Children and Adolescents With Heterozygous Familial Hypercholesterolemia | Disorder of lipid metabolism | Niacin (NPC167400) | |
| NCT03866174 | A Study of Psilocybin for Major Depressive Disorder (MDD) | major depressive disorder | Niacin (NPC167400) | |
| NCT00271817 | To Evaluate Ezetimibe/Simvastatin and Niacin (Extended Release Tablet) in Patients With Type IIa or Type IIb Hyperlipidemia (0653A-091)(COMPLETED) | Hypercholesterolemia | Niacin (NPC167400) | |
| NCT00062556 | Effect of Niacin ER/Lovastatin on Peak Walking Time & Claudication Onset Time in Patients With Intermittent Claudication | intermittent vascular claudication | Niacin (NPC167400) | |
| NCT00071266 | The Dose Response of Niacin ER/Lovastatin on Peak Walking Time (PWT) in Patients With Intermittent Claudication - TROPIC | intermittent vascular claudication | Niacin (NPC167400) |
❱❱❱ Associated Human Diseases and Detailed Association Evidence
How do we define the Plant-Targeted Human Disease Association?
Associated human diseases of an individual plant are summurized based on FOUR types of association evidence, these include:
❶ Association by Therapeutic Target: Bioactive protein targets of the plant were defined in "Molecular Targets" section, target-disease associations collected from TTD database were subsequently used to build the associations between the plant and its targeted human diseases.
❷ Association by Disease Gene Reversion: Plant and a specific disease will be associated when >= 1 plant target gene overlaped with disease's DEGs.
❸ Association by Clinical Trials of Plant: Plant and a specific disease will be associated when >= 1 clinical trial (the plant is the intervetion) can be matched in ClinicalTrials.gov database.
❹ Association by Clinical Trials of Plant Ingredients: Plant and a specific disease will be associated when >= 1 clinical trial (the plant ingredient is the intervetion) can be matched in ClinicalTrials.gov database.
Associated Disease of the Plant |
Association Type & Detailed Evidence |
|---|---|
HyperlipidemiaDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5C80 |
NCT00384293,NCT00536510,NCT00376584,NCT00652431,NCT01583647,NCT00203476,NCT00378833
|
Type 2 diabetes mellitusDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A11 |
NCT01368328,NCT00618995,NCT00485758,NCT03540758,NCT03685773
|
Coronary atherosclerosisDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA80 |
NCT00120289,NCT00127218,NCT00397657,NCT01221402,NCT00150722
|
Ischaemic heart diseases, unspecifiedDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA6Z |
NCT00000553,NCT00000512,NCT00000482,NCT00000599,NCT00000461
|
Mixed hyperlipidaemiaDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5C80.2 |
NCT00289900,NCT00269217,NCT00269204
|
Cerebral ischaemic strokeDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8B11 |
HCAR2
NCT00796887 |
Nicotine use disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6C4A |
CHRNA4
NCT05452772 |
FlushingDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: ME64.4 |
NCT00533611,NCT00536237
|
NADisease Category: NADisease ICD-11 Code: NA |
NCT00062556,NCT00071266
|
Myocardial infarctionDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA41-BA43 |
NCT00120289,NCT00000482
|
Heart failureDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BD10-BD1Z |
NCT01005303,NCT04703361
|
Human immunodeficiency virus disease without mention of associated disease or condition, clinical stage unspecifiedDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1C62.Z |
NCT00152893,NCT02018965
|
Single episode depressive disorder, severe, without psychotic symptomsDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6A70.3 |
NCT03866174,NCT04630964
|
Retinal vascular occlusionDisease Category: 09.Diseases of the visual systemDisease ICD-11 Code: 9B74 |
NCT00493064,NCT00500045
|
Carcinosarcoma of uterusDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C76.43 |
HCAR2,CHRNA4
|
Inborn lipid metabolism errorDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5C52 |
HCAR2
NCT00111891,NCT00943124,NCT00903617,NCT00961636,NCT03510884,NCT00626392,NCT02890992,NCT00664287,NCT00630877,NCT00728910,NCT01104519,NCT01984073,NCT00376584,NCT01071291,NCT00944645 |
HypercholesterolaemiaDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5C80.0 |
NCT00271817,NCT01321034,NCT00378833,NCT00082251,NCT00080275,NCT00269217,NCT01071525,NCT00652431,NCT00536510,NCT03510715,NCT00203476,NCT01054508,NCT00533312,NCT00769132
|
Digestive system diseaseDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DE2Z |
CHRNA4
|
Mood disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6A60-6E23 |
CHRNA4
|
Hypertensive crisisDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA03 |
CHRNA4
|
Chronic arterial occlusive diseaseDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BD4Z |
HCAR2
|
