Collective Molecular Activities of the Plant: Securinega Suffruticosa
Overview of Ingredients
36 All known Ingredients in Total
Unique ingredients have been isolated from this plant.Plant-Ingredients Associations were manually curated from publications or collected from other databases.
34 Ingredients with Acceptable Bioavailablity
Unique ingredients exhibit acceptable human oral bioavailablity, according to the criteria of SwissADME [PMID: 28256516] and HobPre [PMID: 34991690]. The criteria details:SwissADME: six descriptors are used by SwissADME to evaluate the oral bioavailability of a natural product:
☑ LIPO(Lipophility): -0.7 < XLOGP3 < +5.0
☑ SIZE: 150g/mol < MW < 500g/mol
☑ POLAR(Polarity): 20Ų < TPSA < 130Ų
☑ INSOLU(Insolubility): -6 < Log S (ESOL) < 0
☑ INSATU(Insaturation): 0.25 < Fraction Csp3 < 1
☑ FLEX(Flexibility): 0 < Num. rotatable bonds < 9
If 6 descriptors of a natural plant satisfy the above rules, it will be labeled high HOB.
HobPre: A natural plant ingredient with HobPre score >0.5 is labeled high human oral availability (HOB)
22 Ingredients with experimental-derived Activity
Unique ingredients have activity data available.Ingredient Structrual Cards
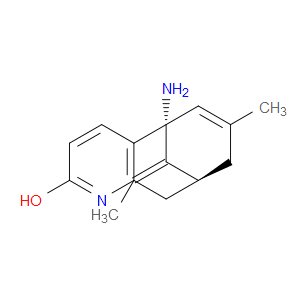
Ingredient ID: NPC78717
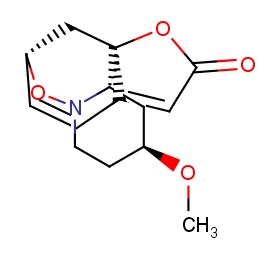
Ingredient ID: NPC75523
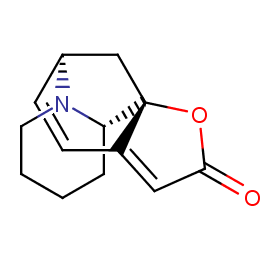
Ingredient ID: NPC72183
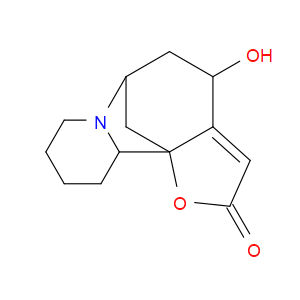
Ingredient ID: NPC71971

Ingredient ID: NPC62065
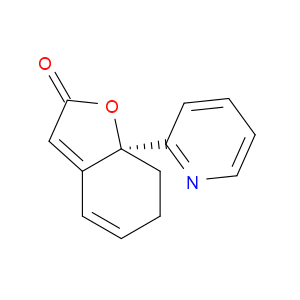
Ingredient ID: NPC479482
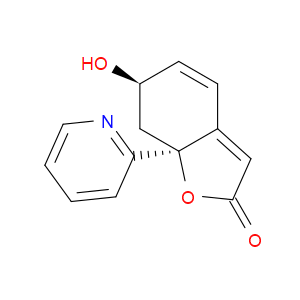
Ingredient ID: NPC479481
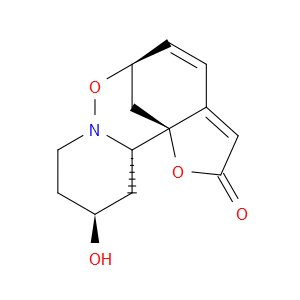
Ingredient ID: NPC479480

Ingredient ID: NPC479479
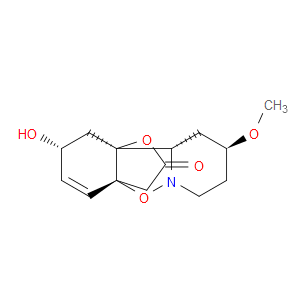
Ingredient ID: NPC479478

Ingredient ID: NPC479477
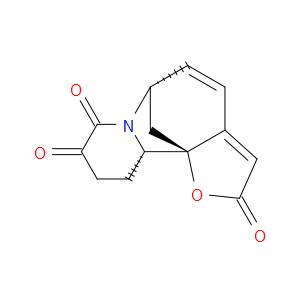
Ingredient ID: NPC479476
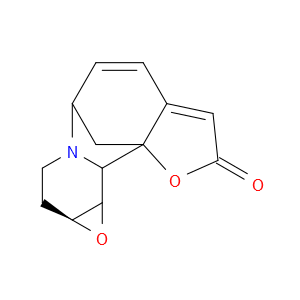
Ingredient ID: NPC479475
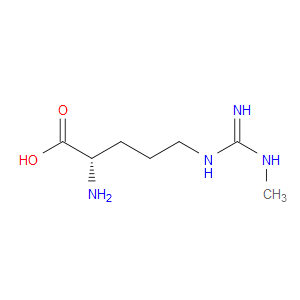
Ingredient ID: NPC38059
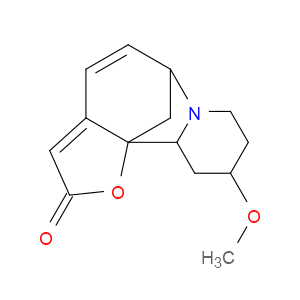
Ingredient ID: NPC329242
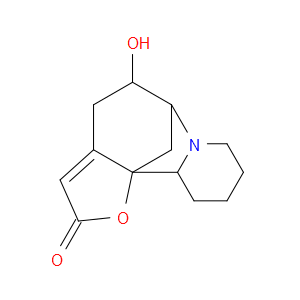
Ingredient ID: NPC324279
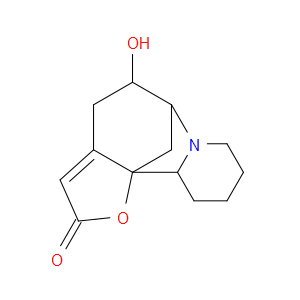
Ingredient ID: NPC318925
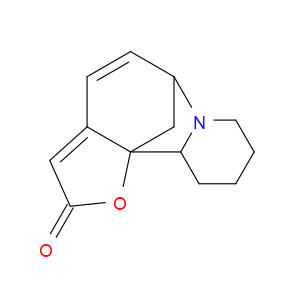
Ingredient ID: NPC317018
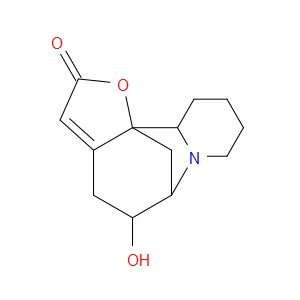
Ingredient ID: NPC312196
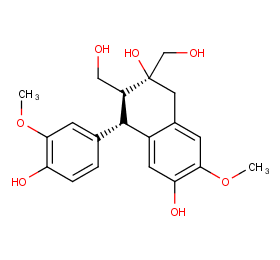
Ingredient ID: NPC311256
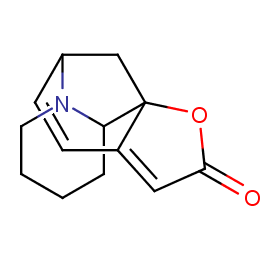
Ingredient ID: NPC299808
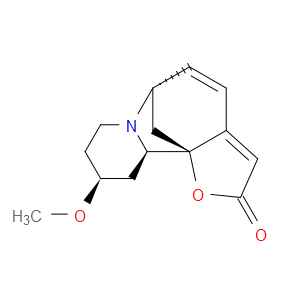
Ingredient ID: NPC298567
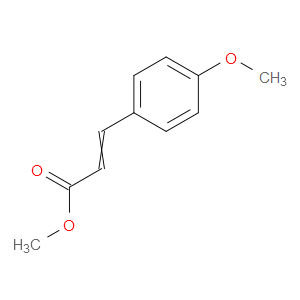
Ingredient ID: NPC28384

Ingredient ID: NPC279703
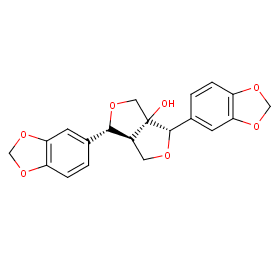
Ingredient ID: NPC266588

Ingredient ID: NPC263986
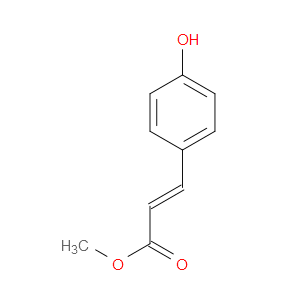
Ingredient ID: NPC253746
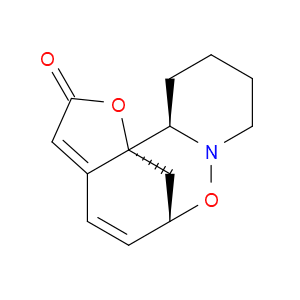
Ingredient ID: NPC249933
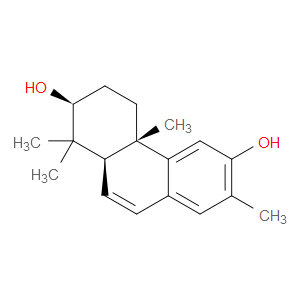
Ingredient ID: NPC22114
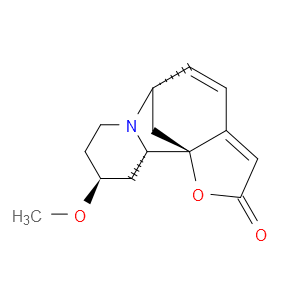
Ingredient ID: NPC219628
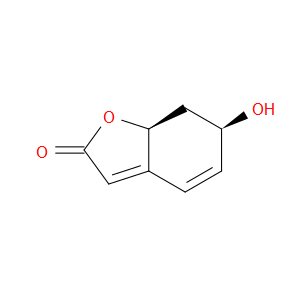
Ingredient ID: NPC194488
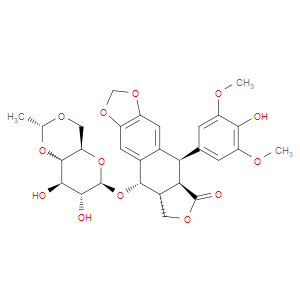
Ingredient ID: NPC185498
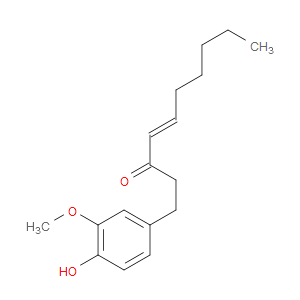
Ingredient ID: NPC164778
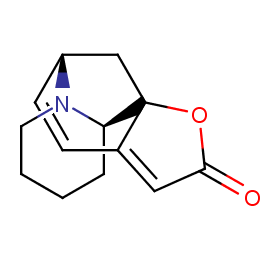
Ingredient ID: NPC118844
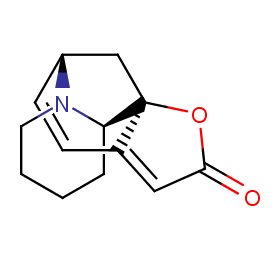
Ingredient ID: NPC110875
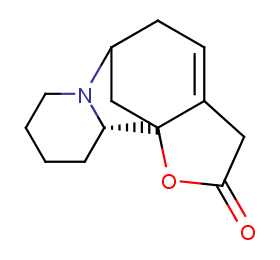
Ingredient ID: NPC109308
Classification of Human Proteins Collectively Targeted by the Plant
Detailed Information of Target Proteins
| Target Type | Protein Class | Gene ID | Protein Name | Uniprot ID | Target ChEMBL ID |
|---|---|---|---|---|---|
| Therapeutic Target | Enzyme | NOS1 | Nitric-oxide synthase, brain | P29475 | CHEMBL3568 |
| Therapeutic Target | Enzyme | NOS3 | Nitric-oxide synthase, endothelial | P29474 | CHEMBL4803 |
| Therapeutic Target | Histone acetyltransferase | NCOA3 | Nuclear receptor coactivator 3 | Q9Y6Q9 | CHEMBL1615382 |
| Therapeutic Target | Isomerase | TOP2B | DNA topoisomerase II beta | Q02880 | CHEMBL3396 |
| Therapeutic Target | Lyase | CA9 | Carbonic anhydrase IX | Q16790 | CHEMBL3594 |
| Therapeutic Target | Lyase | CA2 | Carbonic anhydrase II | P00918 | CHEMBL205 |
| Therapeutic Target | Lyase | CA12 | Carbonic anhydrase XII | O43570 | CHEMBL3242 |
| Therapeutic Target | Lyase | CA7 | Carbonic anhydrase VII | P43166 | CHEMBL2326 |
| Therapeutic Target | Lyase | CA1 | Carbonic anhydrase I | P00915 | CHEMBL261 |
| Therapeutic Target | Lyase | CA14 | Carbonic anhydrase XIV | Q9ULX7 | CHEMBL3510 |
| Therapeutic Target | Transient receptor potential channel | TRPV1 | Vanilloid receptor | Q8NER1 | CHEMBL4794 |
Clinical trials associated with plant from natural product (NP) & plant level:
| Clinical trials type | Number of clinical trials | |
|---|---|---|
| 978 | ||
| NCT ID | Title | Condition | Form in clinical use | Associated by plant or compound |
|---|---|---|---|---|
| NCT02408042 | Study of Pembrolizumab With Chemotherapy in Patients With Advanced Lymphoma (PembroHeme) | Hodgkins lymphoma;non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00003578 | High Dose Chemotherapy With or Without Bone Marrow Transplantation in Treating Patients With Intermediate- or High-Grade Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT02592876 | Treatment Study of Denintuzumab Mafodotin (SGN-CD19A) Plus RICE Versus RICE Alone for Diffuse Large B-Cell Lymphoma | diffuse large B-cell lymphoma;follicular lymphoma | Etoposide (NPC185498) | |
| NCT00002836 | Filgrastim Plus Chemotherapy Compared With Filgrastim Alone In Treating Women Undergoing Peripheral Stem Cell Transplantation For Breast Cancer | breast cancer | Etoposide (NPC185498) | |
| NCT00045162 | S0124: Cisplatin Combined With Irinotecan or Etoposide For Extensive-Stage Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT04380636 | Study of Pembrolizumab With Concurrent Chemoradiation Therapy Followed by Pembrolizumab With or Without Olaparib in Stage III Non-Small Cell Lung Cancer (NSCLC) (MK-7339-012/KEYLYNK-012) | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00521014 | GM-CSF and Rituximab After Autologous Stem Cell Transplant in Treating Patients With Relapsed or Refractory Follicular Non-Hodgkin Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00179920 | Chemotherapy Treatment for Children With Intraocular Germ-Line Retinoblastoma | retinoblastoma | Etoposide (NPC185498) | |
| NCT01134861 | Radiation Therapy Plus Chemotherapy in Treating Patients With Non-small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT00110110 | Combination Chemotherapy and Cyclosporine Followed by Focal Therapy for Bilateral Retinoblastoma | retinoblastoma | Etoposide (NPC185498) | |
| NCT04139304 | A Study of Daratumumab and Dose-Adjusted EPOCH in Plasmablastic Lymphoma | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT01775475 | Intravenous Chemotherapy or Oral Chemotherapy in Treating Patients With Previously Untreated Stage III-IV HIV-Associated Non-Hodgkin Lymphoma | Lymphoma, AIDS-Related | Etoposide (NPC185498) | |
| NCT00003957 | Combination Chemotherapy Plus Peripheral Stem Cell Transplantation in Treating Patients With Relapsed Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT01729845 | Decitabine Followed by Mitoxantrone Hydrochloride, Etoposide, and Cytarabine in Treating Patients With Relapsed or Refractory Acute Myeloid Leukemia or High-Risk Myelodysplastic Syndromes | acute myeloid leukemia;myelodysplastic syndrome | Etoposide (NPC185498) | |
| NCT00079105 | A Phase II Study VEPEMB In Patients With Hodgkin's Lymphoma Aged ≥ 60 Years | lymphoma | Etoposide (NPC185498) | |
| NCT03019640 | Umbilical Cord Blood NK Cells, Rituximab, High-Dose Chemotherapy, and Stem Cell Transplant in Treating Patients With Recurrent or Refractory B-Cell Non-Hodgkin's Lymphoma | diffuse large B-cell lymphoma;follicular lymphoma;Mantle cell lymphoma | Etoposide (NPC185498) | |
| NCT00275015 | Cyclophosphamide and Total Body Irradiation in Treating Patients Who Are Undergoing an Autologous Peripheral Stem Cell Transplant For Chronic Lymphocytic Leukemia | chronic lymphocytic leukemia | Etoposide (NPC185498) | |
| NCT03711305 | Study of Carboplatin Plus Etoposide With or Without SHR-1316 in Participants With Untreated Extensive-Stage (ES) Small Cell Lung Cancer (SCLC) | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00006122 | Gemtuzumab Ozogamicin With or Without Chemotherapy in Treating Older Patients With Acute Myeloid Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT00556127 | Rituximab in Addition to Chemotherapy With Autologous Stem Cell Transplantation as Treatment Diffuse Large B-Cell Lymphoma | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT00025103 | Chemotherapy Followed by Surgery and Radiation Therapy With or Without Stem Cell Transplant in Treating Patients With Relapsed or Refractory Wilms' Tumor or Clear Cell Sarcoma of the Kidney | kidney cancer | Etoposide (NPC185498) | |
| NCT03736616 | Acalabrutinib Plus RICE for Relapsed/Refractory DLBCL | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT00002715 | Combination Chemotherapy in Treating Patients With Advanced Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT04699838 | Chemo-Immunotherapy Followed by Durvalumab and Ceralasertib in Treatment Naïve Patients With Extensive Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT03992911 | Safety and Efficacy of FOLFSIM Plus Toripalimab in the Treatment of Advanced or Metastatic Neuroendocrine Carcinoma | neuroendocrine carcinoma | Etoposide (NPC185498) | |
| NCT00695409 | Yttrium-90 Ibritumomab Tiuxetan Plus High-Dose BEAM Followed By ASCT For Relapsed B-Cell Non-Hodgkin Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT02337712 | Once-daily Simultaneous Modulated Accelerated Thoracic Radiotherapy in Limited Small-cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00003113 | Oral Combination Chemotherapy in Treating Elderly Patients With Intermediate or High-Grade Non-Hodgkin's Lymphomas | lymphoma | Etoposide (NPC185498) | |
| NCT00417248 | Cisplatin/Etoposide/Radiotherapy Followed by Consolidation Sorafenib for Inoperable Stage III Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00002925 | Combination Chemotherapy Plus PSC 833 Followed by Interleukin-2 in Treating Patients With Acute Myelogenous Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT00876031 | Trial for Localised High-risk Rhabdomyosarcoma and Rhabdomyosarcoma-like Soft Tissue Sarcoma | soft tissue sarcoma | Etoposide (NPC185498) | |
| NCT01959698 | Carfilzomib, Rituximab, Ifosfamide, Carboplatin, and Etoposide in Treating Patients With Relapsed or Refractory Stage I-IV Diffuse Large B-cell Lymphoma | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT00439361 | Velcade Plus ICE for Patients With Relapsed Classical Hodgkin Lymphoma | Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00005867 | Combination Chemotherapy in Treating Patients With Aggressive Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00002802 | Therapy Based on Stage of Disease and Risk Assessment in Treating Children With Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT00005780 | Chemotherapy Plus Vaccination to Treat Mantle Cell Lymphoma | Mantle cell lymphoma | Etoposide (NPC185498) | |
| NCT00006258 | Combination Chemotherapy Followed by Radiation Therapy in Treating Patients With Medulloblastoma, Supratentorial Primitive Neuroectodermal Tumor, or Ependymoma | Central Nervous System Neoplasm | Etoposide (NPC185498) | |
| NCT03066778 | A Study of Pembrolizumab (MK-3475) in Combination With Etoposide/Platinum (Cisplatin or Carboplatin) for Participants With Extensive Stage Small Cell Lung Cancer (MK-3475-604/KEYNOTE-604) | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00002531 | Combination Chemotherapy in Treating Adults With Acute Lymphocytic Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT02025881 | Study of Sequential High-dose Chemotherapy in Children With High Risk Medulloblastoma | medulloblastoma | Etoposide (NPC185498) | |
| NCT00003606 | Combination Chemotherapy in Treating Patients With Extensive Stage Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT00186849 | Therapy for Children With Advanced Stage High Risk Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT00512252 | AMD3100 Plus Mitoxantrone, Etoposide and Cytarabine in Acute Myeloid Leukemia | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT00002714 | Combination Chemotherapy Plus Radiation Therapy in Treating Patients With Early-Stage Hodgkin's Disease | lymphoma | Etoposide (NPC185498) | |
| NCT00083551 | UARK 98-026 TT II: Multiple Myeloma Evaluating Anti-Angiogenesis With Thalidomide and Post-Transplant Consolidation Chemotherapy | multiple myeloma | Etoposide (NPC185498) | |
| NCT00002481 | Combination Chemotherapy and Radiation Therapy Plus Bone Marrow Transplantation in Treating Patients With Relapsed or Refractory Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00005964 | Comparison of Two Combination Chemotherapy Regimens in Treating Patients With Previously Untreated Aggressive Stage II, Stage III, or Stage IV Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00004903 | Combination Chemotherapy and Peripheral Stem Cell Transplantation in Treating Patients With Multiple Myeloma | multiple myeloma | Etoposide (NPC185498) | |
| NCT03769935 | Maintenance Therapy for Small-cell Lung Cancer | colorectal cancer | Etoposide (NPC185498) | |
| NCT02899728 | Olaparib, Cediranib Maleate, and Standard Chemotherapy in Treating Patients With Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT03723941 | Adjuvant Chemotherapy vs. Observation/Mitotane After Primary Surgical Resection of Localized Adrenocortical CarcInoma | adrenal cortex carcinoma | Etoposide (NPC185498) | |
| NCT00070200 | Induction Chemotherapy Using Cyclophosphamide and Topotecan in Treating Patients Who Are Undergoing Autologous Peripheral Stem Cell Transplantation for Newly Diagnosed or Progressive Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT00003421 | Combination Chemotherapy in Treating Patients With Advanced Hodgkin's Disease | lymphoma | Etoposide (NPC185498) | |
| NCT00006734 | Comparison of Combination Chemotherapy Regimens in Treating Patients With Ewing's Sarcoma or Neuroectodermal Tumor | sarcoma | Etoposide (NPC185498) | |
| NCT00017368 | Combination Chemotherapy Plus Peripheral Stem Cell Transplantation in Treating Children With Newly Diagnosed Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT03064867 | Venetoclax Plus R-ICE Chemotherapy for Relapsed/Refractory Diffuse Large B-Cell Lymphoma | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT00002829 | Bone Marrow Transplantation in Treating Patients With Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT02371161 | Phase II of High-dose Therapy in Elderly Patients With Relapsed Aggressive NHL or Resistant to First Line Therapy | non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT01492556 | Study of Etoposide in Treating Patients With Recurrent or Metastatic Breast Cancer | breast cancer | Etoposide (NPC185498) | |
| NCT00006487 | S0004: Combination Chemotherapy Plus Radiation Therapy in Treating Patients With Limited-stage Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT00554463 | G-CSF and Pegfilgrastim in Treating Neutropenia in Patients Undergoing Radiation Therapy and Chemotherapy for Limited Stage Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT00003815 | Chemotherapy and Radiation Therapy Plus Bone Marrow Transplantation in Treating Patients With Aggressive Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT03583710 | Mitotane With or Without Cisplatin and Etoposide After Surgery in Treating Patients With Stage I-III Adrenocortical Cancer With High Risk of Recurrence | adrenal cortex carcinoma | Etoposide (NPC185498) | |
| NCT01481272 | Ofatumumab With IVAC Salvage Chemotherapy in Diffuse Large B Cell Lymphoma Patients | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT02905110 | Methotrexate and Etoposide Infusions Into the Fourth Ventricle in Children With Recurrent Posterior Fossa Brain Tumors | brain neoplasm | Etoposide (NPC185498) | |
| NCT04923776 | Liver Directed RT + Chemo-immunotherapy for ES-SCLC | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT03944772 | Phase 2 Platform Study in Patients With Advanced Non-Small Lung Cancer Who Progressed on First-Line Osimertinib Therapy (ORCHARD) | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00379340 | Combination Chemotherapy With or Without Radiation Therapy in Treating Young Patients With Newly Diagnosed Stage III or Stage IV Wilms' Tumor | Wilms tumor | Etoposide (NPC185498) | |
| NCT00060385 | Combination Chemotherapy With or Without Etoposide in Treating Older Patients With Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT04063163 | A Randomized, Double-blind, Placebo Controlled Phase III Study to Investigate Efficacy and Safety of HLX10 + Chemotherapy (Carboplatin- Etoposide) in Patients With Extensive Stage Small Cell Lung Cancer (ES-SCLC) | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT01076231 | Proton Beam Radiation Therapy and Chemotherapy in Treating Patients With Stage III Non-Small Cell Lung Cancer That Can Be Removed By Surgery | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00807755 | Everolimus, Carboplatin, and Etoposide in Treating Patients With Small Cell Lung Cancer or Other Advanced Solid Tumors | lung cancer | Etoposide (NPC185498) | |
| NCT02553460 | Total Therapy for Infants With Acute Lymphoblastic Leukemia (ALL) I | acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT00927875 | A Study of BMS-833923 With Carboplatin and Etoposide Followed by BMS-833923 Alone in Subjects With Extensive-Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00577993 | Fludarabine, Mitoxantrone, and Dexamethasone (FND) Plus Rituximab for Lymphoma Patients | lymphoma | Etoposide (NPC185498) | |
| NCT00014313 | Chemotherapy in Treating Patients With Metastatic Ewing's Sarcoma or Primitive Neuroectodermal Tumor | sarcoma | Etoposide (NPC185498) | |
| NCT00253435 | N2001-02: I-MIBG With Intensive Chemotherapy and Autologous Stem Cell Rescue for High-Risk Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT00878254 | Rituximab and Combination Chemotherapy in Treating Patients With Previously Untreated Mantle Cell Lymphoma | Mantle cell lymphoma | Etoposide (NPC185498) | |
| NCT02631252 | Phase I Study of Mitoxantrone and Etoposide Combined With Hydroxychloroquine, for Relapsed Acute Myelogenous Leukemia | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT01064466 | Pharmacogenomics IND EXEMPT SNP Clinical Study - Etoposide and Single Nucleotide Polymorphisms | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT05403723 | Adaptive SBRT Plus Chemoimmunotherapy for ES-SCLC | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT01055301 | S0833, Bortezomib, Thalidomide, Lenalidomide, Combination Chemotherapy, and Autologous Stem Cell Transplant in Treating Patients With Newly Diagnosed Multiple Myeloma | multiple myeloma | Etoposide (NPC185498) | |
| NCT04981899 | A Study of Safety and Efficacy of PET-adapted Treatment With Nivolumab at the Fixed Dose 40 mg, Ifosfamide, Carboplatin, Etoposide (NICE-40) in Patients With Relapsed/Refractory Hodgkin Lymphoma | Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00187005 | Total Therapy Study XIV for Newly Diagnosed Patients With Acute Lymphoblastic Leukemia | acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT02471911 | KPT-330 Plus RICE for Relapsed/Refractory Aggressive B-Cell Lymphoma | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT00072527 | Cisplatin and Irinotecan Followed by Carboplatin, Etoposide, and Radiation Therapy in Treating Patients With Limited-Stage Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT02870907 | Adjuvant Treatment in Extensive Unilateral Retinoblastoma Primary Enucleated (RB SFCE 2009) | retinoblastoma | Etoposide (NPC185498) | |
| NCT00004086 | Radiolabeled Monoclonal Antibody, Combination Chemotherapy, and Peripheral Stem Cell Transplantation in Treating Patients With Recurrent or Refractory B-Cell Cancer | leukemia;lymphoma | Etoposide (NPC185498) | |
| NCT00968760 | CD19-specific T Cell Infusion in Patients With B-Lineage Lymphoid Malignancies | neoplasm of mature B-cells | Etoposide (NPC185498) | |
| NCT00354107 | Ifosfamide, Carboplatin, Etoposide, and SGN-30 in Treating Young Patients With Recurrent Anaplastic Large Cell Lymphoma | anaplastic large cell lymphoma | Etoposide (NPC185498) | |
| NCT00086944 | Oblimersen, Rituximab and Combination Chemotherapy in Treating Patients With Relapsed or Refractory Aggressive Non-Hodgkin's Lymphoma | diffuse large B-cell lymphoma;follicular lymphoma;Mantle cell lymphoma | Etoposide (NPC185498) | |
| NCT04624204 | Placebo-controlled, Study of Concurrent Chemoradiation Therapy With Pembrolizumab Followed by Pembrolizumab and Olaparib in Newly Diagnosed Treatment-Naïve Limited-Stage Small Cell Lung Cancer (LS-SCLC) (MK 7339-013/KEYLYNK-013) | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT04774380 | Study of Durvalumab in Combination With Platinum and Etoposide for the First Line Treatment of Patients With Extensive-stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00002865 | High-Intensity, Brief-Duration Chemotherapy in Treating Patients With Relapsed or Refractory Acute Lymphocytic Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT01154439 | Everolimus MICE-regimen in Treating Older Patients With Newly Diagnosed Acute Myeloid Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT02003222 | Combination Chemotherapy With or Without Blinatumomab in Treating Patients With Newly Diagnosed BCR-ABL-Negative B Lineage Acute Lymphoblastic Leukemia | acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT00591630 | Zevalin/BEAM/Rituximab vs BEAM/Rituximab With or Without Rituximab in Autologous Stem Cell Transplantation | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT05008224 | Study of Safety and Efficacy of Pembrolizumab and Chemotherapy in Participants With Newly Diagnosed Classical Hodgkin Lymphoma (cHL) (MK-3475-C11/KEYNOTE-C11) | classic Hodgkin lymphoma | Etoposide (NPC185498) | |