Irritable bowel syndromeDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DD91 |
CHRNA4
|
Hyper-lipoproteinaemiaDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5C80 |
HCAR2
|
Attention deficit hyperactivity disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6A05 |
CHRNA4
|
DepressionDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6A70-6A7Z |
CHRNA4
|
Alzheimer diseaseDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8A20 |
CHRNA4
|
Aneurysm/dissectionDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BD50 |
CHRNA4
|
DyslipidemiaDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5C80-5C81 |
HCAR2
|
HypotensionDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA20-BA21 |
CHRNA4
|
Cardiovascular diseaseDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA00-BE2Z |
HCAR2
|
Corneal diseaseDisease Category: 09.Diseases of the visual systemDisease ICD-11 Code: 9A76-9A78 |
CHRNA4
|
Metabolic disorderDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5C50-5D2Z |
NCT00300365
|
Atherosclerotic chronic arterial occlusive disease, unspecifiedDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BD40.Z |
NCT00804843
|
Ischaemic/haemorrhagic strokeDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8B20 |
NCT00120289
|
Human immunodeficiency virus type 1Disease Category: X.Extension CodesDisease ICD-11 Code: XN8LD |
NCT01426438
|
Schizophrenia or other primary psychotic disorders, unspecifiedDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6A2Z |
NCT01720095
|
Acute ischaemic heart disease, unspecifiedDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA4Z |
NCT00855257
|
Asymptomatic stenosis of intracranial or extracranial arteryDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BD55 |
NCT00000599
|
Obsessive-compulsive disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6B20 |
NCT03356483
|
Post-traumatic stress disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6B40 |
NCT03752918
|
Sickle-cell disorderDisease Category: 03.Diseases of the blood or blood-forming organsDisease ICD-11 Code: 3A51 |
NCT00508989
|
Aortic valve stenosisDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BB70 |
NCT02109614
|
Cerebrovascular diseases, unspecifiedDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8B2Z |
NCT00000599
|
HIV-infected patients with tuberculosisDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1B10-1B14 |
NCT00170404
|
Malignant lymphoma, not elsewhere classifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B33.5 |
NCT02702492
|
ObesityDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5B80-5B81 |
NCT01083329
|
Glioblastoma, primary, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH0MB1 |
NCT04677049
|
Unspecified malignant neoplasms of unspecified sitesDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D4Z |
NCT00957359
|
Crohn diseaseDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DD70 |
HCAR2
|
Coeliac diseaseDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DA95 |
HCAR2
|
Serous cystadenoma,borderline malignancy of ovaryDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C73.4 |
HCAR2
|
Inflammatory bowel diseases, unspecifiedDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DD7Z |
HCAR2
|
Idiopathic pulmonary fibrosisDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CB03.4 |
CHRNA4
|
West Nile virus infectionDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D46 |
HCAR2
|
Adenocarcinoma of pancreasDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C10.0 |
HCAR2
|
Mesothelioma of pleuraDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C26.0 |
HCAR2
|
Other specified malignant neoplasms ofcolonDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B90.Y |
HCAR2
|
Cytomegaloviral diseaseDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D82 |
CHRNA4
|
Malignant neoplasms of thyroid gland, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D10.Z |
HCAR2
|
Malignant neoplasms of corpus uteri, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C76.Z |
HCAR2
|
Chikungunya virus diseaseDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D40 |
CHRNA4
|
Malignant neoplasms of thymusDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C27 |
HCAR2
|
Invasive carcinoma of breastDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C61 |
CHRNA4
|
Malaria, unspecifiedDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1F4Z |
HCAR2
|
Glioblastoma of brainDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A00.00 |
HCAR2
|
Renal cell carcinoma, chromophobe typeDisease Category: X.Extension CodesDisease ICD-11 Code: XH6153 |
HCAR2
|
Ebola virus diseaseDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D60.01 |
HCAR2
|
Malignant neoplasms of adrenal glandDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D11 |
CHRNA4
|
Lyme borreliosisDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1C1G |
HCAR2
|