| NCT04665856 | Study of Atezolizumab Plus Carboplatin and Etoposide With or Without Tiragolumab in Participants With Untreated Extensive-Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT03072771 | Blinatumomab Consolidation Post Autologous Stem Cell Transplantation in Patients With Diffuse Large B-Cell Lymphoma (DLBCL) | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT00810017 | Phase 2 Study of Trastuzumab and Etoposide for Her2 Positive Breast Cancer | breast cancer | Etoposide (NPC185498) | |
| NCT03017326 | Paediatric Hepatic International Tumour Trial | Hepatoblastoma | Etoposide (NPC185498) | |
| NCT00274807 | Combination Chemotherapy in Treating Patients With Newly Diagnosed Acute Myeloid Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT04989283 | Testing the Addition of an Immunotherapy Drug, Atezolizumab, to the Usual Chemotherapy Treatment During Radiation Therapy for Superior Sulcus Non-small Cell Lung Cancer | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00002691 | Combination Chemotherapy in Treating Pediatric Patients With Stage III or IV Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00002816 | Combination Chemotherapy in Treating Children With Relapsed Acute Lymphoblastic Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT00145002 | A Study for Aggressive Adult T-cell Leukemia-lymphoma (ATLL) | adult T-cell leukemia/lymphoma | Etoposide (NPC185498) | |
| NCT00005987 | Filgrastim Compared With Sargramostim Plus Chemotherapy, Peripheral Stem Cell Transplantation, and Interferon Alfa in Treating Patients With Multiple Myeloma | multiple myeloma | Etoposide (NPC185498) | |
| NCT03100955 | A Study of Standard Treatment +/- Apatinib in Extensive Stage Small Cell Lung Cancer | colorectal cancer | Etoposide (NPC185498) | |
| NCT00568607 | Study on the Combination Regimen of Dexamethasone Ifosfamide Cisplatin Etoposide in Patients With NK/T Cell Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00667615 | Trial of Vorinostat in Combination With Cyclophosphamide, Etoposide, Prednisone and Rituximab for Elderly Patients With Relapsed Diffuse Large B-Cell Lymphoma (DLBCL) | Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT02631239 | MESA Versus ESA in the Treatment of Early Stage NK/T-cell Lymphoma | extranodal nasal NK/T cell lymphoma | Etoposide (NPC185498) | |
| NCT00072384 | Systemic Chemotherapy and Subtenon Carboplatin, and Local Ophthalmic Therapy in Children With Intraocular Retinoblastoma | retinoblastoma | Etoposide (NPC185498) | |
| NCT02934503 | Study of Pembrolizumab and Chemotherapy With or Without Radiation in Small Cell Lung Cancer (SCLC) | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT05294055 | Mecapegfilgrastim With Chemotherapy for Peripheral Blood Stem Cell Mobilization in MM and Lymphoma | multiple myeloma | Etoposide (NPC185498) | |
| NCT03850067 | A Safety, Tolerability and Preliminary Efficacy Evaluation of CC-90011 Given in Combination With Cisplatin and Etoposide in Subjects With First Line, Extensive Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00027898 | Bortezomib and Combination Chemotherapy in Treating Patients With Advanced Solid Tumors | neoplasm | Etoposide (NPC185498) | |
| NCT00002704 | Radiation Therapy and Chemotherapy in Treating Children With CNS Relapse From Acute Lymphoblastic Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT00042835 | Erlotinib and Radiation Therapy Plus Combination Chemotherapy in Treating Patients With Inoperable Stage III Non-Small Cell Lung Cancer | lung adenocarcinoma;squamous cell lung carcinoma | Etoposide (NPC185498) | |
| NCT05338541 | Tucidinostat Plus Etoposide in the Treatment of Neuroblastoma in Childhood. | neuroblastoma | Etoposide (NPC185498) | |
| NCT00025324 | Chemotherapy, Surgery, Radiation Therapy and Bone Marrow or Peripheral Stem Cell Transplantation in Treating Patients With Primary CNS Germ Cell Tumors | Central Nervous System Neoplasm | Etoposide (NPC185498) | |
| NCT00336024 | Combination Chemotherapy Followed By Peripheral Stem Cell Transplant in Treating Young Patients With Newly Diagnosed Supratentorial Primitive Neuroectodermal Tumors or High-Risk Medulloblastoma | medulloblastoma;primitive neuroectodermal tumor | Etoposide (NPC185498) | |
| NCT00002800 | Chemotherapy in Treating Patients With Newly Diagnosed Acute or Chronic Myelogenous Leukemia or Myelodysplastic Syndrome | neutropenia;leukemia;myelodysplastic syndrome | Etoposide (NPC185498) | |
| NCT00244946 | Combination Chemotherapy Followed By Autologous Stem Cell Transplant, and White Blood Cell Infusions in Treating Patients With Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT01525927 | Study of Chemotherapy Prior to Radiotherapy and Chemotherapy in Patients With HPV Associated Cancer of the Oral Cavity | neoplasm of oropharynx | Etoposide (NPC185498) | |
| NCT01589159 | A Phase Ⅱ Study of an All-Oral Combination of Low-dose Etoposide/Capecitabine in Patients With Metastatic Breast Cancer Previously Treated With Anthracyclines and/or Taxanes | breast cancer | Etoposide (NPC185498) | |
| NCT00878449 | A Study Evaluating the Safety of ABT-263 in Combination With Etoposide/Cisplatin in Subjects With Cancer | neoplasm | Etoposide (NPC185498) | |
| NCT00613223 | Ph I Dose Escalation Trial of Vandetanib in Combo w Etoposide for Malignant Gliomas | gliosarcoma | Etoposide (NPC185498) | |
| NCT00020943 | Chemotherapy and Rituximab With Peripheral Stem Cell Transplantation in Treating Patients With Mantle Cell Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00052299 | Chemotherapy With or Without Gemtuzumab Ozogamicin in Treating Older Patients With Acute Myeloid Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT03077828 | Pembrolizumab and Combination Chemotherapy in Treating Patients With Relapsed or Refractory Hodgkin Lymphoma | classic Hodgkin lymphoma | Etoposide (NPC185498) | |
| NCT01439568 | A Study of LY2510924 in Participants With Extensive-Stage Small Cell Lung Carcinoma | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00569985 | Gene Therapy-Treated Stem Cells in Treating Patients Undergoing Stem Cell Transplant for Intermediate-Grade or High-Grade AIDS-Related Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT01516567 | Intergroup Trial for Children or Adolescents With Primary Mediastinal Large B-Cell Lymphoma: DA-EPOCH-Rituximab Evaluation | lymphoma | Etoposide (NPC185498) | |
| NCT01356680 | HD17 for Intermediate Stage Hodgkin Lymphoma | Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00195871 | Safety and Efficacy of an Adult Acute Lymphoblastic Leukemia Chemotherapy for Adult Lymphoblastic Lymphoma | lymphoblastic lymphoma | Etoposide (NPC185498) | |
| NCT03742115 | Etoposide in the First-line Treatment of Adult EBV-HLH | hemophagocytic syndrome | Etoposide (NPC185498) | |
| NCT02116959 | Alternating Systemic Chemotherapy and Intra-Arterial Melphalan (IAM) Chemotherapy in Children With Intra-Ocular Retinoblastoma | retinoblastoma | Etoposide (NPC185498) | |
| NCT00025636 | Combination Chemotherapy and Peripheral Stem Cell Transplant in Treating Patients With Relapsed Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00049036 | Combination Chemotherapy and Rituximab in Treating Patients With HIV-Associated Stage I, Stage II, Stage III, or Stage IV Non-Hodgkin's Lymphoma | Lymphoma, AIDS-Related | Etoposide (NPC185498) | |
| NCT02219737 | Ibrutinib and Combination Chemotherapy in Treating Patients With Relapsed or Refractory Diffuse Large B-cell Lymphoma | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT03467360 | Inhibition of CArbonic Anhydrase in Combination With Platinum and Etoposide-based Radiochemotherapy in Patients With Localized Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00017225 | Chemotherapy and Radiation Therapy With or Without Peripheral Stem Cell Transplantation in Treating Patients With Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT00144989 | A Study Comparing Irinotecan and Cisplatin (IP) With Etoposide and Cisplatin (EP) Following EP/TRT for LD-SCLC | lung cancer | Etoposide (NPC185498) | |
| NCT00003812 | Chemotherapy Plus Radiation Therapy in Treating Patients With Limited-Stage Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT00958022 | Carboplatin and Etoposide Plus LBH589 for Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00004186 | Combination Chemotherapy in Treating Patients With Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT00304070 | Cisplatin-Based Chemotherapy and/or Surgery in Treating Young Patients With Adrenocortical Tumor | adrenal cortex carcinoma | Etoposide (NPC185498) | |
| NCT00973882 | Carboplatin-Etoposide Combination in Hormone-Resistant Prostate Cancers | prostate cancer | Etoposide (NPC185498) | |
| NCT00003783 | Combination Chemotherapy in Treating Children With Very High Risk Acute Lymphocytic Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT04422210 | A Study Evaluating The Safety, Tolerability, Pharmacokinetics, And Efficacy Of Venetoclax In Combination With Atezolizumab, Carboplatin, And Etoposide In Participants With Untreated Extensive-Stage Small Cell Lung Cancer (ES-SCLC). | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00618501 | Imatinib Mesylate and Combination Chemotherapy With or Without a Donor Stem Cell Transplant in Treating Patients With Newly Diagnosed Acute Lymphoblastic Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT00578864 | Protracted Etoposide During Induction Therapy for High Risk Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT00003402 | Peripheral Stem Cell Transplantation Plus Combination Chemotherapy in Treating Patients With Low-Grade Non-Hodgkin's Lymphoma or Chronic Lymphocytic Leukemia | leukemia;lymphoma | Etoposide (NPC185498) | |
| NCT00924209 | A Phase II Study of Neo-Adjuvant Gemcitabine, Cisplatin and Bevacizumab in Stage IIIA (N2) Non-Squamous Cell Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00016887 | Chemotherapy Followed by Radiation Therapy and Peripheral Stem Cell Transplant Compared With Chemotherapy Plus Interferon Alfa in Treating Patients With Stage III or Stage IV Mantle Cell Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT02227199 | Brentuximab Vedotin, Ifosfamide, Carboplatin, and Etoposide in Treating Patients With Relapsed or Refractory Hodgkin Lymphoma | Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT01300793 | Study of Standard-Dose Rituximab, Ifosfamide, Carboplatin and Etoposide | neoplasm of mature B-cells;non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT04712903 | Durvalumab Plus Chemotherapy in Untreated Patients With Extensive-Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT02405676 | BNHL-2015 for Children or Adolescents in China | neoplasm of mature B-cells | Etoposide (NPC185498) | |
| NCT02763579 | A Study of Carboplatin Plus Etoposide With or Without Atezolizumab in Participants With Untreated Extensive-Stage (ES) Small Cell Lung Cancer (SCLC) | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00003859 | Surgery Plus Radiation Therapy With or Without Chemotherapy in Treating Children With Primitive Neuroectodermal Tumors of the CNS | Central Nervous System Neoplasm | Etoposide (NPC185498) | |
| NCT03023046 | Etoposide, Prednisone, Vincristine Sulfate, Cyclophosphamide, and Doxorubicin in Treating Patients With Acute Lymphoblastic Leukemia or Lymphoblastic Lymphoma | acute lymphoblastic leukemia;lymphoblastic lymphoma | Etoposide (NPC185498) | |
| NCT01947062 | Metronomic Cyclophosphamide in Combination With Standard Chemotherapy for Squamous Cell Lung Carcinoma | squamous cell lung carcinoma | Etoposide (NPC185498) | |
| NCT05142865 | Camrelizumab Combined With Chemotherapy and Apatinib for Extrapulmonary Neuroendocrine Carcinomas | neuroendocrine carcinoma | Etoposide (NPC185498) | |
| NCT01329900 | Chemotherapy Plus Ofatumumab Followed by G-CSF for Mobilization of Peripheral Blood Stem Cells in Patients With Non-Hodgkin's Lymphomas | lymphoma | Etoposide (NPC185498) | |
| NCT00146120 | Risk-Adapted Therapy of Acute Myeloid Leukemia of Adults (18-60 Years) According to the Cytogenetic Result | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT00058292 | Radiolabeled Monoclonal Antibody Therapy and High-Dose Chemotherapy Followed By Autologous Peripheral Stem Cell Transplant in Treating Patients With Relapsed or Refractory Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00168896 | A Study for Small Cell Lung Cancer (SCLC) in Extensive Disease Stage | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT01722292 | A Study of LY2940680 in Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT02883049 | Combination Chemotherapy in Treating Young Patients With Newly Diagnosed High-Risk B Acute Lymphoblastic Leukemia and Ph-Like TKI Sensitive Mutations | acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT01197560 | Study of Lenalidomide to Evaluate Safety and Effectiveness in Patients With Diffuse Large B-Cell Lymphoma (DLBCL) | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT00137995 | R-ICE Versus R-DHAP in Patients Aged 18-65 With Relapse Diffuse Large B-cell Lymphoma | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT00712582 | Therapy for Patients With Untreated Age-Adjusted International Prognostic Index Low-Intermediate Risk, High-Intermediate Risk, or High Risk Diffuse Large B Cell Lymphoma | non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT05270057 | Loncastuximab Tesirine in Combination With DA-EPOCH-R in Patients With Previously Untreated Aggressive B-cell Lymphoid Malignancies | Burkitts lymphoma | Etoposide (NPC185498) | |
| NCT04177004 | Human Lysozyme Goat Milk for the Prevention of Graft Versus Host Disease in Patients With Blood Cancer Undergoing a Donor Stem Cell Transplant | hematopoietic and lymphoid cell neoplasm | Etoposide (NPC185498) | |
| NCT01390584 | Chemotherapy Based on PET Scan in Treating Patients With Stage I or Stage II Hodgkin Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT01294670 | Clinical Study of Vorinostat in Combination With Etoposide in Pediatric Patients < 21 Years at Diagnosis With Refractory Solid Tumors | sarcoma | Etoposide (NPC185498) | |
| NCT01117441 | International Collaborative Treatment Protocol For Children And Adolescents With Acute Lymphoblastic Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT00005978 | N99-01: Combination Chemotherapy, Radiation Therapy, and Stem Cell Transplantation in Treating Patients With Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT03117751 | Total Therapy XVII for Newly Diagnosed Patients With Acute Lymphoblastic Leukemia and Lymphoma | acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT03793478 | Safety and Efficacy of Quizartinib in Children and Young Adults With Acute Myeloid Leukemia (AML), a Cancer of the Blood | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT03349346 | Idelalisib With Rituximab, Ifosfamide, Carboplatin, Etoposide (RICE) in Children and Adolescents | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT00012051 | Chemotherapy and Peripheral Stem Cell Transplant With or Without Monoclonal Antibody Therapy in Treating Patients With Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT02773732 | Clinical Trial of Oral Ciprofloxacin and Etoposide in Subjects With Resistant Acute Myeloid Leukemia (AML)(UF-AML-CE-101) | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT01097057 | Rituximab, Combination Chemotherapy, Filgrastim (G-CSF), and Plerixafor in Treating Patients With Non-Hodgkin Lymphoma Undergoing Mobilization of Autologous Peripheral Blood Stem Cells | non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00483561 | Gefitinib and Etoposide in Treating Patients With Advanced Prostate Cancer That Did Not Respond to Hormone Therapy | prostate cancer | Etoposide (NPC185498) | |
| NCT04047862 | Study of Ociperlimab (BGB-A1217) in Combination With Tislelizumab in Advanced Solid Tumors | neoplasm | Etoposide (NPC185498) | |
| NCT03564704 | Precision Diagnosis Directing HDACi Chidamide Target Therapy for Adult T-LBL/ALL | lymphoblastic lymphoma | Etoposide (NPC185498) | |
| NCT00882076 | Clofarabine, Etoposide, and Mitoxantrone for Relapsed and Refractory Acute Leukemias | leukemia | Etoposide (NPC185498) | |
| NCT05092412 | Low-dose Radiotherapy Combined With Durvalumab, Chemotherapy(EP) in the Treatment of ES-SCLC | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT02400281 | Study of Crenolanib Combined With Chemotherapy in FLT3-mutated Acute Myeloid Leukemia Patients | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT00006455 | Combination Chemotherapy in Treating Children With Anaplastic Large Cell Lymphoma (ALCL 99) | lymphoma | Etoposide (NPC185498) | |
| NCT00002757 | TITLE:Less Intensive Therapy for Children With Non-Hodgkin's Lymphoma | leukemia;lymphoma | Etoposide (NPC185498) | |
| NCT00003621 | Combination Chemotherapy Plus Radiation Therapy in Treating Patients With Anaplastic Astrocytoma | Central Nervous System Neoplasm | Etoposide (NPC185498) | |
| NCT00049595 | Comparison of Two Combination Chemotherapy Regimens in Treating Patients With Stage III or Stage IV Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00057837 | Comparison of Two Combination Chemotherapy Regimens in Treating Patients With Extensive-Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT03570983 | A Trial Comparing Single Agent Melphalan to Carmustine, Etoposide, Cytarabine, and Melphalan (BEAM) as a Preparative Regimen for Patients With Multiple Myeloma Undergoing High Dose Therapy Followed by Autologous Stem Cell Reinfusion | multiple myeloma | Etoposide (NPC185498) | |
| NCT00151242 | Study on All-Trans Retinoic Acid, Induction and Consolidation Therapy, and Pegfilgrastim After Consolidation Therapy in Younger Patients With Newly Diagnosed Acute Myeloid Leukemia | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT02574728 | Sirolimus in Combination With Metronomic Chemotherapy in Children With Recurrent and/or Refractory Solid and CNS Tumors | cancer | Etoposide (NPC185498) | |
| NCT03041311 | Carboplatin, Etoposide, and Atezolizumab With or Without Trilaciclib (G1T28), a CDK4/6 Inhibitor, in Extensive-Stage SCLC | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00906945 | Chemosensitization With Plerixafor Plus G-CSF in Acute Myeloid Leukemia | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT00002642 | SWOG-9416: Chemotherapy, Radiation Therapy, and Surgery in Treating Patients With Stage III Non-small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT02703272 | A Safety and Efficacy Study of Ibrutinib in Pediatric and Young Adult Participants With Relapsed or Refractory Mature B-cell Non-Hodgkin Lymphoma | non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00264953 | HD11 for Intermediate Stages | lymphoma | Etoposide (NPC185498) | |
| NCT01746173 | CHOEP + High Dose Therapy + Auto SCT for T-Cell Lymphoma | T-cell non-Hodgkin lymphoma | Etoposide (NPC185498) | |
| NCT00002510 | Chemotherapy and Radiation Therapy Followed by Peripheral Stem Cell Transplantation in Treating Patients With Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT02768558 | Cisplatin and Etoposide Plus Radiation Followed By Nivolumab/Placebo For Locally Advanced NSCLC | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00084838 | Chemotherapy Combined With Radiation Therapy for Newly Diagnosed CNS AT/RT | Central Nervous System Neoplasm | Etoposide (NPC185498) | |
| NCT02794571 | Safety and Pharmacokinetics (PK) of Escalating Doses of Tiragolumab as a Single Agent and in Combination With Atezolizumab and/or Other Anti-Cancer Therapies in Locally Advanced or Metastatic Tumors | neoplasm | Etoposide (NPC185498) | |
| NCT00004128 | Combination Chemotherapy, Interleukin-2, and Peripheral Stem Cell Transplant in Treating Patients With Acute Myeloid Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT02987998 | Neoadjuvant Chemoradiation Plus Pembrolizumab Followed By Consolidation Pembrolizumab in NSCLC | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00008190 | Combination Chemotherapy Followed by Peripheral Stem Cell Transplantation and Interleukin-2 in Treating Patients With Acute Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT00049218 | Chemotherapy Followed By Vaccine Therapy in Treating Patients With Extensive-Stage Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT01857934 | Therapy for Children With Advanced Stage Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT02661503 | HD21 for Advanced Stages | classic Hodgkin lymphoma | Etoposide (NPC185498) | |
| NCT04996771 | Surufatinib Combined With Chemotherapy Plus Toripalimab or Not in the Treatment of Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT01415765 | MLN4924 Compared With MLN4924 Plus Chemotherapy for Large B-cell Lymphoma | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT00250718 | Study: Treatment of Relapsed Lymphoid Malignancies With an Anti-Angiogenic Approach | non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT01178645 | Study of Busulfan, Etoposide, Cytarabine, and Melphalan (BuEAM) Conditioning for Autologous Stem Cell Transplantation (ASCT) to Treat B Cell Lymphoma Except for Diffuse Large B Cell Lymphoma | non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00003558 | Chemotherapy in Treating Patients With Cancer of Unknown Primary Origin | carcinoma | Etoposide (NPC185498) | |
| NCT00002557 | Combination Chemotherapy in Patients With Advanced or Recurrent Mycosis Fungoides | lymphoma | Etoposide (NPC185498) | |
| NCT00003423 | Combination Chemotherapy in Treating Children With Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT01604863 | A Study of HGS1036 in Combination With Chemotherapy in Subjects With Advanced Solid Malignancies | cancer | Etoposide (NPC185498) | |
| NCT01458366 | Bendamustine in Combination With Ofatumumab, Carboplatin and Etoposide for Refractory or Relapsed Aggressive B-Cell Lymphomas | non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00098839 | Chemoimmunotherapy With Epratuzumab in Relapsed Acute Lymphoblastic Leukemia (ALL) | childhood acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT00002936 | Altretamine and Etoposide in Treating Patients With HIV-Related Cancer | lymphoma;sarcoma | Etoposide (NPC185498) | |
| NCT03991884 | Inotuzumab Ozogamicin and Chemotherapy in Treating Patients With Recurrent or Refractory B-cell Acute Lymphoblastic Leukemia | acute lymphoblastic leukemia;lymphoblastic lymphoma | Etoposide (NPC185498) | |
| NCT00811993 | A Study of R1507 in Combination With Multiple Standard Chemotherapy Treatments in Patients With Advanced Solid Tumors | neoplasm | Etoposide (NPC185498) | |
| NCT00630565 | Stem Cell Transplant in Treating Patients With Acute Myeloid Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT05091567 | A Phase III, Open-Label Study of Maintenance Lurbinectedin in Combination With Atezolizumab Compared With Atezolizumab in Participants With Extensive-Stage Small-Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT03504410 | Efficacy/Safety of CPI-613 in Combination With HD Cyt. and Mito. vs HD Cyt. and Mito. in Older Patients With R/R AML | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT00003631 | Chemotherapy Plus Radiation Therapy in Treating Patients With Refractory or Relapsed Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00000660 | Phase I Study of Weekly Oral VP-16 for AIDS-Associated Kaposi's Sarcoma | Kaposi's sarcoma | Etoposide (NPC185498) | |
| NCT00187161 | Treatment of Burkitt Lymphoma/Leukemia and B Large Cell NHL | acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT04878016 | A Study of Carboplatin Plus Etoposide With or Without ZKAB001 (Anti-PD-L1 Antibody) in Patients With ES-SCLC | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00007852 | Rituximab and Combination Chemotherapy Followed by Bone Marrow or Peripheral Stem Cell Transplantation in Treating Patients With Relapsed or Refractory Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00392886 | Combination Chemotherapy With or Without Etoposide Followed By an Autologous Stem Cell Transplant in Treating Young Patients With Previously Untreated Malignant Brain Tumors | Central Nervous System Neoplasm | Etoposide (NPC185498) | |
| NCT00003158 | S9712: Radiation Therapy and Combination Chemotherapy in Treating Patients With Stage III Non-small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT00002638 | Combination Chemotherapy Followed by Peripheral Stem Cell Transplantation in Treating Children With Relapsed Acute Lymphocytic Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT04256421 | A Study of Atezolizumab Plus Carboplatin and Etoposide With or Without Tiragolumab in Patients With Untreated Extensive-Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00003625 | Combination Chemotherapy Plus Radiation Therapy in Treating Patients With Newly Diagnosed Brain Stem Glioma | brain neoplasm | Etoposide (NPC185498) | |
| NCT03792815 | Busulfan, Melphalan and Etoposide as Conditioning for Autologous Transplantation for Non-Hodgkin's Lymphoma (NHL) | non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00018954 | Chemotherapy in Treating Patients With Acute Lymphoblastic Leukemia and Diffuse Non-Hodgkin's Lymphoma | leukemia;lymphoma | Etoposide (NPC185498) | |
| NCT00567567 | Comparing Two Different Myeloablation Therapies in Treating Young Patients Who Are Undergoing a Stem Cell Transplant for High-Risk Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT00369317 | Combination Chemotherapy in Treating Young Patients With Down Syndrome and Acute Myeloid Leukemia or Myelodysplastic Syndromes | acute basophilic leukemia;acute erythroleukemia;acute megakaryoblastic leukaemia;acute monocytic leukemia;acute myeloblastic leukemia without maturation;acute myelomonocytic leukemia;myelodysplastic syndrome | Etoposide (NPC185498) | |
| NCT00003996 | Combination Chemotherapy Plus Radiation Therapy in Treating Patients With Newly Diagnosed Glioblastoma Multiforme | Central Nervous System Neoplasm | Etoposide (NPC185498) | |
| NCT05354700 | A Clinical Study to Evaluate the Efficacy and Safety of HLX07 Combination Therapy in Patients With Extensive Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT01563601 | Efficacy and Safety of Obatoclax Mesylate in Combination With Carboplatin and Etoposide Compared With Carboplatin and Etoposide Alone in Chemotherapy-Naive Patients With Extensive-Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT01092182 | Phase II Study of Dose-Adjusted EPOCH-Rituximab in Adults With Untreated Burkitt Lymphoma and c-MYC+ Diffuse Large B-Cell Lymphoma | Burkitts lymphoma;diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT00061893 | Vinblastine, Celecoxib, and Combination Chemotherapy in Treating Patients With Newly-Diagnosed Metastatic Ewing's Sarcoma Family of Tumors | sarcoma | Etoposide (NPC185498) | |
| NCT00812266 | Topotecan and Cisplatin Versus Etoposide and Carboplatin in 1st Line Treatment of Patients With Small Cell Lung Cancer and Extensive Disease (SCLC-ED) | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00499018 | Dose Dense Chemotherapy + Rituximab +/-Intensified High Dose Chemoimmunotherapy With Support of Peripheral Autologous Stem Cell in Diffuse Large B-Cell Lymphoma | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT02632708 | Safety Study of AG-120 or AG-221 in Combination With Induction and Consolidation Therapy in Participants With Newly Diagnosed Acute Myeloid Leukemia (AML) With an IDH1 and/or IDH2 Mutation | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT01987232 | Phase 1b/2 Study of Carfilzomib, Carboplatin, and Etoposide in Patients With Previously Untreated Extensive Stage Small-cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00009815 | Combination Chemotherapy in Treating Patients With Advanced Solid Tumors | neoplasm | Etoposide (NPC185498) | |
| NCT01063439 | BuEAM Conditioning for Autologous Stem Cell Transplantation (ASCT) to Treat Diffuse Large B Cell Lymphoma (DLCBL) | lymphoma | Etoposide (NPC185498) | |
| NCT05058651 | Evaluating the Addition of the Immunotherapy Drug Atezolizumab to Standard Chemotherapy Treatment for Advanced or Metastatic Neuroendocrine Carcinomas That Originate Outside the Lung | small cell carcinoma | Etoposide (NPC185498) | |
| NCT00180908 | Comparison of High-Dose Methotrexate (HDM) Plus Doxorubicin to HDM Plus Etoposide-Ifosfamide in Osteosarcoma Children | osteosarcoma | Etoposide (NPC185498) | |
| NCT04203511 | INCMGA00012 in Combination With Chemoradiation in Participants With Stage III Non-Small Cell Lung Cancer (POD1UM-301) | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT03385096 | Mel vs BUCY+VP-16 Conditioning Regimen for MM Undergoing Auto-HSCT | multiple myeloma | Etoposide (NPC185498) | |
| NCT00003084 | Combination Chemotherapy With Ketoconazole in Treating Patients With Prostate Cancer | prostate cancer | Etoposide (NPC185498) | |
| NCT02595424 | Cisplatin, Carboplatin and Etoposide or Temozolomide and Capecitabine in Treating Patients With Neuroendocrine Carcinoma of the Gastrointestinal Tract or Pancreas That Is Metastatic or Cannot Be Removed by Surgery | pancreatic endocrine carcinoma | Etoposide (NPC185498) | |
| NCT00003215 | Combination Chemotherapy Plus Peripheral Stem Cell Transplantation in Treating Patients With Newly Diagnosed Aggressive Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00926640 | A Phase I Study of Belinostat in Combination With Cisplatin and Etoposide in Adults With Small Cell Lung Carcinoma and Other Advanced Cancers | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT01169636 | Panobinostat Plus Ifosfamide, Carboplatin, and Etoposide (ICE) Compared With ICE For Relapsed Hodgkin Lymphoma | Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00481832 | Autologous Followed by Non-myeloablative Allogeneic Transplantation for Non-Hodgkin's Lymphoma | non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00732498 | Rituximab, Yttrium Y 90 Ibritumomab Tiuxetan in Patients W/Relapsed Stage II, III, or IV Follicular NHL | lymphoma | Etoposide (NPC185498) | |
| NCT05367856 | Efficacy and Safety of Chidamide Combined With BEAM Pretreatment Regimen in Autologous Transplantation for T-cell Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00004898 | Radiation Therapy and Chemotherapy Plus Peripheral Stem Cell Transplantation in Treating Patients With High-Grade Lymphoma or Acute Lymphoblastic Leukemia | leukemia;lymphoma | Etoposide (NPC185498) | |
| NCT03382561 | Cisplatin/Carboplatin and Etoposide With or Without Nivolumab in Treating Patients With Extensive Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00749723 | Therapy Optimization Trial for the Treatment of Relapsed or Refractory Brain Tumors in Children | medulloblastoma | Etoposide (NPC185498) | |
| NCT03617432 | Chidamide Combined With CHOPE Regimen for Peripheral T-cell Lymphoma Patients | neoplasm | Etoposide (NPC185498) | |
| NCT00005612 | Combination Chemotherapy Followed by Peripheral Stem Cell Transplantation in Treating Patients With Ovarian Epithelial Cancer | ovarian cancer | Etoposide (NPC185498) | |
| NCT04778410 | Study of Magrolimab Combinations in Participants With Myeloid Malignancies | cancer | Etoposide (NPC185498) | |
| NCT00004224 | Combination Chemotherapy Followed by Radiation Therapy in Treating Children With Localized Ependymoma | Central Nervous System Neoplasm | Etoposide (NPC185498) | |
| NCT03553238 | Precision Diagnosis Directing HDACi Chidamide Target Therapy for Adult ETP-ALL | acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT04501718 | Apatinib Combined With Temozolomide and Etoposide Capsules in the Treatment of Recurrent Medulloblastoma in Children | medulloblastoma | Etoposide (NPC185498) | |
| NCT00003309 | Combination Chemotherapy Plus Radiation Therapy in Treating Adult Patients With Brain Cancer | Central Nervous System Neoplasm | Etoposide (NPC185498) | |
| NCT01840566 | High Dose Therapy and Autologous Stem Cell Transplantation Followed by Infusion of Chimeric Antigen Receptor (CAR) Modified T-Cells Directed Against CD19+ B-Cells for Relapsed and Refractory Aggressive B Cell Non-Hodgkin Lymphoma | non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00001563 | EPOCH Chemotherapy +/- IL-12 for Previously Untreated and EPOCH Plus Rituximab for Previously Treated Patients With AIDS-Associated Lymphoma | Lymphoma, AIDS-Related | Etoposide (NPC185498) | |
| NCT05142696 | A Safety Study of [177Lu]Lu-DOTA-TATE in Newly Diagnosed Extensive Stage Small Cell Lung Cancer (ES-SCLC) Patients in Combination With Carboplatin, Etoposide and Tislelizumab | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT01871766 | Risk-Adapted Focal Proton Beam Radiation and/or Surgery in Patients With Low, Intermediate and High Risk Rhabdomyosarcoma Receiving Standard or Intensified Chemotherapy | rhabdomyosarcoma | Etoposide (NPC185498) | |
| NCT00004176 | Amifostine, Chemotherapy, and Radiation Therapy in Treating Patients With Limited-Stage Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT04730999 | Efficacy and Safety Study of a New Therapeutic Strategy in the Treatment of Extended-Disease Small-Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00871013 | Trial for Patients Not Qualifying for TT4 and TT5 Protocols Because of Prior Therapy | multiple myeloma | Etoposide (NPC185498) | |
| NCT00039130 | Rituximab, Chemotherapy, and Filgrastim in Treating Patients With Burkitt's Lymphoma or Burkitt's Leukemia | leukemia;lymphoma | Etoposide (NPC185498) | |
| NCT04745689 | Study of AZD2811 + Durvalumab in ES-SCLC | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00005589 | Combination Chemotherapy Plus Peripheral Stem Cell Transplantation With or Without Rituximab in Treating Patients With Relapsed Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00720109 | Dasatinib and Combination Chemotherapy in Treating Young Patients With Newly Diagnosed Acute Lymphoblastic Leukemia | acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT00005090 | S9901 Combination Chemotherapy With or Without Peripheral Stem Cell Transplantation in Treating Men With Stage III or Stage IV Hodgkin's Disease | lymphoma | Etoposide (NPC185498) | |
| NCT00470223 | Combined Chemotherapy With or Without Zoledronic Acid for Patients With Osteosarcoma | sarcoma | Etoposide (NPC185498) | |
| NCT01569204 | Targeted BEACOPP Variants in Patients With Newly Diagnosed Advanced Classical Hodgkin Lymphoma | Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT03643276 | Treatment Protocol for Children and Adolescents With Acute Lymphoblastic Leukemia - AIEOP-BFM ALL 2017 | acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT00912392 | Etoposide-Carboplatin Alone or With Endostar for Extensive Disease Small Cell Lung Cancer (ED-SCLC) | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT02385110 | Alemtuzumab or Tocilizumab in Combination With Etoposide and Dexamethasone for the Treatment of Adult Patients With Hemophagocytic Lymphohistiocytosis | leukemia | Etoposide (NPC185498) | |
| NCT01059071 | Safety Study for Refractory or Relapsed Neuroblastoma With DFMO Alone and in Combination With Etoposide | neuroblastoma | Etoposide (NPC185498) | |
| NCT05298423 | Study of Pembrolizumab/Vibostolimab (MK-7684A) in Combination With Concurrent Chemoradiotherapy Followed by Pembrolizumab/Vibostolimab Versus Concurrent Chemoradiotherapy Followed by Durvalumab in Participants With Stage III Non-small Cell Lung Cancer (MK-7684A-006/KEYVIBE-006) | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT01435018 | Three Chemo Regimens as an Adjunct to ART for Treatment of Advanced AIDS-KS | HIV-1 infection | Etoposide (NPC185498) | |
| NCT00039195 | Chemotherapy and Rituximab With or Without Total-Body Irradiation and Peripheral Stem Cell Transplant in Treating Patients With Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT02298283 | Brentuximab Vedotin as Consolidation Treatment in Patients With Stage I/II HL and PET Positivity After 2 Cycles of ABVD | Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00024193 | Chemotherapy and Surgery Followed by Peripheral Stem Cell Transplantation in Treating Patients With Metastatic Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT00365755 | Combination Chemotherapy in Treating Young Patients Who Are Undergoing Surgery and an Autologous Bone Marrow Transplant for Disseminated Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT00003190 | Combination Chemotherapy With or Without Valspodar in Treating Patients With Previously Untreated Acute Myeloid Leukemia | acute erythroleukemia;acute megakaryoblastic leukaemia;acute monocytic leukemia;acute myeloblastic leukemia with maturation;acute myeloblastic leukemia without maturation;acute myelomonocytic leukemia | Etoposide (NPC185498) | |
| NCT00001339 | A Study of Combination Chemotherapy and Surgical Resection in the Treatment of Adrenocortical Carcinoma: Continuous Infusion Doxorubicin, Vincristine and Etoposide With Daily Mitotane Before and After Surgical Resection | carcinoma | Etoposide (NPC185498) | |
| NCT01411267 | AC220 for Children With Relapsed/Refractory ALL or AML | acute lymphoblastic leukemia;acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT04293562 | A Study to Compare Standard Chemotherapy to Therapy With CPX-351 and/or Gilteritinib for Patients With Newly Diagnosed AML With or Without FLT3 Mutations | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT02366663 | BEAM vs. 90-Yttrium Ibritumomab Tiuxetan (Zevalin®)/BEAM With ASCT for Relapsed DLBCL | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT00003967 | Topotecan Plus Etoposide in Treating Patients With Recurrent Ovarian, Peritoneal, or Fallopian Tube Cancer | fallopian tube cancer;peritoneum cancer;ovarian cancer | Etoposide (NPC185498) | |
| NCT01864109 | Irinotecan and Temozolomide in Combination With Existing High Dose Alkylator Based Chemotherapy for Treatment of Patients With Newly Diagnosed Ewing Sarcoma | Ewing sarcoma | Etoposide (NPC185498) | |
| NCT00916669 | A Randomized Study to Evaluate the Effect of Two Different Doses of Enoxaparin Sodium in Combination With Standard Chemotherapy in Patients With Stage Small Cell Lung Cancer (SCLC) | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT03135977 | Apatinib and Etoposide as the Maintenance Therapy in Extensive-stage Small Cell Lung Cancer After First-line Chemotherapy | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00274950 | Observation and/or Combination Chemotherapy After Surgery or Biopsy in Treating Young Patients With Extracranial Germ Cell Tumors | childhood germ cell tumor;ovarian cancer | Etoposide (NPC185498) | |
| NCT00003401 | Peripheral Stem Cell Transplantation Plus Combination Chemotherapy in Treating Patients With Multiple Myeloma | multiple myeloma | Etoposide (NPC185498) | |
| NCT01180322 | Trial Evaluating Induction Therapy With Idarubicin and Etoposide Plus Sequential or Concurrent Azacitidine and Maintenance Therapy With Azacitidine | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT04000295 | Apatinib and Etoposide Capsule Versus Weekly Paclitaxel in Patients With Platinum Resistant Ovarian Cancer | ovarian cancer | Etoposide (NPC185498) | |
| NCT00005022 | Radiation Therapy Plus Combination Chemotherapy in Treating Patients With Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT04101357 | Safety, Pharmacokinetics, Pharmacodynamics, and Preliminary Efficacy Trial of BNT411 | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT02289690 | Dose Escalation and Double-blind Study of Veliparib in Combination With Carboplatin and Etoposide in Treatment-naive Extensive Stage Disease Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00003941 | Combination Chemotherapy With or Without Peripheral Stem Cell Transplant in Treating Men With Previously Untreated Germ Cell Cancer | mediastinal cancer;testicular neoplasm | Etoposide (NPC185498) | |
| NCT00416676 | Combination Chemotherapy and Surgery With or Without Radiation Therapy in Treating Patients With Stage 2 or Stage 3 Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT00094497 | Trial in Locally Advanced and Metastatic Adrenocortical Carcinoma Treatment (FIRM-ACT) | carcinoma | Etoposide (NPC185498) | |
| NCT02018861 | A Phase 1/2, Open-Label, Dose Escalation, Safety and Tolerability Study of INCB050465 and Itacitinib in Subjects With Previously Treated B-Cell Malignancies (CITADEL-101) | cancer | Etoposide (NPC185498) | |
| NCT05221645 | Pembrolizumab in Combination With R-ICE Chemotherapy in Relapsed/Refractory Diffuse Large B-cell Lymphoma | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT02940990 | SBRT in Multi-metastatic NSCLC Patients Which Are Pan-negative for Driver Mutations | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00002610 | Chemotherapy With or Without Surgery, Radiation Therapy, or Stem Cell Transplantation in Treating Young Patients With Kidney Tumors | kidney cancer | Etoposide (NPC185498) | |
| NCT02359162 | Efficacy and Safety Study of P-Gemox vs.EPOCH as First-line Chemotherapy to Treat NK/T-cell Lymphoma With Early Stage | lymphoma | Etoposide (NPC185498) | |
| NCT00401609 | G-Step: Gemcitabine Based Small-cell Lung Cancer Treatment in Elderly Patients | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00006260 | Combination Chemotherapy in Treating Patients With Metastatic Breast Cancer | breast cancer | Etoposide (NPC185498) | |
| NCT00776802 | GCS-100LE in Combination With Etoposide and Dexamethasone in Relapsed or Refractory Diffuse Large B-Cell Lymphoma | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT00002598 | Combination Chemotherapy and Interferon Alfa in Treating Patients With Chronic Myelogenous Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT01251107 | Study Comparing ABVD vs BEACOPP in Advanced Hodgkin's Lymphoma | Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00002854 | High-Dose Combination Chemotherapy Plus Peripheral Stem Cell Transplantation in Treating Patients With Advanced Cancer | cancer | Etoposide (NPC185498) | |
| NCT01859741 | A Phase 1b/2 Study of OMP-59R5 (Tarextumab) in Combination With Etoposide and Platinum Therapy | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT01958372 | Radiation Therapy, Chemotherapy, and Soy Isoflavones in Treating Patients With Stage IIIA-IIIB Non-Small Cell Lung Cancer | lung adenocarcinoma | Etoposide (NPC185498) | |
| NCT00006708 | S0019 Combination Chemotherapy With or Without Rituximab in Treating Patients With Relapsed or Refractory Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00002926 | Combination Chemotherapy With or Without Peripheral Stem Cell Transplantation in Treating Patients With Myelodysplastic Syndrome or Acute Myelogenous Leukemia | leukemia;myelodysplastic syndrome | Etoposide (NPC185498) | |
| NCT02421939 | A Study of ASP2215 Versus Salvage Chemotherapy in Patients With Relapsed or Refractory Acute Myeloid Leukemia (AML) With FMS-like Tyrosine Kinase (FLT3) Mutation | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT00559104 | Combination Chemotherapy With or Without Total-Body Irradiation Followed By Stem Cell Transplant in Treating Patients With Non-Hodgkin Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00002675 | Chemotherapy in Treating Patients With Retinoblastoma | retinoblastoma | Etoposide (NPC185498) | |
| NCT00002550 | Chemotherapy Plus Radiation Therapy With or Without Surgery in Treating Patients With Stage IIIA Non-small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT00002590 | Combination Chemotherapy in Treating Children With Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT04833114 | Polatuzumab Vedotin Plus Rituximab, Ifosfamide, Carboplatin and Etoposide (Pola-R-ICE) Versus R-ICE Alone in Second Line Treatment of Diffuse Large B-cell Lymphoma (DLBCL) | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT02848183 | Optimal Treatment Strategy Based on for Pediatric AML | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT00002766 | Comparison of Two Combination Chemotherapy Regimens in Treating Adults With Previously Untreated Leukemia or Lymphoma | leukemia;lymphoma | Etoposide (NPC185498) | |
| NCT00020709 | Combination Chemotherapy and Radiation Therapy With or Without Gefitinib in Treating Patients With Stage III Non-Small Cell Lung Cancer That Cannot Be Removed By Surgery | lung adenocarcinoma;squamous cell lung carcinoma | Etoposide (NPC185498) | |
| NCT03794167 | BuCE Versus BuME as Conditioning Therapy in Non-Hodgkin's Lymphoma | non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT01565772 | Proton Radiation Therapy With Cisplatin and Etoposide Followed by Surgery in Stage III Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00193596 | Gemcitabine/Irinotecan/ZD1839 vs Paclitaxel/Carboplatin/Etoposide/ZD1839 in Carcinoma of Unknown Primary Site | neoplasm | Etoposide (NPC185498) | |
| NCT00430118 | Combination Chemotherapy Based on Risk of Relapse in Treating Young Patients With Acute Lymphoblastic Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT01573338 | Clinical Study to Evaluate the Maximum Tolerated Dose of BAY1000394 When Given Together With Chemotherapy and the Effectiveness of This Combination Treatment in Shrinking a Specific Type of Lung Tumors (Small Cell Lung Cancer) | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00002514 | Stem Cell Transplantation Compared With Standard Chemotherapy in Treating Patients With Acute Lymphoblastic Leukemia in First Remission | leukemia | Etoposide (NPC185498) | |
| NCT02819999 | A Study of Rovalpituzumab Tesirine (SC16LD6.5) in the Frontline Treatment of Patients With Extensive Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT01899066 | Efficacy Study of Lucentis in the Treatment of Retinoblastoma | retinoblastoma | Etoposide (NPC185498) | |
| NCT00004006 | Combination Chemotherapy, Radiation Therapy, and Bone Marrow Transplantation in Treating Patients With Retinoblastoma | retinoblastoma | Etoposide (NPC185498) | |
| NCT02329080 | New Combination of Chemoimmunotherapy for Systemic B-cell Lymphoma With Central Nervous System Involvement | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT00967369 | Combination Chemotherapy With or Without Bortezomib in Treating Patients With Classical Hodgkin Lymphoma That Has Returned or Does Not Respond to Prior Treatment. | classic Hodgkin lymphoma | Etoposide (NPC185498) | |
| NCT00571493 | VELCADE®-BEAM and Autologous Hematopoietic Stem Cell Transplantation for Non-Hodgkin's Lymphoma, or Mantle Cell Lymphoma | Mantle cell lymphoma | Etoposide (NPC185498) | |
| NCT05384015 | Study of Pembrolizumab, Lenvatinib and Chemotherapy Combination in First Line Extensive-stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00078988 | High-Dose Chemotherapy Plus Autologous Stem Cell Transplantation Compared With Intermediate-Dose Chemotherapy Plus Autologous Stem Cell Transplantation With or Without Isotretinoin in Treating Young Patients With Recurrent High-Grade Gliomas | brain neoplasm | Etoposide (NPC185498) | |
| NCT00881166 | Safety of MP470 in Combination With Standard-of-Care Chemotherapy Regimens to Treat Solid Tumors | cancer | Etoposide (NPC185498) | |
| NCT00776698 | A Study of Avastin (Bevacizumab) in Combination With Thoracic Radiation and Chemotherapy in Patients With Advanced Non-Squamous Non-Small Cell Lung Cancer. | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00398554 | Combination Chemotherapy in Treating Young Male Patients With Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00948090 | PK-directed Dose Adjustment of IV Busulfan Conditioning Regimen for Autologous Stem Cell Transplant in Lymphoma Patients | lymphoma | Etoposide (NPC185498) | |
| NCT00066742 | Tirapazamine Combined With Chemo and RT in Limited-Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT03367143 | Lenalidomide Plus ICE in the Treatment of Refractory and Relapsed DLBCL | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT00276731 | Combination Chemotherapy Followed By Surgery With or Without Radiation Therapy in Treating Young Patients With Stage II or Stage III Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT00002768 | Chemotherapy Plus Peripheral Stem Cell Transplantation in Treating Patients With Acute Myeloid Leukemia in Second Remission | leukemia | Etoposide (NPC185498) | |
| NCT03206671 | Treatment Protocol of the NHL-BFM and the NOPHO Study Groups for Mature Aggressive B-cell Lymphoma and Leukemia in Children and Adolescents | neoplasm of mature B-cells | Etoposide (NPC185498) | |
| NCT04660097 | Alotinib Plus Durvalumab-Platinum-Etoposide in First-line Treatment Extensive Small-cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00576979 | Intensity-Modulated Radiation Therapy, Etoposide, and Cyclophosphamide Followed By Donor Stem Cell Transplant in Treating Patients With Relapsed or Refractory Acute Lymphoblastic Leukemia or Acute Myeloid Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT00002517 | Combination Chemotherapy in Treating Children With Newly Diagnosed Acute Myeloid Leukemia or Myelodysplastic Syndrome | leukemia;myelodysplastic syndrome | Etoposide (NPC185498) | |
| NCT03669783 | Clinical Trial for Patients With a Stage IV Childhood Renal Tumor, Comparing Upfront Vincristine, Actinomycin-D and Doxorubicin (Standard Arm) With Upfront Vincristine, Carboplatin and Etoposide (Experimental Arm) | childhood kidney neoplasm | Etoposide (NPC185498) | |
| NCT00023998 | Chemotherapy With or Without Trastuzumab in Treating Patients With Metastatic Osteosarcoma | osteosarcoma | Etoposide (NPC185498) | |
| NCT00003114 | Combination Chemotherapy in Treating Patients With AIDS-Related Hodgkin's Disease | lymphoma | Etoposide (NPC185498) | |
| NCT00439556 | Bortezomib and Chemotherapy in Treating Participants With Lymphoid Malignancies Undergoing Stem Cell Transplant | lymphoma | Etoposide (NPC185498) | |
| NCT02372409 | Using MRI-Guided Laser Heat Ablation to Induce Disruption of the Peritumoral Blood Brain Barrier to Enhance Delivery and Efficacy of Treatment of Pediatric Brain Tumors | anaplastic astrocytoma;glioblastoma multiforme;oligoastrocytoma;oligodendroglioma;optic nerve glioblastoma;pilocytic astrocytoma | Etoposide (NPC185498) | |
| NCT00006461 | Combination Chemotherapy Followed by Second-Look Surgery and Radiation Therapy in Treating Children With Nonmetastatic Medulloblastoma or Primitive Neuroectodermal Tumor | medulloblastoma;primitive neuroectodermal tumor | Etoposide (NPC185498) | |
| NCT00682981 | A Phase I/II Study of Carboplatin and Etoposide With or Without Obatoclax in Extensive-stage Small Cell Lung Cancer (ES-SCLC) | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT03389087 | Study of Apatinib Plus Etoposide Capsule as the Therapy of Advanced Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00003937 | Combination Chemotherapy Plus Dexrazoxane in Treating Patients With Newly Diagnosed Nonmetastatic Osteosarcoma | sarcoma | Etoposide (NPC185498) | |
| NCT01411098 | Radiation Therapy, Cisplatin, and Etoposide in Treating Patients With Non-small Cell Lung Cancer That Cannot Be Removed by Surgery | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00002471 | Combination Chemotherapy in Treating Patients With Acute B-Lymphoblastic Leukemia or Non-Hodgkin's Lymphoma | leukemia;lymphoma | Etoposide (NPC185498) | |
| NCT01555541 | Intensive Consolidation and Stem Cell Mobilization Therapy Followed by Autologous Stem Cell Transplantation in High-risk Patients With Relapsed/Refractory Diffuse Large B-Cell Lymphoma | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT00002961 | Chemotherapy With or Without Total-Body Irradiation Prior to Bone Marrow Transplantation in Treating Children With Acute Lymphoblastic Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT00028769 | S0032, Combination Chemotherapy Plus Hormone Therapy in Treating Patients With Metastatic Prostate Cancer | prostate cancer | Etoposide (NPC185498) | |
| NCT00004906 | Combination Chemotherapy and Peripheral Stem Cell Transplantation in Treating Women With Metastatic Breast Cancer | breast cancer | Etoposide (NPC185498) | |
| NCT00002565 | Combination Chemotherapy in Treating Patients With Intermediate-Grade or Immunoblastic Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT02443077 | Ibrutinib Before and After Stem Cell Transplant in Treating Patients With Relapsed or Refractory Diffuse Large B-cell Lymphoma | neoplasm of mature B-cells | Etoposide (NPC185498) | |
| NCT00554788 | Combination Chemotherapy, Autologous Stem Cell Transplant, and/or Radiation Therapy in Treating Young Patients With Extraocular Retinoblastoma | retinoblastoma | Etoposide (NPC185498) | |
| NCT00003093 | Combination Chemotherapy in Treating Children With Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT04585893 | Safety and Efficacy of Rituximab for Treatment of Multicentric Castleman Disease in Malawi | Castleman disease | Etoposide (NPC185498) | |
| NCT00577629 | Chemotherapy With Monoclonal Antibody and Radioimmunotherapy for High-Risk B-Cell Non-Hodgkins Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00041015 | Combination Chemotherapy in Treating Patients With Extensive-Stage Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT01181271 | Tandem Auto-Allo Transplant for Lymphoma | chronic lymphocytic leukemia;diffuse large B-cell lymphoma;Hodgkins lymphoma;Mantle cell lymphoma | Etoposide (NPC185498) | |
| NCT00641381 | Carmustine, Etoposide, Cyclophosphamide, and Stem Cell Transplant in Treating Patients With HIV-Associated Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00083681 | DCEP in Combination With Thalidomide as Salvage Therapy for Post Transplantation Relapse | multiple myeloma | Etoposide (NPC185498) | |
| NCT05453500 | Chemotherapy (DA-EPOCH+/-R) and Targeted Therapy (Tafasitamab) for the Treatment of Newly-Diagnosed Philadelphia Chromosome Negative B Acute Lymphoblastic Leukemia | acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT02881086 | Optimization of Therapy in Adult Patients With Newly Diagnosed Acute Lymphoblastic Leukemia or Lymphoblastic Lymphoma by Individualised, Targeted and Intensified Treatment | acute lymphoblastic leukemia;lymphoblastic lymphoma | Etoposide (NPC185498) | |
| NCT01055314 | Temozolomide, Cixutumumab, and Combination Chemotherapy in Treating Patients With Metastatic Rhabdomyosarcoma | alveolar rhabdomyosarcoma;embryonal rhabdomyosarcoma | Etoposide (NPC185498) | |
| NCT00716066 | Autologous Stem Cell Transplant for Neurologic Autoimmune Diseases | chronic inflammatory demyelinating polyneuropathy;Myasthenia gravis;neuromyelitis optica;Opsoclonus-Myoclonus Syndrome;Stiff-Person syndrome | Etoposide (NPC185498) | |
| NCT03926624 | Trial of DFP-10917 vs Non-Intensive or Intensive Reinduction for AML Patients in 2nd/3rd/4th Salvage | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT00003399 | Peripheral Stem Cell Transplantation Plus Combination Chemotherapy in Treating Patients With Multiple Myeloma | multiple myeloma | Etoposide (NPC185498) | |
| NCT01178658 | Study of Busulfan, Etoposide, Cytarabine, and Melphalan (BuEAM) Conditioning for Autologous Stem Cell Transplantation (ASCT) to Treat T Cell or Natural Killer (NK) Cell Lymphoma | non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00005946 | Chemotherapy Plus Donor White Blood Cell Infusion in Treating Patients With Relapsed Hematologic Cancer Following Donor Peripheral Stem Cell Transplantation | leukemia;multiple myeloma;myelodysplastic syndrome | Etoposide (NPC185498) | |
| NCT00410423 | Study of VELCADE® With Mitoxantrone and Etoposide for Leukemias | leukemia | Etoposide (NPC185498) | |
| NCT00002868 | Interferon-alfa With or Without Chemotherapy and Peripheral Stem Cell Transplantation in Treating Patients With Newly Diagnosed Chronic Myelogenous Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT00083161 | Cisplatin, Etoposide, and Cyclophosphamide in Treating Patients With Extensive-Stage Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT04684017 | Anlotinib Plus Etoposide and Carboplatin as First-line Treatment for Extensive-stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT01451515 | NHL16: Study For Newly Diagnosed Patients With Acute Lymphoblastic Lymphoma | lymphoblastic lymphoma | Etoposide (NPC185498) | |
| NCT03407144 | Safety and Efficacy of Pembrolizumab (MK-3475) in Children and Young Adults With Classical Hodgkin Lymphoma (MK-3475-667/KEYNOTE-667) | Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00003727 | Chemotherapy and Peripheral Stem Cell Transplantation Followed by Immunotherapy in Treating Patients With Chronic Myelogenous Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT01914718 | Dose-adjusted EPOCH-R in MYC Positive DLBCL | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT04675697 | Anlotinib Plus Platinum-etoposide in First-line of Extensive-stage Small-cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT05076786 | Chidamide Plus Etoposide and Cisplatin/Carboplatin as First-line Treatment for Extrapulmonary Neuroendocrine Carcinoma | neuroendocrine carcinoma | Etoposide (NPC185498) | |
| NCT04372927 | ADMIRAL Trial: Adaptive Mediastinal Radiation With Chemo-Immunotherapy | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00864318 | Dose Intensification Study in Refractory Germ Cell Tumors With Relapse and Bad Prognosis | germ cell tumor | Etoposide (NPC185498) | |
| NCT04189094 | Chemoradiotherapy With or Without Sintilimab in Limited-stage SCLC | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT03059667 | Immunotherapy as Second-line in Patient With Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00006047 | Combination Therapy With Oral 9-Nitrocamptothecin & Oral Etoposide | neoplasm | Etoposide (NPC185498) | |
| NCT04924101 | Efficacy and Safety of Pembrolizumab Plus Investigational Agents in Combination With Chemotherapy as First-Line Treatment in Extensive-Stage Small Cell Lung Cancer (ES-SCLC) (MK-3475-B99/ KEYNOTE-B99) | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT03004287 | 2015-12: A Study Exploring the Use of Early and Late Consolidation/Maintenance Therapy | multiple myeloma | Etoposide (NPC185498) | |
| NCT03118466 | Mitoxantrone, Etoposide, and Cytarabine (MEC) Plus Lenalidomide for Relapsed or Refractory Acute Myeloid Leukemia | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT05309629 | Safety and Efficacy Study of First-line Treatment With QL1706 Plus Chemotherapy in Extensive-Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00682383 | Combined Modality Therapy With Growth Factor Support in Locally Advanced Non-small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00006045 | Combination Chemotherapy With or Without Monoclonal Antibody Therapy in Treating Patients With Refractory or Relapsed Acute Myelogenous Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT04326439 | AflacLL1901 (CHOA-AML) | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT00002827 | Chemotherapy Followed by Radiation Therapy in Treating Young Patients With Newly Diagnosed Hodgkin's Disease | lymphoma | Etoposide (NPC185498) | |
| NCT00124644 | Combination Chemotherapy and Tipifarnib in Treating Patients With Newly Diagnosed Acute Myeloid Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT00003262 | Combination Chemotherapy in Treating Patients With Hodgkin's Disease and HIV Infection | lymphoma | Etoposide (NPC185498) | |
| NCT00085098 | Radiation Therapy Compared With Chemotherapy and Radiation Therapy in Treating Patients With Newly Diagnosed Primary Central Nervous System (CNS) Germ Cell Tumor | brain neoplasm | Etoposide (NPC185498) | |
| NCT02356159 | Study of Palifermin (Kepivance) in Persons Undergoing Unrelated Donor Allogeneic Hematopoietic Cell Transplantation | Hodgkins lymphoma;leukemia;multiple myeloma;myelodysplastic syndrome;non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT03535961 | Apatinib Combined With Oral Etoposide in Metastatic HER2 Negative Breast Cancer | breast cancer | Etoposide (NPC185498) | |
| NCT01756989 | ANGIOCOMB Antiangiogenic Therapy for Pediatric Patients With Diffuse Brain Stem and Thalamic Tumors | glioma | Etoposide (NPC185498) | |
| NCT00288626 | High-Dose Immunosuppression and Autologous Transplantation for Multiple Sclerosis (HALT MS) Study | relapsing-remitting multiple sclerosis | Etoposide (NPC185498) | |
| NCT00025402 | Chemotherapy and Biological Therapy With or Without Bone Marrow or Peripheral Stem Cell Transplant in Treating Patients With Chronic Myelogenous Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT00109928 | S0350 Combination Chemotherapy in Treating Patients With Newly Diagnosed Stage II, Stage III, or Stage IV Peripheral T-Cell Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00349778 | High-Dose Sequential Therapy and Single Autologous Transplantation for Multiple Myeloma | multiple myeloma | Etoposide (NPC185498) | |
| NCT00002658 | Combination Chemotherapy, Biological Therapy, and Bone Marrow Transplantation in Treating Patients With Acute Myeloid Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT00458848 | Chemotherapy With or Without Imatinib and/or Peripheral Stem Cell Transplant in Acute Lymphoblastic Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT03432598 | Anti-PD-1 in Combination With Chemotherapy as First-Line Treatment to Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT04002947 | Acalabrutinib With DA-EPOCH-R or R-CHOP for People With Untreated Diffuse Large B-cell Lymphoma | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT00072280 | Surgery and/or Chemotherapy in Treating Children With Infantile, Congenital, or Childhood Fibrosarcoma | sarcoma | Etoposide (NPC185498) | |
| NCT01237808 | Study of Low-Dose Cytarabine and Etoposide With or Without All-Trans Retinoic Acid in Older Patients Not Eligible for Intensive Chemotherapy With Acute Myeloid Leukemia and NPM1 Mutation | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT00020566 | Combination Chemotherapy With or Without Peripheral Stem Cell Transplantation, Radiation Therapy, and/or Surgery in Treating Patients With Ewing's Sarcoma | sarcoma | Etoposide (NPC185498) | |
| NCT00176605 | Phase II Metronomic Dosing, Etoposide, Cyclophosphamide, D0 Prostate Cancer | prostate cancer | Etoposide (NPC185498) | |
| NCT00017251 | Combination Chemotherapy Plus Oblimersen in Treating Patients With Previously Untreated Extensive-Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00126191 | Intensive Chemotherapy and Rituximab in the Treatment of Burkitt Lymphoma | Burkitts lymphoma | Etoposide (NPC185498) | |
| NCT00002835 | Combination Chemotherapy in Treating Patients With Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00057811 | Rituximab, Rasburicase, and Combination Chemotherapy in Treating Young Patients With Newly Diagnosed Advanced B-Cell Leukemia or Lymphoma | anaplastic large cell lymphoma;Burkitts lymphoma;childhood acute lymphoblastic leukemia;diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT00053352 | Surgery and Combination Chemotherapy in Treating Children With Extracranial Germ Cell Tumors | Ovarian Embryonal Carcinoma;Ovarian Yolk Sac Tumor;teratoma;Testicular Choriocarcinoma;Testicular Yolk Sac Tumor | Etoposide (NPC185498) | |
| NCT00003597 | Colony-Stimulating Factors in Treating Children With Recurrent or Refractory Solid Tumors | cancer | Etoposide (NPC185498) | |
| NCT01096368 | Maintenance Chemotherapy or Observation Following Induction Chemotherapy and Radiation Therapy in Treating Patients With Newly Diagnosed Ependymoma | anaplastic ependymoma | Etoposide (NPC185498) | |
| NCT00006373 | Combination Chemotherapy Followed by Bone Marrow or Peripheral Stem Cell Transplantation in Treating Patients With Non-Hodgkin's Lymphoma or Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00742924 | Zoledronic Acid and Combination Chemotherapy in Treating Patients With Newly Diagnosed Metastatic Osteosarcoma | sarcoma | Etoposide (NPC185498) | |
| NCT00808899 | Neuroblastoma Protocol 2008: Therapy for Children With Advanced Stage High Risk Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT00003397 | Peripheral Stem Cell Transplantation Plus Combination Chemotherapy and Monoclonal Antibody Therapy in Treating Patients With Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT03980925 | Platinum-doublet Chemotherapy and Nivolumab for the Treatment of Subjects With Neuroendocrine Neoplasms (NENs) of the Gastroenteropancreatic (GEP) Tract or of Unknown (UK) Origin. | digestive system neuroendocrine neoplasm | Etoposide (NPC185498) | |
| NCT00720447 | Donor Stem Cell Transplant in Treating Patients With Mantle Cell Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00006695 | Monoclonal Antibody Therapy, Combination Chemotherapy, and Peripheral Stem Cell Transplant in Treating Patients With Relapsed or Refractory Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT04683198 | Camrelizumab Combined With Apatinib ,Carboplatin and Etoposide in Participants With ES-SCLC | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00025649 | Combination Chemotherapy Followed by Surgery and Peripheral Stem Cell or Bone Marrow Transplantation in Treating Infants With Newly Diagnosed Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT00363415 | Study of Pemetrexed and Carboplatin Compared With Etoposide Carboplatin to Treat Extensive-Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT01979536 | Brentuximab Vedotin or Crizotinib and Combination Chemotherapy in Treating Patients With Newly Diagnosed Stage II-IV Anaplastic Large Cell Lymphoma | anaplastic large cell lymphoma | Etoposide (NPC185498) | |
| NCT05484583 | Durvalumab Combined With Consolidation Radiotherapy After First-line Treatment in Extensive Stage Small Cell Lung Cancer With Oligometastases | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00047281 | Thalidomide, Celecoxib, and Combination Chemotherapy in Treating Patients With Relapsed or Refractory Malignant Glioma | Central Nervous System Neoplasm | Etoposide (NPC185498) | |
| NCT00002876 | Etoposide Plus Cisplatin in Treating Patients With Recurrent Ependymomas | Central Nervous System Neoplasm | Etoposide (NPC185498) | |
| NCT00003141 | Chemotherapy Plus Peripheral Stem Cell Transplantation in Treating Infants With Malignant Brain or Spinal Cord Tumors | brain neoplasm;neuroblastoma;sarcoma | Etoposide (NPC185498) | |
| NCT00079482 | Study of CEP-701 (Lestaurtinib) in Patients With Acute Myeloid Leukemia (AML) | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT00276809 | Combination Chemotherapy, Total-Body Irradiation, and Alemtuzumab in Treating Patients Undergoing an Autologous Stem Cell Transplant for Stage I, Stage II, Stage III, or Stage IV Chronic Lymphocytic Leukemia | chronic lymphocytic leukemia | Etoposide (NPC185498) | |
| NCT00351195 | Etoposide, Oxaliplatin and Capecitabine in Advanced Hepatocellular Carcinoma (HCC) | hepatocellular carcinoma | Etoposide (NPC185498) | |
| NCT03811002 | Testing the Addition of a New Immunotherapy Drug, Atezolizumab (MPDL3280A), to the Usual Chemoradiation (CRT) Therapy Treatment for Limited Stage Small Cell Lung Cancer (LS-SCLC) | lung cancer | Etoposide (NPC185498) | |
| NCT00032019 | Combination Chemotherapy and Monoclonal Antibody Therapy in Treating Patients With Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00041327 | Combination Chemotherapy Followed By Antiviral Therapy and Interferon Alfa in Treating Patients With HTLV-1-Related Adult T-Cell Leukemia/Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00660036 | Phase I/II Study of Mitoxantrone, Etoposide and Gemtuzumab Ozogamicin for Acute Myeloid Leukemia | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT00069238 | Campath-1H and EPOCH to Treat Non-Hodgkin's T- and NK-Cell Lymphomas | lymphoma | Etoposide (NPC185498) | |
| NCT00003632 | Combination Chemotherapy Plus Peripheral Stem Cell Transplantation in Treating Patients With Newly Diagnosed Central Nervous System Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00006669 | Rituximab Followed by Combination Chemotherapy in Treating Patients With Refractory or Recurrent Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00079040 | Cisplatin, Etoposide, and Bevacizumab in Treating Patients With Previously Untreated Extensive Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT02562365 | Adjuvant Chemotherapy of Three-step Regimen in Ovarian Cancer | ovarian cancer | Etoposide (NPC185498) | |
| NCT00002941 | Combination Chemotherapy Followed by Peripheral Stem Cell Transplantation in Treating Children With Recurrent or Refractory Hodgkin's or Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT03161054 | Metronomic Chemotherapy in Elderly Non-fit Patients With Aggressive B-Cell Lymphomas | non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00006012 | Combination Chemotherapy and Radiation Therapy in Treating Patients With Limited-Stage Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT02605421 | Myeloablative Consolidation Therapy and Tandem Autologous Stem Cell Rescue in Patients With High-Risk Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT00003860 | Combination Chemotherapy Plus Peripheral Stem Cell Transplantation in Treating Patients With Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT03168594 | Study to Compare Irinotecan Combined With Cisplatin (IP) Versus Etoposide Combined With Cisplatin (EP) in Advanced and Metastatic Gastrointestinal Pancreatic and Esophageal Neuroendocrine Carcinoma | neuroendocrine carcinoma | Etoposide (NPC185498) | |
| NCT04740021 | Study of LP002 in Combination With Chemotherapy for Patients With Extensive Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT03164057 | A Trial of Epigenetic Priming in Patients With Newly Diagnosed Acute Myeloid Leukemia | acute myeloid leukemia;myelodysplastic syndrome | Etoposide (NPC185498) | |
| NCT03923179 | Phase II Clinical Study of Pyrotinib Combined With Etoposide to Treat HER2-positive Advanced Breast Cancer | breast cancer | Etoposide (NPC185498) | |
| NCT00983944 | Rituximab and Combination Chemotherapy With or Without Bleomycin Sulfate in Treating Patients With Primary Mediastinal Large B-Cell Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00004054 | Hormone Therapy Plus Radiation Therapy With or Without Combination Chemotherapy in Treating Patients With Prostate Cancer | prostate cancer | Etoposide (NPC185498) | |
| NCT03418038 | Ascorbic Acid and Combination Chemotherapy for the Treatment of Relapsed or Refractory Lymphoma or Clonal Cytopenia of Undetermined Significance | Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00005613 | Peripheral Stem Cell Transplantation in Treating Patients With Non-Hodgkin's Lymphoma or Hodgkin's Disease | lymphoma | Etoposide (NPC185498) | |
| NCT04346914 | Study of ZKAB001 Combined With Carboplatin and Etoposide in the Extensive Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT03020030 | Treatment of Newly Diagnosed Acute Lymphoblastic Leukemia in Children and Adolescents | acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT00002567 | High-Dose Chemotherapy and Radiation Therapy Followed by Peripheral Stem Cell Transplantation in Treating Patients With Acute Leukemia in Remission | leukemia | Etoposide (NPC185498) | |
| NCT05124951 | The Efficacy of WVI in Patients With Localized Basal Ganglia Intracranial Germ Cell Tumors | central nervous system cancer | Etoposide (NPC185498) | |
| NCT02782754 | Chemotherapy Plus Reduced Radiotherapy in Intracranial Germinoma | germ cell tumor | Etoposide (NPC185498) | |
| NCT02631109 | L-DEP Regimen as a Salvage Therapy for Refractory Epstein Barr Virus-induced Hemophagocytic Lymphohistiocytosis | hemophagocytic syndrome | Etoposide (NPC185498) | |
| NCT00002912 | Combination Chemotherapy Plus PSC-833 in Treating Children With Refractory or Relapsed Acute Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT00151060 | Estramustine, Etoposide and Paclitaxel Treatment for Hormonally Responsive Adenocarcinoma of the Prostate | prostate cancer | Etoposide (NPC185498) | |
| NCT02142049 | Ibrutinib and Lenalidomide With Dose Adjusted EPOCH-R in Subjects With Relapsed/Refractory Diffuse Large B-cell Lymphoma | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT00426101 | Treatment Protocol for Hemophagocytic Lymphohistiocytosis 2004 | hemophagocytic syndrome | Etoposide (NPC185498) | |
| NCT00627354 | Mitoxantrone, Etoposide, and Vinorelbine As Second-Line Therapy in Treating Patients With Metastatic Prostate Cancer That Did Not Respond to Hormone Therapy | prostate cancer | Etoposide (NPC185498) | |
| NCT05348213 | Novel Targeted Drugs Combined With R-ICE Regimen in Relapsed and Refractory Diffuse Large B-cell Lymphoma | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT00002701 | Combination Chemotherapy With or Without Bone Marrow Transplantation in Treating Patients With Acute Promyelocytic Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT00400946 | Treatment of Acute Lymphoblastic Leukemia in Children | leukemia | Etoposide (NPC185498) | |
| NCT01237678 | A Study of IMGN901 for Patients With Advanced Solid Tumors and Extensive Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT03623087 | SIMPLE Chemotherapy for NK Lymphoma/Leukaemia | non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00002798 | Combination Chemotherapy With or Without Bone Marrow Transplantation in Treating Children With Acute Myelogenous Leukemia or Myelodysplastic Syndrome | acute erythroleukemia;acute megakaryoblastic leukaemia;acute monocytic leukemia;acute myeloblastic leukemia without maturation;acute myelomonocytic leukemia;chronic myelomonocytic leukemia;refractory anemia;refractory anemia with excess blasts | Etoposide (NPC185498) | |
| NCT02582697 | Accelerated v's Standard BEP Chemotherapy for Patients With Intermediate and Poor-risk Metastatic Germ Cell Tumours | germ cell tumor | Etoposide (NPC185498) | |
| NCT00143455 | Study Of Irinotecan Hydrochloride (Campto(R)) And Cisplatin Versus Etoposide And Cisplatin In Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00427791 | Rituximab to the Preparative Regimen of Etoposide and Total Body Irradiation in Acute Lymphoblastic Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT04234607 | A Study of TQB2450 or Placebo Combined With Anlotinib, Etoposide and Carboplatin Versus Etoposide and Carboplatin in Subjects With Extensive Small Cell Lung Cancer (ETER701) | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT03321890 | Chidamide Combined With PECM in Relapsed or Refractory Peripheral T-cell Lymphoma (PTCL) | unspecified peripheral T-cell lymphoma | Etoposide (NPC185498) | |
| NCT00544596 | R-(-)-Gossypol Acetic Acid, Cisplatin, and Etoposide in Treating Patients With Advanced Solid Tumors or Extensive Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00791154 | QUILT-2.013: A Trial of AMG 479 or AMG 102 in Combination With Platinum-based Chemotherapy as First-Line Treatment for Extensive Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT03213977 | R-DA-EDOCH Versus R-CEOP90, w/o Upfront Auto-HSCT in Young Patients With Intermediate/High-risk DLBCL | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT00114738 | EPOCH-R Chemotherapy Plus Bortezomib to Treat Mantle Cell Lymphoma | Mantle cell lymphoma | Etoposide (NPC185498) | |
| NCT00004905 | Combination Chemotherapy Plus Peripheral Stem Cell Transplantation in Treating Patients With Chronic Myelogenous Leukemia or Acute Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT02538926 | Etoposide, Prednisone, Vincristine Sulfate, Cyclophosphamide, and Doxorubicin Hydrochloride With Asparaginase in Treating Patients With Acute Lymphoblastic Leukemia or Lymphoblastic Lymphoma | T-lymphoblastic lymphoma;T-cell acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT02686346 | Brentuximab Vedotin in Refractory/Relapsed Hodgkin Lymphoma Treated by ICE | Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00632827 | Treatment of PTCL With Aggressive Induction Therapy Followed by Autologous SCT Using Denileukin Diftitox (Ontak) | unspecified peripheral T-cell lymphoma | Etoposide (NPC185498) | |
| NCT00073918 | Iodine I 131 Tositumomab, Etoposide and Cyclophosphamide Followed by Autologous Stem Cell Transplant in Treating Patients With Relapsed or Refractory Non-Hodgkin's Lymphoma | anaplastic large cell lymphoma;MALT lymphoma;Waldenstrom macroglobulinemia;Burkitts lymphoma;diffuse large B-cell lymphoma;follicular lymphoma;lymphoblastic lymphoma;Mantle cell lymphoma | Etoposide (NPC185498) | |
| NCT01949129 | Allogeneic Stem Cell Transplantation for Children and Adolescents With Acute Lymphoblastic Leukaemia | acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT00005834 | S9922 Combination Chemo Plus Filgrastim With or Without Thalidomide in Refractory Multiple Myeloma | multiple myeloma | Etoposide (NPC185498) | |
| NCT00021112 | Combination Chemotherapy Plus Radiation Therapy Followed by Surgery in Treating Patients With Stage IIIB Non-Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT03517137 | Very Early PET-response Adapted Targeted Therapy for Advanced Hodgkin Lymphoma: a Single -Arm Phase II Study | Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00002974 | Whole-Body Hyperthermia Plus Chemotherapy in Treating Patients With Advanced Sarcoma | sarcoma | Etoposide (NPC185498) | |
| NCT04728230 | Olaparib and Durvalumab With Carboplatin, Etoposide, and/or Radiation Therapy for the Treatment of Extensive-Stage Small Cell Lung Cancer, PRIO Trial | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT03018626 | R-ACVBP and DA-EPOCH-R in Patients With Non-GCB DLBCL | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT01030900 | Phase II Trial of Alemtuzumab (Campath) and Dose-Adjusted EPOCH-Rituximab (DA-EPOCH-R) in Relapsed or Refractory Diffuse Large B-Cell and Hodgkin Lymphomas | diffuse large B-cell lymphoma;Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT04221035 | High-Risk Neuroblastoma Study 2 of SIOP-Europa-Neuroblastoma (SIOPEN) | neuroblastoma | Etoposide (NPC185498) | |
| NCT02570542 | Study of the Impact of CD34+ Cell Dose on Absolute Lymphocyte Count Following High-Dose Therapy and Autologous Stem Cell Transplantation for Relapsed and Refractory Diffuse Large B-cell Lymphoma (DLBCL) | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT00920153 | Three Different Therapy Regimens in Treating Patients With Previously Untreated Hodgkin Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT02639650 | Study of Paclitaxel Plus Cisplatin as the First-line Chemotherapy in High Risk Gestational Trophoblastic Tumor | gestational trophoblastic neoplasm | Etoposide (NPC185498) | |
| NCT00162695 | Rhabdomyosarcoma and Malignant Soft Tissue Tumours of Childhood | rhabdomyosarcoma | Etoposide (NPC185498) | |
| NCT00135135 | Therapy for Children With Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT02228512 | Study of Pomalidomide Combined With Modified DA-EPOCH and Rituximab in KSHV-Associated Lymphomas | Castleman disease;diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT00354744 | High-Dose Combination Chemotherapy and Radiation Therapy in Treating Patients With Newly Diagnosed Metastatic Rhabdomyosarcoma or Ectomesenchymoma | sarcoma | Etoposide (NPC185498) | |
| NCT05384821 | Metronomic Chemotherapy in Wilms Tumor (MetroWilms-1906) | Wilms tumor | Etoposide (NPC185498) | |
| NCT01818908 | Trial of DA-EPOCH Regimen for NHL With HLH | hemophagocytic syndrome | Etoposide (NPC185498) | |
| NCT00613457 | Combination Chemotherapy Based on Risk of Relapse in Treating Young Patients With Acute Lymphoblastic Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT00007865 | Combination Chemotherapy Plus Rituximab in Treating Patients With Recurrent or Refractory Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT05116007 | Anti-PD-1 and VEGF Bispecific Antibody AK112 in Combination With Chemotherapy in Patients With ES-SCLC | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT01579929 | Combination of the Hedgehog Inhibitor, LDE225, With Etoposide and Cisplatin in the First-Line Treatment of Patients With Extensive Stage Small Cell Lung Cancer (ES-SCLC) | lung cancer | Etoposide (NPC185498) | |
| NCT02911142 | Lenalidomide Combined With Modified DA-EPOCH and Rituximab (EPOCH-R2) in Primary Effusion Lymphoma or KSHV-associated Large Cell Lymphoma | B-cell neoplasm | Etoposide (NPC185498) | |
| NCT00504140 | Recombinant Interferon Alpha and Etoposide in Relapsed Osteosarcoma | osteosarcoma | Etoposide (NPC185498) | |
| NCT00047138 | Chemotherapy Before and After Surgery in Treating Children With Wilm's Tumor | kidney cancer | Etoposide (NPC185498) | |
| NCT00002905 | Combination Chemotherapy in Treating Patients With HIV-Related Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00598169 | Bortezomib, Ifosfamide, Carboplatin, and Etoposide, With or Without Rituximab, in Treating Patients With Relapsed or Refractory AIDS-Related Non-Hodgkin Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT04542369 | Pilot Study of PD-1inhibitor (Tislelizumab) Plus Chemotherapy as Neoadjuvant Therapy for Limited-Stage Small-Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT02653196 | A Multi-Institutional Pilot Study of Allogeneic Hematopoietic Stem Cell Transplantation for Patients With Malignant Neuro-Epithelial and Other Solid Tumors | neuroepithelial neoplasm | Etoposide (NPC185498) | |
| NCT00002532 | Combination Chemotherapy in Treating Patients With Relapsed or Refractory Acute Lymphocytic Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT00387699 | Bevacizumab, Cisplatin, Etoposide, and Radiation Therapy in Treating Patients With Limited-Stage Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT00482053 | Phase 2 Poor Risk DLBCL of TLI and ATG Followed by Matched Allogeneic HT as Consolidation to Autologous HCT | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT03567642 | A Study of the Combination of Osimertinib, Platinum and Etoposide for Patients With Metastatic EGFR Mutant Lung Cancers | lung cancer | Etoposide (NPC185498) | |
| NCT03293173 | Biomarker Driven Intensified ChemoImmunotherapy With Early CNS Prophylaxis | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT02738346 | SCLC on the 2nd Line With Relapsed After Response to Chemotherapy GFPC 01-2013 | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00653068 | Combination Chemotherapy, Radiation Therapy, and an Autologous Peripheral Blood Stem Cell Transplant in Treating Young Patients With Atypical Teratoid/Rhabdoid Tumor of the Central Nervous System | neoplasm | Etoposide (NPC185498) | |
| NCT00041054 | Combination Chemotherapy and Exisulind in Treating Patients With Extensive-Stage Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT00453154 | Cisplatin or Carboplatin, and Etoposide With or Without Sunitinib Malate in Treating Patients With Extensive-Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT01014767 | Intercontinental Multidisciplinary Registry and Treatment Optimization Study for Choroid Plexus Tumors | brain cancer;choroid plexus neoplasm | Etoposide (NPC185498) | |
| NCT00003364 | Radiation Therapy Plus Combination Chemotherapy In Treating Patients With Limited Stage Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT00416832 | Combination Chemotherapy and Radiation Therapy in Treating Young Patients With Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00041210 | Combination Chemotherapy in Treating Patients With Previously Untreated Advanced Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00003728 | Combination Chemotherapy Plus Steroid Therapy in Treating Children With Acute Lymphoblastic Leukemia or Lymphoblastic Non-Hodgkin's Lymphoma | leukemia;lymphoma | Etoposide (NPC185498) | |
| NCT02949258 | Neoadjuvant Chemotherapy With SEEOX Regimen for Borrmann Type 4 Gastric Cancer | gastric cancer | Etoposide (NPC185498) | |
| NCT05458180 | CMOEP in the Treatment of Untreated Peripheral T-cell Lymphoma | unspecified peripheral T-cell lymphoma | Etoposide (NPC185498) | |
| NCT02063022 | Efficacy of Dose Intensification in Patients With Non-metastatic Ewing Sarcoma | Ewing sarcoma | Etoposide (NPC185498) | |
| NCT04154189 | A Study to Compare the Efficacy and Safety of Ifosfamide and Etoposide With or Without Lenvatinib in Children, Adolescents and Young Adults With Relapsed and Refractory Osteosarcoma | osteosarcoma | Etoposide (NPC185498) | |
| NCT00791947 | A Nordic Phase II Study of PTCL Based on Dose-intensive Induction and High-dose Consolidation With ASCT | unspecified peripheral T-cell lymphoma | Etoposide (NPC185498) | |
| NCT00350181 | Sirolimus & Mycophenolate Mofetil as GVHD Prophylaxis in Myeloablative, Matched Related Donor HCT | acute graft vs. host disease;leukemia;non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT02964013 | Study of Vibostolimab Alone and in Combination With Pembrolizumab in Advanced Solid Tumors (MK-7684-001) ( KEYVIBE-001) | neoplasm | Etoposide (NPC185498) | |
| NCT00387868 | Preoperative Treatment of Patients With High Risk Thymoma | Thymoma | Etoposide (NPC185498) | |
| NCT00191139 | Gemcitabine or Gemcitabine Plus Docetaxel After Cisplatin, Etoposide and Radiation in Non Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT04765098 | Tamoxifen Versus Etoposide After First Recurrence in GBM Patients | glioblastoma multiforme | Etoposide (NPC185498) | |
| NCT00002549 | Combination Chemotherapy Followed by Bone Marrow or Peripheral Stem Cell Transplantation in Treating Patients With Acute Myelogenous Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT01906814 | Adjuvant Chemotherapy for High-risk Retinoblastoma After Enucleation | retinoblastoma | Etoposide (NPC185498) | |
| NCT02262325 | Hypofractionated Boost Before Chemoradiation for Patients With Stage II-III Non-small Cell Lung Cancer Unsuitable for Surgery | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00022737 | Combination Chemotherapy With or Without Peripheral Stem Cell Transplant in Treating Children With Acute Lymphoblastic Leukemia | childhood acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT05001412 | Chemotherapy Combined With Camrelizumab and Apatinib in First-line Treatment of ES-SCLC | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00134030 | Combination Chemotherapy, PEG-Interferon Alfa-2b, and Surgery in Treating Patients With Osteosarcoma | osteosarcoma | Etoposide (NPC185498) | |
| NCT01733589 | Recombinant Human Endostatin Continued Pumping Into Vein Combining With CCRT in Unresectable Stage III NSCLC | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00242996 | Rituximab, Cyclophosphamide, and G-CSF Followed By Combination Chemotherapy in Treating Patients Who Are Undergoing Autologous Stem Cell Transplant Followed By Rituximab and GM-CSF for Refractory Diffuse Large B-Cell Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT02043587 | Chemotherapy With Liposomal Cytarabine CNS Prophylaxis for Adult Acute Lymphoblastic Leukemia & Lymphoblastic Lymphoma | acute lymphoblastic leukemia;lymphoblastic lymphoma | Etoposide (NPC185498) | |
| NCT00689169 | Targeted Intensification With ZBEAM and Autologous Stem Cell Transplantation in Patients With High-grade B-Cell Lymphoma | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT00100477 | Use of Topotecan in Patients With Refractory Acute Leukemia | leukemia;non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT04696575 | Lamivudine in Combination With Chemoimmunotherapy for the Treatment of Extensive Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT02660762 | Modified MRCUKALLⅫ/ECOGE2993 Regimen for ALL | leukemia | Etoposide (NPC185498) | |
| NCT05468489 | To Evaluate Efficacy and Safety of HLX10 + Chemotherapy (Carboplatin- Etoposide) in US Patients With ES-SCLC | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT02070458 | Ixazomib, Mitoxantrone Hydrochloride, Etoposide, and Intermediate-Dose Cytarabine in Relapsed or Refractory Acute Myeloid Leukemia | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT00613626 | Cisplatin + Etoposide +/- Concurrent ZD6474 in Previously Untreated Extensive Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00031590 | Low-Dose Radiation and Combination Chemotherapy Following Surgery in Children With Newly Diagnosed Medulloblastoma | medulloblastoma | Etoposide (NPC185498) | |
| NCT00052923 | Stem Cell Transplantation With or Without Rituximab in Treating Patients With Relapsed or Progressive B-Cell Diffuse Large Cell Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT02845882 | LBL-2016 for Children or Adolescents in China | lymphoblastic lymphoma | Etoposide (NPC185498) | |
| NCT00549848 | Total Therapy Study XVI for Newly Diagnosed Patients With Acute Lymphoblastic Leukemia | acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT00002649 | Interleukin-2 or Observation Following Radiation Therapy, Combination Chemotherapy, and Peripheral Stem Cell Transplantation in Treating Patients With Recurrent Non-Hodgkin's Lymphoma | Burkitts lymphoma;diffuse large B-cell lymphoma;follicular lymphoma | Etoposide (NPC185498) | |
| NCT02836639 | BEB Conditioning Regimen for Autologous Stem-cell Transplantation(ASCT) in Non-Hodgkin's Lymphoma | non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00255723 | Combination Chemotherapy and Radiation Therapy in Treating Patients Who Are Undergoing an Autologous Stem Cell Transplant for Relapsed or Refractory Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT04543890 | A Prospective Study Comparing Two Radiotherapy Dose/Fraction and Omitting CTVs of the Primary Tumor in Limited SCLC | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00025597 | Combination Chemotherapy Followed by Surgery in Treating Infants With Newly Diagnosed Neuroblastoma. | neuroblastoma | Etoposide (NPC185498) | |
| NCT04696939 | Combined Atezolizumab and Chemotherapy (Carboplatin Plus Etoposide) in Neoadjuvant Treating Limited-Stage Small Cell Lung Cancer Patients | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT02937116 | First in Human Study of IBI308 in Chinese Subjects With Advanced Solid Tumors | cancer | Etoposide (NPC185498) | |
| NCT00774046 | High-dose Cytarabine/Mitoxantrone Followed by Autotransplantation for t-MDS/t-AML | acute myeloid leukemia;myelodysplastic syndrome | Etoposide (NPC185498) | |
| NCT00539500 | Autologous Stem Cell Rescue With CD133+ Selected Cells in High-Risk Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT00202800 | Phase II Trial to Evaluate Gemcitabine and Etoposide for Locally Advanced or Metastatic Pancreatic Cancer | pancreatic carcinoma | Etoposide (NPC185498) | |
| NCT03839446 | Phase II Study of the Combination of Mitoxantrone, Etoposide and Gemtuzumab Ozogamicin (MEGO) for Patients With Acute Myeloid Leukemia Refractory to Initial Standard Induction Therapy | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT00723658 | S0629, Observation or Combination Chemotherapy, Bortezomib, Thalidomide, and Rituximab Followed By Two Autologous Peripheral Blood Stem Cell Transplants in Treating Patients With Waldenstrom Macroglobulinemia | lymphoma | Etoposide (NPC185498) | |
| NCT00193505 | Ifosfamide/Carboplatin/Etoposide/Rituxan Followed by Zevalin in Relapsed/Refractory Intermediate Grade B-Cell Lymphoma | non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT02722369 | STUDY 15 - Comparing Gemcitabine/Carboplatin and Hydroxychloroquine Versus Carboplatin/Etoposide Therapy Alone in Small Cell Lung Cancer (SCLC) | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT04469530 | Sirolimus in Combination With Metronomic Chemotherapy in Children With High-Risk Solid Tumors | neoplasm | Etoposide (NPC185498) | |
| NCT01258634 | A Study of Pre-Operative Treatment of Newly-Diagnosed, Surgically-Resectable Osteosarcoma With Doxorubicin, Ifosfamide, Etoposide, and Cisplatin With Early Metabolic Assessment of Response | osteosarcoma | Etoposide (NPC185498) | |
| NCT00058461 | Combination Chemotherapy and Rituximab in Treating Young Patients With Recurrent or Refractory Non-Hodgkin's Lymphoma or Acute Lymphoblastic Leukemia | Burkitts lymphoma;diffuse large B-cell lymphoma;lymphoblastic lymphoma | Etoposide (NPC185498) | |
| NCT00061919 | Carboplatin and Etoposide With or Without Thalidomide in Treating Patients With Limited-Stage or Extensive-Stage Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT02987244 | Chidamide Plus CHOEP Combined With Upfront ASCT in Untreated Peripheral T-cell Lymphoma | non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00320359 | Topotecan Plus Cisplatin Versus Etoposide Plus Cisplatin In 1st Line Extensive Disease Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00315705 | A Study of Clofarabine in Combination With Etoposide and Cyclophosphamide in Children With Acute Leukemias. | acute lymphoblastic leukemia;acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT00335738 | Vincristine, Carboplatin, and Etoposide or Observation Only in Treating Patients Who Have Undergone Surgery for Newly Diagnosed Retinoblastoma | retinoblastoma | Etoposide (NPC185498) | |
| NCT00005578 | Combination Chemotherapy With or Without Dexrazoxane in Treating Children With Hodgkin's Disease | lymphoma | Etoposide (NPC185498) | |
| NCT00049439 | Combination Chemotherapy in Treating Patients With AIDS-Related Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00051311 | Modified Stem Cell Transplant Procedure to Treat Patients With Blood and Immune System Cancers | hematopoietic and lymphoid cell neoplasm | Etoposide (NPC185498) | |
| NCT00003935 | Combination Chemotherapy Plus Radiation Therapy in Treating Children With Newly Diagnosed Brain Stem Glioma | brain neoplasm | Etoposide (NPC185498) | |
| NCT03007147 | Imatinib Mesylate and Combination Chemotherapy in Treating Patients With Newly Diagnosed Philadelphia Chromosome Positive Acute Lymphoblastic Leukemia | T-cell acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT00660504 | Phase3 Study of Amrubicin With Cisplatin Versus Etoposide-cisplatin for Extensive Disease Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT00334867 | Combination Chemotherapy With or Without Topotecan in Treating Patients With Newly Diagnosed Localized Ewing's Sarcoma | sarcoma | Etoposide (NPC185498) | |
| NCT05192889 | Trial Treating Relapsed Acute Lymphoblastic Leukemia With Venetoclax and Navitoclax | acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT03663166 | Radiation and Chemotherapy With Ipilimumab Followed by Nivolumab for Patients With Stage III Unresectable Non-Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00003243 | Combination Chemotherapy Plus Infusion of White Blood Cells in Treating Patients With Hematologic Cancer | myelodysplastic/myeloproliferative disease;leukemia;lymphoma;myelodysplastic syndrome | Etoposide (NPC185498) | |
| NCT03531281 | Human Lysozyme Goat Milk in Treating Patients With Blood Cancer Undergoing Donor Stem Cell Transplant | hematopoietic and lymphoid cell neoplasm | Etoposide (NPC185498) | |
| NCT00025610 | Combination Chemotherapy in Treating Infants With Newly Diagnosed Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT00003389 | Combination Chemotherapy With or Without Radiation Therapy in Treating Patients With Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00006374 | Combination Chemotherapy in Treating Patients With Extensive-Stage Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT00809341 | R-ICE and High-Dose Cyclophosphamide With PET/CT for Diffuse Large B-Cell Non-Hodgkin's Lymphoma | non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT02429700 | TC or BEP in Treating Patients With Ovarian Malignant Sex Cord-Stromal Tumors | ovarian sex cord-stromal tumor | Etoposide (NPC185498) | |
| NCT02504359 | Combination Chemotherapy and Donor Stem Cell Transplant Followed by Ixazomib Citrate Maintenance Therapy in Treating Patients With Relapsed High-Risk Multiple Myeloma | plasma cell leukemia;multiple myeloma | Etoposide (NPC185498) | |
| NCT01822496 | Erlotinib Hydrochloride or Crizotinib and Chemoradiation Therapy in Treating Patients With Stage III Non-small Cell Lung Cancer | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00109031 | Palifermin for the Reduction of Oral Mucositis in Single-dose Evaluation (PROMISE) | leukemia;lymphoma;cancer | Etoposide (NPC185498) | |
| NCT00002688 | Cyclosporine and Combination Chemotherapy in Treating Patients With Relapsed or Refractory Acute Myeloid Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT00726986 | Sorafenib, Cisplatin, and Etoposide in Treating Patients With Extensive-Stage Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT00274924 | Rituximab and Combination Chemotherapy in Treating Patients With Stage II, Stage III, or Stage IV Diffuse Large B-Cell Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00026208 | Combination Chemotherapy Plus Low-Dose Radiation Therapy in Treating Patients With Stage I or Stage IIA Hodgkin's Lymphoma | Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT04681417 | Ocular Conservative Treatment for Retinoblastoma : Efficacy of the New Management Strategies and Visual Outcome | retinoblastoma | Etoposide (NPC185498) | |
| NCT00002805 | Combination Chemotherapy in Treating Patients With Acute Myeloid Leukemia or Myelodysplastic Syndrome | leukemia | Etoposide (NPC185498) | |
| NCT00345865 | Autologous Peripheral Stem Cell Transplant in Treating Patients With Non-Hodgkin's Lymphoma or Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT04044222 | A Study to Evaluate the Efficacy and Safety of a Sintilimab Plus ICE Regimen Versus ICE Regimen in Classic Hodgkin's Lymphoma Patients (cHL) Who Have Failed First-line Standard Chemotherapy | classic Hodgkin lymphoma | Etoposide (NPC185498) | |
| NCT02338518 | Comparison of SEEOX and SOX Regimens in Stage ⅢB/ⅢC Gastric Cancer Patients | gastric cancer | Etoposide (NPC185498) | |
| NCT03568994 | Atovaquone (Mepron®) Combined With Conventional Chemotherapy for de Novo Acute Myeloid Leukemia (AML) | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT00005802 | Chemotherapy Followed by Donor White Blood Cells Plus Interleukin-2 in Treating Patients With Acute Myeloid or Lymphocytic Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT00002516 | Combination Chemotherapy Plus Surgery and Radiation Therapy in Treating Patients With Ewing's Sarcoma | sarcoma | Etoposide (NPC185498) | |
| NCT00006040 | Radiolabeled Monoclonal Antibody Therapy and Etoposide Followed by Peripheral Stem Cell Transplantation in Treating Patients With Advanced Myelodysplastic Syndrome or Refractory Leukemia | leukemia;myelodysplastic syndrome;myeloproliferative disorder | Etoposide (NPC185498) | |
| NCT00186888 | Study of Treatment for Patients With Cancer of the Eye -Retinoblastoma | retinoblastoma | Etoposide (NPC185498) | |
| NCT00970385 | Study About Treatment of Newly Diagnosed Non Cutaneous Peripheral T Cell Lymphoma | unspecified peripheral T-cell lymphoma | Etoposide (NPC185498) | |
| NCT00030719 | Combination Chemotherapy With or Without Filgrastim Before Surgery, High-Dose Chemotherapy, and Radiation Therapy Followed by Isotretinoin With or Without Monoclonal Antibody in Treating Patients With Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT04490421 | Camrelizumab Combined With Apatinib, Etoposide and Cisplatin Treat Small-cell Lung Cancer. | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00003052 | Combination Chemotherapy With or Without Hyperthermia Therapy in Treating Patients With Soft Tissue Sarcoma | sarcoma | Etoposide (NPC185498) | |
| NCT00262210 | A Comparative Study for Non-Hodgkin's Lymphoma in Hepatitis B Virus Carriers | non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00003602 | Combination Chemotherapy in Treating Older Patients With Acute Myeloid Leukemia | leukemia;myelodysplastic syndrome | Etoposide (NPC185498) | |
| NCT00504751 | Phase II Study of "VIPER" Chemotherapy in Rel/Ref DLBCL | non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00869232 | UARK 2008-02 A Trial for High-risk Myeloma Evaluating Accelerating and Sustaining Complete Remission | multiple myeloma | Etoposide (NPC185498) | |
| NCT00054665 | PS-341 Alone and PS-341 Plus EPOCH Chemotherapy to Treat Non-Hodgkin's Lymphoma | neoplasm of mature B-cells | Etoposide (NPC185498) | |
| NCT00003573 | Etoposide Plus Radiation Therapy Followed by Combination Chemotherapy in Treating Children With Newly Diagnosed Advanced Medulloblastoma | brain neoplasm | Etoposide (NPC185498) | |
| NCT00098774 | Rituximab and Combination Chemotherapy in Treating Patients With Newly Diagnosed Primary CNS Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT01356290 | Antiangiogenic Therapy for Children With Recurrent Medulloblastoma, Ependymoma and ATRT | medulloblastoma | Etoposide (NPC185498) | |
| NCT00379457 | Combination Chemotherapy in Treating Young Patients With Nonmetastatic Rhabdomyosarcoma | sarcoma | Etoposide (NPC185498) | |
| NCT00002571 | SWOG-9320 Combination Chemotherapy, Radiation Therapy, and Antiviral Therapy in Treating Patients With AIDS-Related Lymphoma | Lymphoma, AIDS-Related | Etoposide (NPC185498) | |
| NCT00193531 | Paclitaxel, Carboplatin, and Oral Etoposide Followed by Weekly Paclitaxel in High Grade Neuroendocrine Carcinoma | neuroendocrine carcinoma | Etoposide (NPC185498) | |
| NCT00573690 | Sorafenib Combined With Cisplatin and Etoposide or Carboplatin and Pemetrexed in Treating Patients With Metastatic Solid Tumors | neoplasm | Etoposide (NPC185498) | |
| NCT00002494 | Combination Chemotherapy in Treating Patients With Non-Hodgkin's Lymphoma or Acute Lymphocytic Leukemia | leukemia;lymphoma | Etoposide (NPC185498) | |
| NCT00231582 | High-dose Chemotherapy With Autologous Stem Cell Transplantation in Poor Prognosis Germ-cell Tumors: TAXIF II | testicular neoplasm | Etoposide (NPC185498) | |
| NCT00025428 | Combination Chemotherapy Before Surgery in Treating Children With Localized Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT00070174 | Gemtuzumab Ozogamicin in Treating Young Patients With Newly Diagnosed Acute Myeloid Leukemia Undergoing Remission Induction and Intensification Therapy | leukemia | Etoposide (NPC185498) | |
| NCT02436707 | Novel Combination Therapy in the Treatment of Relapsed and Refractory Aggressive B-Cell Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT04520074 | Adjuvant Chemotherapy of Three-step Regimen in BRCA1/2 Wide Type Ovarian Cancer (ACTS-2) | ovarian cancer | Etoposide (NPC185498) | |
| NCT00514540 | Carboplatin Plus Docetaxel (Taxotere) in Anaplastic Prostate Cancer | prostate cancer | Etoposide (NPC185498) | |
| NCT00678327 | Fludeoxyglucose F 18-PET/CT Imaging in Assessing Response to Chemotherapy in Patients With Newly Diagnosed Stage II, Stage III, or Stage IV Hodgkin Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00083915 | DTPACE Followed by Tandem Transplant With Melphalan (MEL) 200 Versus MEL/Dexamethasone/Thalidomide (DT) Platinol/Adriamycin/Etoposide (PACE) Hybrid and DTPACE Consolidation | multiple myeloma | Etoposide (NPC185498) | |
| NCT03061656 | Tandem High Dose Chemotherapy With 131I-MIBG Treatment in High Risk Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT00002566 | Combination Chemotherapy With Bone Marrow Transplantation in Treating Men With Germ Cell Tumors | testicular neoplasm | Etoposide (NPC185498) | |
| NCT00002740 | Combination Chemotherapy Plus Peripheral Stem Cell Transplantation Followed by Surgery and/or Radiation Therapy in Treating Young Patients With Advanced Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT01013779 | Merkel Positron Emission Tomography (PET) Protocol | Merkel cell skin cancer | Etoposide (NPC185498) | |
| NCT00977561 | A Study Of Cisplatin (Or Carboplatin) And Etoposide With Or Without Figitumumab (CP-751,871) In Patients With Extensive-Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00053768 | Combination Chemotherapy Followed By Radiation Therapy in Treating Patients With Aggressive Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT05389423 | Pomalidomide and Dose-Adjusted EPOCH +/- Rituximab for HIV-Associated Lymphomas | Burkitts lymphoma;diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT00070187 | Immunotherapy Using Cyclosporine, Interferon Gamma, and Interleukin-2 After High-Dose Myeloablative Chemotherapy With Autologous Stem Cell Transplantation in Treating Patients With Refractory or Relapsed Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00137111 | Therapy for Newly Diagnosed Patients With Acute Lymphoblastic Leukemia | acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT01046825 | Mature B-Cell Lymphoma And Leukemia Study III | lymphoma | Etoposide (NPC185498) | |
| NCT02518750 | Re-Induction Therapy for Relapsed Pediatric T-Cell Acute Lymphoblastic Leukemia or Lymphoma | acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT01025778 | Haploidentical Stem Cell Transplantation for Children With Therapy Resistant Leukemia | acute lymphoblastic leukemia;acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT02771743 | Response-based Treatment of High-risk Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT00003816 | Combination Chemotherapy and Donor Stem Cell Transplant in Treating Patients With Aplastic Anemia or Hematologic Cancer | chronic myeloproliferative disorder;leukemia;lymphoma;myelodysplastic syndrome | Etoposide (NPC185498) | |
| NCT03700359 | A Study of Lobaplatin/Etoposide With or Without Anlotinib Maintenance Therapy in Patients With ES-SCLC | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00887159 | A Randomized Phase II Study of Cisplatin and Etoposide in Combination With Either Hedgehog Inhibitor GDC-0449 or IGF-1R MOAB IMC-A12 for Patients With Extensive Stage | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00080886 | Rituximab and Carmustine, Cytarabine, Etoposide, and Melphalan Followed By Autologous Hematopoietic Stem Cell Transplantation in Treating Patients With B-Cell Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT03519971 | Study of Durvalumab Given With Chemoradiation Therapy in Patients With Unresectable Non-small Cell Lung Cancer | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00216125 | Cisplatin/Etoposide/Radiotherapy +/- Consolidation Docetaxel in Advanced Stage III Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00666484 | Combination Chemotherapy in Treating Younger Patients With Hodgkin Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00002697 | Combination Chemotherapy Plus Radiation Therapy Followed by Peripheral Stem Cell Transplantation in Treating Patients With Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00372593 | Combination Chemotherapy With or Without Gemtuzumab in Treating Young Patients With Newly Diagnosed Acute Myeloid Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT00131027 | High-Dose Methotrexate (MTX) for Adult Acute Lymphoblastic Leukemia (ALL) | acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT01993810 | Comparing Photon Therapy To Proton Therapy To Treat Patients With Lung Cancer | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT02502708 | Study of the IDO Pathway Inhibitor, Indoximod, and Temozolomide for Pediatric Patients With Progressive Primary Malignant Brain Tumors | gliosarcoma;ependymoma;medulloblastoma | Etoposide (NPC185498) | |
| NCT03937843 | Reduced Intensity Radio-chemotherapy for Stage IIA/B Seminoma | seminoma;testicular carcinoma | Etoposide (NPC185498) | |
| NCT01423500 | ALL-SCT BFM International- HSCT in Children and Adolescents With ALL | acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT00186875 | Therapy for Pediatric Relapsed or Refractory Acute Lymphoblastic Leukemia | acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT00005998 | Peripheral Stem Cell Transplantation With Specially Treated Stem Cells in Treating Patients With Non-Hodgkin's Lymphoma or Hodgkin's Disease | lymphoma | Etoposide (NPC185498) | |
| NCT03901118 | Chiauranib in Combination With Chemotherapy in Patients With Ovarian Cancer | ovarian cancer | Etoposide (NPC185498) | |
| NCT02137928 | Carboplatin Periocular Injection for Retinoblastoma | retinoblastoma | Etoposide (NPC185498) | |
| NCT00027846 | Observation or Radiation Therapy and/or Chemotherapy and Second Surgery in Treating Children Who Have Undergone Surgery for Ependymoma | brain neoplasm | Etoposide (NPC185498) | |
| NCT00798148 | 131I-MIBG With Myeloablative Chemotherapy and Autologous Stem Cell Transplantation for the Treatment of High-risk Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT00529503 | A Randomized Phase IIb Placebo-Controlled Study of R-ICE Chemotherapy With and Without SGN-40 for Patients With DLBCL | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT02523976 | Dasatinib Combined With Chemotherapy in Philadelphia Chromosome-positive Acute Lymphoblastic Leukemia | lymphoid leukemia | Etoposide (NPC185498) | |
| NCT00003380 | Liposomal Doxorubicin and Etoposide in Treating Patients With Recurrent or Persistent Ovarian, Fallopian Tube, or Peritoneal Cancer | fallopian tube cancer;peritoneum cancer;ovarian cancer | Etoposide (NPC185498) | |
| NCT04560972 | LB-100, Carboplatin, Etoposide, and Atezolizumab for the Treatment of Untreated Extensive-Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00004110 | Monoclonal Antibody Therapy Plus Etoposide in Treating Patients With Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT01920932 | Adcetris (Brentuximab Vedotin), Combination Chemotherapy, and Radiation Therapy in Treating Younger Patients With Stage IIB, IIIB and IV Hodgkin Lymphoma | Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT01226849 | Feasibility Study Of Adding Bortezomib to R-ICE Chemotherapy To Treat Relapsed/ Refractory Diffuse Large B-Cell Lymphoma | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT00984997 | Combined Modality Treatment for Resectable Non-Small Cell Superior Sulcus Tumors | lung cancer | Etoposide (NPC185498) | |
| NCT00002488 | Combination Chemotherapy in Treating Patients With Intermediate-Grade or High-Grade Non-Hodgkin's Lymphoma Who Have Not Responded to Anthracycline-Containing Combination Chemotherapy | lymphoma | Etoposide (NPC185498) | |
| NCT00500890 | Treatment of Tumors of the Choroid Plexus Epithelium | choroid plexus neoplasm | Etoposide (NPC185498) | |
| NCT00209833 | Treatment of Adults Aged Up to 60 Years With De Novo Acute Myeloblastic Leukaemia,Secondary AML, or RAEB-T | refractory anemia | Etoposide (NPC185498) | |
| NCT00551005 | Etoposide and Celecoxib in Patients With Advanced Cancer | neoplasm | Etoposide (NPC185498) | |
| NCT01032070 | Erlotinib Versus Oral Etoposide in Patients With Recurrent or Refractory Pediatric Ependymoma | childhood ependymoma | Etoposide (NPC185498) | |
| NCT00352027 | Chemotherapy With Low-Dose Radiation for Pediatric Hodgkin Lymphoma | Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT04951115 | A Trial With Chemotherapy, Immunotherapy, and Radiotherapy for Patients With Newly Diagnosed Stage IV Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00388960 | Study of Amrubicin With or Without Cisplatin Versus Etoposide-cisplatin for Extensive Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT01511562 | Combination Chemotherapy With or Without Autologous Stem Cell Transplant in Treating Patients With Central Nervous System B-Cell Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT02199184 | Dose Adjusted EPOCH Regimen in Combination With Ofatumumab or Rituximab in Treating Patients With Newly Diagnosed or Relapsed or Refractory Burkitt Lymphoma or Relapsed or Refractory Acute Lymphoblastic Leukemia | AIDS;acute lymphoblastic leukemia;Burkitts lymphoma | Etoposide (NPC185498) | |
| NCT00004010 | Combination Chemotherapy and Radiation Therapy in Treating Children With Previously Untreated Stage II, Stage III, or Stage IV Hodgkin's Disease | lymphoma | Etoposide (NPC185498) | |
| NCT00003650 | Combination Chemotherapy in Treating Children With Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT02815592 | Trial of BMS-986012 in Combination With Platinum and Etoposide | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00499343 | G-CSF Versus G-CSF Plus GM-CSF for Stem Cell Mobilization in NHL Patients | lymphoma | Etoposide (NPC185498) | |
| NCT05364424 | A Study Evaluating the Efficacy, Safety, and Pharmacokinetics of Glofitamab in Combination With Rituximab Plus Ifosfamide, Carboplatin Etoposide Phosphate in Participants With Relapsed/Refractory Transplant or CAR-T Therapy Eligible Diffuse B-Cell Lymphoma | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT03699956 | RRx-001 Sequentially With a Platinum Doublet or a Platinum Doublet in Third-Line or Beyond in Patients With Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00780104 | Sirolimus in Combination With MEC in High Risk Myeloid Leukemias | chronic myelogenous leukemia;acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT02825147 | Pegaspargase and Methotrexate Based Regimens for Newly Diagnosed Extranodal NK/T Cell Lymphoma | extranodal nasal NK/T cell lymphoma | Etoposide (NPC185498) | |
| NCT00450801 | R-MACLO-IVAM and Thalidomide in Untreated Mantle Cell Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00589056 | Nelfinavir, Radiation Therapy, Cisplatin, and Etoposide in Treating Patients With Stage III Non-Small Cell Lung Cancer That Cannot Be Removed By Surgery | lung cancer | Etoposide (NPC185498) | |
| NCT01104025 | Hybrid Immunotherapy for Hemophagocytic LymphoHistiocytosis | hemophagocytic syndrome | Etoposide (NPC185498) | |
| NCT02265770 | An International Clinical Program for the Diagnosis and Treatment of Children With Ependymoma | childhood ependymoma | Etoposide (NPC185498) | |
| NCT00536978 | Natural Killer (NK) Cell Adback After Allogeneic Stem Cell Transplant With Campath-IH Plus Chemorx for Patients With Lymphoid Malignancies | leukemia;lymphoma | Etoposide (NPC185498) | |
| NCT00002858 | Combination Chemotherapy Plus Radiation Therapy in Treating Patients With Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT01220297 | Sirolimus & Mycophenolate Mofetil as GvHD Prophylaxis in Myeloablative, Matched Related Donor HCT | Abnormality of blood and blood-forming tissues | Etoposide (NPC185498) | |
| NCT00088530 | BBR 2778 for Relapsed, Aggressive Non-Hodgkin's Lymphoma (NHL) | non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00324467 | Tailoring Treatment for B Cell Non-hodgkin's Lymphoma Based on PET Scan Results Mid Treatment | neoplasm of mature B-cells;non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00568464 | Study on VCD/IE in the Patients With Ewing's Sarcoma Family of Tumors (ESFT) | Ewing sarcoma | Etoposide (NPC185498) | |
| NCT00515138 | A Pilot Study of Standard-Dose Rituximab, Ifosfamide, Carboplatin and Etoposide (RICE) Plus Bortezomib (Velcade) in a Dose-Escalating Fashion for Patients With Relapsed or Primary Refractory Aggressive B-Cell Non-Hodgkin's Lymphoma Who Are Candidates for Autologous Stem Cell Transplant | lymphoma | Etoposide (NPC185498) | |
| NCT01710176 | Localized High-Risk Soft Tissue Sarcomas Of The Extremities And Trunk Wall In Adults: An Integrating Approach Comprising Standard Vs Histotype-Tailored Neoadjuvant Chemotherapy | soft tissue sarcoma | Etoposide (NPC185498) | |
| NCT00117598 | Study Evaluating Temsirolimus (CCI-779) In Mantle Cell Lymphoma (MCL) | lymphoma | Etoposide (NPC185498) | |
| NCT00077285 | Irinotecan and Carboplatin as Upfront Window Therapy in Treating Patients With Newly Diagnosed Intermediate-Risk or High-Risk Rhabdomyosarcoma | sarcoma | Etoposide (NPC185498) | |
| NCT04472949 | Thoracic Radiotherapy Plus Maintenance Durvalumab After First Line Carboplatin and Etoposide Plus Durvalumab in Extensive-stage Disease Small Cell Lung Cancer (ED-SCLC). | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00985855 | Feasibility of Cetuximab Associated With Concomitant Radio-Chemotherapy in Patients With Locally Advanced Non Small Cell Lung Cancer | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT05026593 | A Study of IBI110 in Combination With Sintilimab and Chemotherapy in Patients With Untreated Extensive-Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00025272 | Combination Chemotherapy in Treating Patients With Extensive-Stage Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT00040690 | Combination Chemotherapy in Treating Patients With Burkitt's Lymphoma or Burkitt's Leukemia | leukemia;lymphoma | Etoposide (NPC185498) | |
| NCT00002785 | Combination Chemotherapy, Bone Marrow Transplantation, and Radiation Therapy in Treating Infants With Acute Lymphoblastic Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT01165112 | Bendamustine Hydrochloride, Rituximab, Etoposide, and Carboplatin in Treating Patients With Relapsed or Refractory Diffuse Large B-cell Lymphoma or Hodgkin Lymphoma | diffuse large B-cell lymphoma;Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00574080 | UARK 2006-15: A Study of Tandem Transplants With or Without Bortezomib and Thalidomide | multiple myeloma | Etoposide (NPC185498) | |
| NCT04322318 | A Study of Combination Chemotherapy for Patients With Newly Diagnosed DAWT and Relapsed FHWT | kidney Wilms tumor | Etoposide (NPC185498) | |
| NCT00002987 | Combination Chemotherapy Given With Radiation Therapy or Radiation Therapy Alone in Treating Patients With Early-Stage Hodgkin's Disease | lymphoma | Etoposide (NPC185498) | |
| NCT00526396 | STAD-1 Small Cell Lung Cancer Toxicity Adjusted Dosing Study | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT03586999 | Nivolumab With Standard of Care Chemotherapy for Peripheral T Cell Lymphomas | unspecified peripheral T-cell lymphoma | Etoposide (NPC185498) | |
| NCT03229616 | BUCY+VP-16 vs BUCY Conditioning Regimen for DLBCL Undergoing Auto-HSCT | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT01887340 | Therapeutic Strategy Guided by PET-TDM for Patients With Seminoma | seminoma | Etoposide (NPC185498) | |
| NCT00936936 | High-dose Chemotherapy for Poor-Prognosis Relapsed Germ-Cell Tumors | testicular carcinoma | Etoposide (NPC185498) | |
| NCT03188198 | Risk Adapted Therapy in Diffuse Large B Cell Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT01681537 | Lenalidomide Plus Chemotherapy for AML | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT04622228 | Study of Low-Dose Radiotherapy (LDRT) Concurrent Cisplatin/Carboplatin Plus Etoposide With Atezolizumab for Patients With Extensive-Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00209222 | Efficacy of R-CHOP vs R-CHOP/R-DHAP in Untreated MCL | Mantle cell lymphoma | Etoposide (NPC185498) | |
| NCT03458260 | Study of Pixantrone in CD20+ Relapsed/Refractory Aggressive Non-Hodgkin Lymphoma | non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00187083 | A Study of Children With Refractory or Relapsed ALL | acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT03328104 | Everolimus in Combination With Nelarabine, Cyclophosphamide and Etoposide in Lymphoblastic Leukemia/Lymphoma | lymphoid leukemia;lymphoblastic lymphoma | Etoposide (NPC185498) | |
| NCT05161533 | Hypofractionated Radiation Therapy After Durvalumab and Chemotherapy for the Treatment of Stage IV Extensive Stage Small Cell Lung Cancer, CASPIAN-RT Trial | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT01900509 | Bendamustine Hydrochloride, Clofarabine, and Etoposide in Treating Younger Patients With Relapsed or Refractory Hematologic Malignancies | Hodgkins lymphoma;leukemia;non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT04005716 | Study of Platinum Plus Etoposide With or Without BGB-A317 in Participants With Untreated Extensive-Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT02412267 | Study of Ofatumomab in Combination With ICE-chemotherapy in Patients With Diffuse Large B-cell Lymphoma (DLBCL) | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT00002823 | Combination Chemotherapy Compared With No Treatment Following Surgery in Treating Patients With Non-small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT03298074 | Apatinib Plus Etoposide Versus Etoposide Alone for Platinum-resistant Recurrent Ovarian Cancer | ovarian cancer | Etoposide (NPC185498) | |
| NCT01331590 | Disrupting the Bone Marrow Microenvironment With G-CSF in Acute Lymphoblastic Leukemia | acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT00764907 | Risk-Adjusted Combination Chemotherapy in Treating Young Patients With Acute Lymphoblastic Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT01555710 | Study of Palifosfamide-tris in Combination With Carboplatin and Etoposide in Chemotherapy Naïve Patients With Extensive-Stage Small Cell Lung Cancer (The MATISSE Study) | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00225173 | Combination Chemotherapy +/- Radiation in High Risk Hodgkin's Disease | Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT02402920 | Pembrolizumab and Concurrent Chemoradiotherapy or Radiation Therapy in Treating Patients With Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00011921 | Chemotherapy Followed by Peripheral Stem Cell or Bone Marrow Transplant Compared With Chemotherapy Alone in Treating Patients With Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT01830777 | Brentuximab Vedotin + Re-induction Chemotherapy for AML | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT00028665 | Cyclophosphamide W/or W/Out Rituximab and Peripheral Stem Cell Transplantation in Patients With Recurrent Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00829205 | Se-Methyl-Seleno-L-Cysteine, Rituximab, Ifosfamide, Carboplatin, and Etoposide in Treating Patients With Diffuse Large B-Cell Lymphoma That Has Relapsed or Not Responded to Treatment | lymphoma | Etoposide (NPC185498) | |
| NCT00002657 | SWOG-9239 Reduction of Immunosuppression Plus Interferon Alfa and Combination Chemotherapy in Treating Patients With Malignant Tumors That Develop After Organ Transplant | multiple myeloma | Etoposide (NPC185498) | |
| NCT00717938 | A Study of Standard Treatment +/- Enoxaparin in Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00042978 | Carboplatin and Etoposide With or Without Oblimersen Sodium in Treating Patients With Extensive Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT01704716 | High Risk Neuroblastoma Study 1.8 of SIOP-Europe (SIOPEN) | neuroblastoma | Etoposide (NPC185498) | |
| NCT05224141 | Pembrolizumab/Vibostolimab (MK-7684A) or Atezolizumab in Combination With Chemotherapy in First Line Treatment of Extensive-Stage Small Cell Lung Cancer (MK-7684A-008, KEYVIBE-008) | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00003284 | High-Dose Combination Chemotherapy Followed by Peripheral Stem Cell Transplantation in Treating Patients With Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT00734877 | UARK 2013-13, Total Therapy 4B - Formerly 2008-01 - A Phase III Trial for Low Risk Myeloma | multiple myeloma | Etoposide (NPC185498) | |
| NCT00002982 | Combination Chemotherapy and Peripheral Stem Cell Transplantation in Treating Older Patients With Refractory or Relapsed Intermediate-Grade Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT02896582 | Efficacy of Upfront and Maintenance Obinutuzumab in Mantle Cell Lymphoma Treated by DHAP and MRD Driven Maintenance | Mantle cell lymphoma | Etoposide (NPC185498) | |
| NCT01873326 | Paclitaxel, Ifosfamide and Cisplatin (TIP) Versus Bleomycin, Etoposide and Cisplatin (BEP) for Patients With Previously Untreated Intermediate- and Poor-risk Germ Cell Tumors | germ cell tumor | Etoposide (NPC185498) | |
| NCT00004188 | Combination Chemotherapy and Peripheral Stem Cell Transplantation in Treating Patients With Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT00043927 | Extensive Small Cell Lung Cancer Treatment Using An Investigational Drug Plus Chemotherapy In Chemotherapy-Naive Adults | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT03860844 | Isatuximab in Combination With Chemotherapy in Pediatric Patients With Relapsed/Refractory Acute Lymphoblastic Leukemia or Acute Myeloid Leukemia | acute lymphoblastic leukemia;acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT00002788 | High-Dose Chemotherapy Followed by Total-Body Irradiation and Peripheral Stem Cell Transplantation in Treating Patients With Chronic Lymphocytic Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT00006032 | Combination Chemotherapy Followed by Peripheral Stem Cell Transplantation in Treating Women With Metastatic Breast Cancer | breast cancer | Etoposide (NPC185498) | |
| NCT01423747 | Allogeneic Stem Cell Transplantation in Children and Adolescents With Acute Lymphoblastic Leukaemia | acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT00002719 | Combination Chemotherapy With or Without G-CSF in Treating Older Patients With Acute Myeloid Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT00265031 | HD12 for Advanced Stages | lymphoma | Etoposide (NPC185498) | |
| NCT02633514 | Adjuvant Treatment for Incomplete Resection Thymoma or Thymic Carcinoma | thymus neoplasm | Etoposide (NPC185498) | |
| NCT00869752 | MK-0646, Etoposide, and Cisplatin in Treating Patients With Extensive-Stage Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT00025259 | Chemotherapy With or Without Additional Chemotherapy and/or Radiation Therapy in Treating Children With Newly Diagnosed Hodgkin's Disease | classic Hodgkin lymphoma | Etoposide (NPC185498) | |
| NCT02638428 | Genomics-Based Target Therapy for Children With Relapsed or Refractory Malignancy | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT00537511 | A Phase I/II Study to Determine the Maximum Tolerated Dose (MTD) and Safety of CC-4047 (Pomalidomide) Administered in Conjunction With Cisplatin and Etoposide | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT05422183 | Envafolimab, Lenvatinib Combined With VP-16 in Platinum-resistant Recurrent Epithelial Ovarian Cancer | ovarian cancer | Etoposide (NPC185498) | |
| NCT00621361 | Phase I Safety and Tolerability Study of Cediranib (RECENTIN™, AZD2171) in Combination With Chemo in First Line Lung Cancer Patients | lung cancer | Etoposide (NPC185498) | |
| NCT00002524 | Combination Chemotherapy in Treating Patients With AIDS-Related Lymphoma | Lymphoma, AIDS-Related | Etoposide (NPC185498) | |
| NCT00612430 | Ph II Bevacizumab + Etoposide for Pts w Recurrent MG | gliosarcoma | Etoposide (NPC185498) | |
| NCT00003583 | Amifostine to Prevent Side Effects in Patients Who Are Receiving Chemotherapy and Radiation Therapy for Limited-Stage Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT03591510 | A Global Study of Midostaurin in Combination With Chemotherapy to Evaluate Safety, Efficacy and Pharmacokinetics in Newly Diagnosed Pediatric Patients With FLT3 Mutated AML | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT00005977 | Combination Chemotherapy in Treating Patients With Non-Hodgkin's Lymphoma or Acute Lymphocytic Leukemia | leukemia;lymphoma | Etoposide (NPC185498) | |
| NCT00180791 | High Risk Primitive Neuroectodermal (PNET) Brain Tumors in Childhood | medulloblastoma | Etoposide (NPC185498) | |
| NCT00012311 | Chemotherapy Plus Peripheral Stem Cell Transplantation Compared With Chemotherapy Alone in Treating Women With Stage IV Breast Cancer | breast cancer | Etoposide (NPC185498) | |
| NCT02748889 | Carboplatin Plus Etoposide With or Without MPDL3280A in Untreated Extensive Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT05245994 | An Open Label, Multicenter Phase 2 Study of Durvalumab (MEDI4736) + Olaparib as Maintenance Therapy in Chinese | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00367497 | Safety and Efficacy Study of Salvage Chemotherapy (R-ESHAP) to Treat Relapsed and Refractory Aggressive Non-Hodgkin's Lymphoma | non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT05466318 | ChiCGB vs BEAM in High-risk or R/R Lymphomas | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT01677949 | Clofarabine, Cyclophosphamide and Etoposide for Minimal Residual Disease Positive Acute Leukemia | acute lymphoblastic leukemia;acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT04077905 | Pegylated Liposomal Doxorubicin as a Induction Therapy for Lymphoma Induced Hemophagocytic Lymphohistiocytosis. | hemophagocytic syndrome | Etoposide (NPC185498) | |
| NCT00003436 | Combination Chemotherapy With or Without Bone Marrow Transplantation in Treating Children With Acute Myeloid Leukemia | leukemia;myelodysplastic syndrome | Etoposide (NPC185498) | |
| NCT00388349 | Gemcitabine and Hodgkin's Disease Chemotherapy Followed by Peripheral Blood Stem Cell Rescue for Hodgkin's Disease | Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00151255 | All-Trans Retinoic Acid in Combination With Standard Induction and Consolidation Therapy in Older Patients With Newly Diagnosed Acute Myeloid Leukemia | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT01027923 | IV Plerixafor With Mitoxantrone Etoposide and Cytarabine for Acute Myeloid Leukemia (AML) | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT00025064 | Combination Chemotherapy With or Without Radiation Therapy and Peripheral Stem Cell Transplant in Treating Children With Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00534469 | Auto BMT for Non-M3 AML in 1st Remission in Pts | leukemia | Etoposide (NPC185498) | |
| NCT02499770 | Trilaciclib (G1T28), a CDK 4/6 Inhibitor, in Combination With Etoposide and Carboplatin in Extensive Stage Small Cell Lung Cancer (SCLC) | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00005631 | Rituximab and Combination Chemotherapy in Treating Patients With Relapsed or Refractory Large Cell Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT01476839 | Radiolabeled Monoclonal Antibody Therapy and Combination Chemotherapy Before Stem Cell Transplant in Treating Patients With Primary Refractory or Relapsed Hodgkin Lymphoma | Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00472056 | Rituximab in Addition to Autologous Transplantation With BEAM for Patients With Lymphoid Malignancies | lymphoma | Etoposide (NPC185498) | |
| NCT02861573 | Study of Pembrolizumab (MK-3475) Combination Therapies in Metastatic Castration-Resistant Prostate Cancer (MK-3475-365/KEYNOTE-365) | prostate cancer | Etoposide (NPC185498) | |
| NCT00562978 | Yttrium Y 90 Ibritumomab Tiuxetan, Etoposide, Cyclophosphamide, and an AHSCT in Non-Hodgkin's Lymphoma Patients | lymphoma | Etoposide (NPC185498) | |
| NCT00043979 | Stem Cell Transplantation in Patients With High-Risk and Recurrent Pediatric Sarcomas | sarcoma | Etoposide (NPC185498) | |
| NCT04028050 | A Study of Atezolizumab in Combination With Carboplatin Plus Etoposide to Investigate Safety and Efficacy in Patients With Untreated Extensive-Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00217503 | Bortezomib and Antiviral Therapy Followed By Effusion Drainage, Bevacizumab, and Combination Chemotherapy in Treating Patients With Primary Effusion Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00040872 | Multiple Therapies in Treating Patients With Advanced Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT00476190 | ALL Adult Consortium Trial: Adult ALL Trial | acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT02194049 | Cisplatin, Etoposide and PI3K Inhibitor BKM120 in Treating Patients With Advanced Solid Tumors or Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00880243 | Effect of Priming During Induction and Consolidations in Younger Acute Myeloid Leukemia (AML) | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT00104676 | Combination Chemotherapy in Treating Patients With Stage II or Stage III Germ Cell Tumors | teratoma;testicular neoplasm | Etoposide (NPC185498) | |
| NCT02179528 | Study of Small Doses of Etoposide as Maintenance Treatment in Small Cell Lung Cancer(SCLC) | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00002822 | Combination Chemotherapy Followed by Radiation Therapy in Patients With Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT00324298 | Bleomycin, Etoposide, and Cisplatin in Treating Patients With Metastatic Germ Cell Cancer of the Testicles | testicular neoplasm | Etoposide (NPC185498) | |
| NCT00005823 | Intensive Compared With Nonintensive Chemotherapy in Treating Older Patients With Acute Myeloid Leukemia or Myelodysplastic Syndrome | leukemia;myelodysplastic syndrome;myeloproliferative disorder | Etoposide (NPC185498) | |
| NCT00499616 | Combination Chemotherapy and Surgery With or Without Isotretinoin in Treating Young Patients With Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT00278278 | Combination Chemotherapy and Radiation Therapy With or Without Methotrexate in Treating Young Patients With Newly Diagnosed Gliomas | Central Nervous System Neoplasm | Etoposide (NPC185498) | |
| NCT00572169 | UARK 2006-66, Total Therapy 3B: An Extension of UARK 2003-33 Total Therapy | multiple myeloma | Etoposide (NPC185498) | |
| NCT00002509 | High-Dose Combination Chemotherapy Followed by Peripheral Stem Cell Transplantation in Treating Patients With Poor-Prognosis Breast Cancer | breast cancer | Etoposide (NPC185498) | |
| NCT03273829 | Carfilzomib in Combination With Cyclophosphamide and Etoposide for Children | leukemia | Etoposide (NPC185498) | |
| NCT04254471 | This Phase II/III, Multicenter Study is Designed to Evaluate the Safety and Clinical Activity of AL3810 in Patients | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00005863 | Combination Chemotherapy With or Without Filgrastim and/or Tretinoin in Treating Patients With Acute Myeloid Leukemia | leukemia;myelodysplastic syndrome;myeloproliferative disorder | Etoposide (NPC185498) | |
| NCT00240097 | Study of Sequential Topoisomerase, Irinotecan/Oxaliplatin - Etoposide /Carboplatin in Extensive Small Cell Lung Cancer (SCLC) | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT04356690 | Etoposide in Patients With COVID-19 Infection | COVID-19 | Etoposide (NPC185498) | |
| NCT02039726 | (QuANTUM-R): An Open-label Study of Quizartinib Monotherapy vs. Salvage Chemotherapy in Acute Myeloid Leukemia (AML) Subjects Who Are FLT3-ITD Positive | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT00004031 | SWOG-9704 Chemoradiotherapy and Peripheral Stem Cell Transplantation Compared With Combination Chemotherapy in Treating Patients With Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT03781869 | Study of Anlotinib as the Maintenance Therapy for Extensive-stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT03016871 | Nivolumab, Ifosfamide, Carboplatin, and Etoposide as Second-Line Therapy in Treating Patients With Refractory or Relapsed HL | Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT03116971 | Phase Ib/II Study of M3814 With Etoposide and Cisplatin in Small Cell Lung Cancer (SCLC) Extensive Disease (ED) | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT02580994 | Pembrolizumab in Untreated Extensive SCLC | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00936156 | Treatment of High-Risk Cerebral Primitive Neuroectodermal Tumors in Children Aged Over 5 Years | primitive neuroectodermal tumor | Etoposide (NPC185498) | |
| NCT00005985 | Filgrastim and Chemotherapy Followed by Peripheral Stem Cell Transplant in Treating Patients With Hodgkin's Lymphoma or Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00003119 | Surgery in Treating Children With Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT00025623 | Combination Chemotherapy Followed by Surgery in Treating Infants With Newly Diagnosed Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT01000285 | EPOCH Chemotherapy and Bortezomib for Associated T-Cell Leukemia Lymphoma | T-cell acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT00693719 | Irinotecan and Etoposide in Treating Patients With Recurrent, Locally Advanced, or Metastatic Breast Cancer | breast cancer | Etoposide (NPC185498) | |
| NCT00933673 | New Combination Regimen of L-asparaginase, Dexamethasone, Ifosfamide, Cisplatin and Etoposide in NK/T-Cell Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT04871607 | Yttrium-90 Labeled Anti-CD25 Monoclonal Antibody Combined With BEAM Chemotherapy Conditioning for the Treatment of Primary Refractory or Relapsed Hodgkin Lymphoma | Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00908180 | Carmustine, Etoposide, Cytarabine, Melphalan, and Alemtuzumab Followed by Donor Stem Cell Transplant in Treating Patients With Relapsed or Refractory Hodgkin Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00066222 | Cisplatin, Etoposide, and Radiation Therapy in Treating Patients With Limited-Stage Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT04363255 | First-line Chemotherapy Followed by Toripalimab Combined With Anlotinib for Maintenance in ES-SCLC | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00002611 | Combination Chemotherapy Alone or With Radiation Therapy in Treating Children With Kidney Cancer | kidney cancer | Etoposide (NPC185498) | |
| NCT00045617 | S0122: Combination Chemotherapy, Radiation Therapy, and Vaccine Therapy in Limited-Stage Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT00002596 | Combination Chemotherapy With or Without Bone Marrow or Stem Cell Transplantation in Treating Men With Untreated Germ Cell Tumors | childhood germ cell tumor;testicular neoplasm | Etoposide (NPC185498) | |
| NCT00616785 | Combination Chemotherapy and Radiation Therapy in Treating Patients With Stage III Non-Small Cell Lung Cancer That Cannot Be Removed by Surgery | lung cancer | Etoposide (NPC185498) | |
| NCT00003696 | Combination Chemotherapy in Treating Patients Who Have Extensive-Stage Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT00032149 | Combination Chemotherapy Plus Filgrastim in Treating Patients With HIV-Related Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT02776605 | Ponatinib With Chemotherapy for Young Adults Ph Positive Acute Lymphoblastic Leukemia | acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT00278252 | Etoposide in Treating Young Patients With Relapsed Ependymoma | Central Nervous System Neoplasm | Etoposide (NPC185498) | |
| NCT00002945 | High Dose Chemotherapy, Peripheral Stem Cell Transplantation, and Interleukin-2 in Treating Patients With Acute Myeloid Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT00004907 | Chemotherapy and Radiation Therapy Plus Bone Marrow or Peripheral Stem Cell Transplantation in Treating Patients With Relapsed or Refractory T-cell Lymphoma, Hodgkin's Lymphoma, or Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT04012606 | Toripalimab in Combination With Platinum Plus Etoposidein Patients With Extensive-Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00062439 | S0220: Chemoradiotherapy Followed By Surgery and Docetaxel in Treating Patients With Pancoast Tumors | lung cancer | Etoposide (NPC185498) | |
| NCT04551131 | Use Of A Response-Adapted Ruxolitinib-Containing Regimen For The Treatment Of Hemophagocytic Lymphohistiocytosis | hemophagocytic syndrome | Etoposide (NPC185498) | |
| NCT00691210 | Phase I Vorinostat in Combination With Niacinamide and Etoposide for Lymphoid Malignancies | Hodgkins lymphoma;non-Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00204646 | Neoadjuvant Adriamycin and Ifosfamide Plus High-Dose ICE in Patients With Soft Tissue Sarcoma (STS) | sarcoma | Etoposide (NPC185498) | |
| NCT03755804 | Pediatric Classical Hodgkin Lymphoma Consortium Study: cHOD17 | Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00136084 | Treatment of Patients With Newly Diagnosed Acute Myeloid Leukemia or Myelodysplasia | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT00004169 | Chemotherapy, Radiation Therapy, and Peripheral Stem Cell Transplantation in Treating Patients With Hodgkin's Disease | lymphoma | Etoposide (NPC185498) | |
| NCT05361395 | First-Line Tarlatamab in Combination With Carboplatin, Etoposide, and PD-L1 Inhibitor in Subjects With Extensive Stage Small Cell Lung Cancer (ES-SCLC) | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT03182244 | A Study of ASP2215 Versus Salvage Chemotherapy In Patients With Relapsed or Refractory Acute Myeloid Leukemia (AML) With FLT3 Mutation | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT00703820 | Clofarabine Plus Cytarabine Versus Conventional Induction Therapy And A Study Of NK Cell Transplantation In Newly Diagnosed Acute Myeloid Leukemia | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT02166463 | Brentuximab Vedotin and Combination Chemotherapy in Treating Children and Young Adults With Stage IIB, Stage IIIB, IVA, or IVB Hodgkin Lymphoma | classic Hodgkin lymphoma | Etoposide (NPC185498) | |
| NCT04452370 | Oral Etoposide Combined With Anlotinib in Advanced Triple Negative Breast Cancer | breast cancer | Etoposide (NPC185498) | |
| NCT02949895 | Study of BMS-986012 in Subjects With Small Cell Lung Caner | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT04092283 | Testing the Addition of an Antibody to Standard Chemoradiation Followed by the Antibody for One Year to Standard Chemoradiation Followed by One Year of the Antibody in Patients With Unresectable Stage III Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00658580 | Randomized Study of Cisplatin-Etoposide Versus an Etoposide Regimen Without Cisplatin in Extensive Small-Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00002634 | Chemotherapy, Radiation Therapy, Immunotherapy, and Bone Marrow Transplantation in Treating Patients With Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT00265889 | Autologous Stem Cell Transplant in Treating Patients With Progressive or Recurrent Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00006363 | Combination Chemotherapy With or Without PSC 833, Peripheral Stem Cell Transplantation, and/or Interleukin-2 in Treating Patients With Acute Myeloid Leukemia | acute basophilic leukemia;acute erythroleukemia;acute megakaryoblastic leukaemia;acute monocytic leukemia;acute myeloblastic leukemia without maturation;acute myelomonocytic leukemia;myelodysplastic syndrome | Etoposide (NPC185498) | |
| NCT00702962 | Carboplatin and Etoposide in Combination With Vorinostat for Patients With Extensive Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT04061772 | A Study of Bortezomib Combined With CHEP in Peripheral T Cell Lymphoma | unspecified peripheral T-cell lymphoma | Etoposide (NPC185498) | |
| NCT00002887 | Hydroxyurea Plus Combination Chemotherapy in Patients With Non-small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT02947113 | Combination of Platinum Doublets and Hypofractionated Radiotherapy in NSCLC | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT02867956 | Apatinib and Etoposide in Patients With Platinum Resistant or Refractory Ovarian Cancer | ovarian cancer | Etoposide (NPC185498) | |
| NCT00310128 | Combination Chemotherapy Followed by Rituximab and Yttrium Y 90 Ibritumomab Tiuxetan in Treating Patients With Relapsed or Refractory AIDS-Related Non-Hodgkin's Lymphoma | anaplastic large cell lymphoma;Lymphoma, AIDS-Related | Etoposide (NPC185498) | |
| NCT01783535 | Protocol for the Study and Treatment of Participants With Intraocular Retinoblastoma | retinoblastoma | Etoposide (NPC185498) | |
| NCT03786783 | Dinutuximab, Sargramostim, and Combination Chemotherapy in Treating Patients With Newly Diagnosed High-Risk Neuroblastoma | ganglioneuroblastoma;neuroblastoma | Etoposide (NPC185498) | |
| NCT01450761 | Trial in Extensive-Disease Small Cell Lung Cancer (ED-SCLC) Subjects Comparing Ipilimumab Plus Etoposide and Platinum Therapy to Etoposide and Platinum Therapy Alone | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00304018 | Donor Umbilical Cord Blood Transplant in Treating Patients With Advanced Hematologic Cancer | leukemia;multiple myeloma;myelodysplastic syndrome | Etoposide (NPC185498) | |
| NCT00632853 | Radiation Therapy Regimens in Treating Patients With Limited-Stage Small Cell Lung Cancer Receiving Cisplatin and Etoposide | lung cancer | Etoposide (NPC185498) | |
| NCT00002756 | Induction Intensification in Treating Infants With Newly Diagnosed Acute Lymphoblastic Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT00686959 | Chemotherapy and Radiation in Treating Participants With Stage 3 Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00002552 | Chemotherapy Plus Bone Marrow Transplantation in Treating Patients With Refractory Non-Hodgkin's Lymphoma, Hodgkin's Disease, or Multiple Myeloma | multiple myeloma | Etoposide (NPC185498) | |
| NCT02323737 | Phase II Study of Irinotecan and Cisplatin in Extensive-stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00939653 | T2007-002 Clofarabine, Etoposide, Cyclophosphamide in Relapsed Acute Myelogenous Leukemia (AML) | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT02733107 | The Efficacy and Safety of Apatinib Combined With Etoposide in Heavily Pretreated Advanced Non-small Cell Lung Cancer | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00083538 | Study of Tumor Antigen-Pulsed Autologous Dendritic Cell Vaccination Administrated Subcutaneously or Intranodally | multiple myeloma | Etoposide (NPC185498) | |
| NCT00020722 | Chemotherapy Followed by Peripheral Stem Cell Transplantation Plus Biological Therapy in Treating Women With Stage IV Breast Cancer | breast cancer | Etoposide (NPC185498) | |
| NCT02157792 | M6620 First in Human Study | neoplasm | Etoposide (NPC185498) | |
| NCT01457040 | Intensified Conditioning Regimen With High-Dose-Etoposide for Allo-HSCT for Adult Acute Lymphoblastic Leukemia | acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT00002522 | Combination Chemotherapy Followed by Bone Marrow or Peripheral Stem Cell Transplantation in Treating Patients With Relapsed or Refractory Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT02101853 | Blinatumomab in Treating Younger Patients With Relapsed B-cell Acute Lymphoblastic Leukemia | acute lymphoblastic leukemia | Etoposide (NPC185498) | |
| NCT00186966 | Treatment of Children and Adolescents With Refractory or Relapsed Acute Myeloid Leukemia | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT00006184 | Chemotherapy, Stem Cell Transplantation and Donor and Patient Vaccination for Treatment of Multiple Myeloma | multiple myeloma | Etoposide (NPC185498) | |
| NCT02176967 | Response and Biology-Based Risk Factor-Guided Therapy in Treating Younger Patients With Non-high Risk Neuroblastoma | ganglioneuroblastoma;neuroblastoma | Etoposide (NPC185498) | |
| NCT02626338 | Pilot Study of Crenolanib Combined With Standard Salvage Chemotherapy in Subjects With R/R AML | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT00003541 | Combination Chemotherapy, Radiation Therapy, and Peripheral Stem Cell Transplantation in Treating Patients With Stage III or Stage IV Mantle Cell Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT02875457 | Apatinib as the Maintenance Therapy for Extensive Stage Small Cell Lung Cancer After Combined With Etoposide/Cisplatin | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT02243436 | Brentuximab Vedotin in Pre-transplant Induction and Consolidation for Relapsed or Refractory Hodgkin Lymphoma | classic Hodgkin lymphoma | Etoposide (NPC185498) | |
| NCT02784015 | Response Based Treatment for Children With Unresectable Localized Soft Tissue Sarcoma | soft tissue sarcoma | Etoposide (NPC185498) | |
| NCT01653418 | Phase II Study of V-BEAM Conditioning Regimen Prior to Second Autologous Stem Cell Transplantation | multiple myeloma | Etoposide (NPC185498) | |
| NCT00002588 | Combination Chemotherapy in Treating Patients With Recurrent or Refractory Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT00003194 | Combination Chemotherapy and Peripheral Stem Cell Transplantation in Treating Patients With Recurrent or Refractory Solid Tumors | neoplasm | Etoposide (NPC185498) | |
| NCT04917250 | GPED for Patients With Relapsed/Refractory or Advanced NK/T-cell Lymphoma | extranodal nasal NK/T cell lymphoma | Etoposide (NPC185498) | |
| NCT01548573 | Tandem Auto Transplantation in Myeloma Patients With <12 Months of Prior Treatment | multiple myeloma | Etoposide (NPC185498) | |
| NCT02636322 | Smart Start: A Phase II Study of Rituximab, Lenalidomide, and Ibrutinib | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT03628131 | Combination Chemotherapy With Pazopanib in Children and Adolescents With Relapsed/Refractory Solid Tumors | neoplasm | Etoposide (NPC185498) | |
| NCT00025363 | Comparison of Chemotherapy Regimens in Treating Children With Relapsed or Progressive Rhabdomyosarcoma | rhabdomyosarcoma | Etoposide (NPC185498) | |
| NCT00002674 | Combination Chemotherapy Followed by Peripheral Stem Cell Transplantation in Treating Patients With Chronic Myelogenous Leukemia | leukemia | Etoposide (NPC185498) | |
| NCT02341989 | Adjuvant Bleomycin, Etoposide and Cisplatin (BEP) Versus Carboplatin in Stage I Seminomatous Testicular Cancer | seminoma;testicular neoplasm | Etoposide (NPC185498) | |
| NCT03613597 | Etoposide Plus Lobaplatin Versus Etoposide Plus Cisplatin for Patients With Limited Small-cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT05353257 | A Study to Evaluate the Efficacy and Safety of HLX10 in Combination With Chemotherapy and Concurrent Radiotherapy in Patients With Limited-Stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT01857453 | Interest of a Dose Decrease for Radiotherapy Associated With Chemotherapy for Treatment of Standard Risk Adult Medulloblastomas | medulloblastoma | Etoposide (NPC185498) | |
| NCT00822120 | S0816 Fludeoxyglucose F 18-PET/CT Imaging and Combination Chemotherapy With or Without Additional Chemotherapy and G-CSF in Treating Patients With Stage III or Stage IV Hodgkin Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00793845 | Tandem High-dose Chemotherapy and Autologous Stem Cell Rescue in Patients With High-risk Neuroblastoma | neuroblastoma | Etoposide (NPC185498) | |
| NCT00004904 | Stem Cell Transplantation in Treating Patients With Hematologic Cancer | graft versus host disease;chronic myeloproliferative disorder;leukemia;multiple myeloma;myelodysplastic syndrome | Etoposide (NPC185498) | |
| NCT03840902 | M7824 With cCRT in Unresectable Stage III Non-small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00003299 | Cisplatin Plus Etoposide With or Without Paclitaxel in Treating Patients With Extensive-Stage Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT00033696 | Combination Chemotherapy and Radiation Therapy in Treating Patients With Limited-Stage Small Cell Lung Cancer | lung cancer | Etoposide (NPC185498) | |
| NCT02912702 | L-DEP as an Initial Treatment for EBV-HLH | hemophagocytic syndrome | Etoposide (NPC185498) | |
| NCT04638790 | First Line Chemotherapy for Classical Hodgkin Lymphoma in Russia (HL-Russia-1) | Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT01371981 | Bortezomib and Sorafenib Tosylate in Treating Patients With Newly Diagnosed Acute Myeloid Leukemia | myeloid sarcoma | Etoposide (NPC185498) | |
| NCT00826644 | Trial of Belotecan/Cisplatin in Chemotherapy Naive Small Cell Lung Cancer Patient | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00233987 | S0410 Tandem Stem Cell Transplantation in Treating Patients With Progressive or Recurrent Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00801918 | Denileukine Diftitox for Relapsed ALCL | lymphoma | Etoposide (NPC185498) | |
| NCT02512926 | Carfilzomib in Combination With Cyclophosphamide and Etoposide for Children | leukemia | Etoposide (NPC185498) | |
| NCT05006664 | Brentuximab Vedotin in Combination With CHEP in Patient With PTCL | lymphoma | Etoposide (NPC185498) | |
| NCT04702880 | A Study of BMS-986012 in Combination With Carboplatin, Etoposide, and Nivolumab as First-line Therapy in Extensive-stage Small Cell Lung Cancer | small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT03383406 | A Phase II Trial of Ifosfamide, Etoposide, Cytarabine, and Methotrexate (IVAM) Chemotherapy for Refractory or Relapsed Diffuse Large B Cell Lymphoma | diffuse large B-cell lymphoma | Etoposide (NPC185498) | |
| NCT00003053 | Combination Chemotherapy in Treating Patients With Stage I, II, or IIIA Non-small Cell Lung Cancer That Has Been Surgically Removed | lung cancer | Etoposide (NPC185498) | |
| NCT00005793 | A Phase I/II Study of Induction Chemotherapy With Daunorubicin, Cytarabine, Topotecan and Etoposide | leukemia | Etoposide (NPC185498) | |
| NCT00004900 | Chemotherapy Plus Peripheral Stem Cell Transplantation in Treating Women With Metastatic Breast Cancer | breast cancer | Etoposide (NPC185498) | |
| NCT01222572 | Stereotactic Boost for Locally Advanced Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Etoposide (NPC185498) | |
| NCT00303849 | Carboplatin, Melphalan, Etoposide Phosphate, Mannitol, and Sodium Thiosulfate in Treating Patients With Previously Treated Brain Tumors | anaplastic oligodendroglioma | Etoposide (NPC185498) | |
| NCT00002461 | Combination Chemotherapy Followed by Bone Marrow or Peripheral Stem Cell Transplantation in Treating Patients With Refractory Hodgkin's Disease or Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00003575 | Interleukin-12 Following Chemotherapy in Treating Patients With Refractory HIV-Associated Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00002521 | Combination Chemotherapy and Bone Marrow and/or Peripheral Stem Cell Transplantation in Treating Patients With Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT00118209 | Rituximab and Combination Chemotherapy in Treating Patients With Diffuse Large B-Cell Non-Hodgkin's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT02349178 | Bridging Study to Eliminate Presence of MRD for Acute Leukemia Before HCT | acute lymphoblastic leukemia;acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT02306291 | Study to Determine Safety, Pharmacokinetics and Efficacy of GMI-1271 in Combination With Chemotherapy in AML | acute myeloid leukemia | Etoposide (NPC185498) | |
| NCT01468740 | Prospective Study on HIV-related Hodgkin Lymphoma | Hodgkins lymphoma | Etoposide (NPC185498) | |
| NCT00392834 | Rituximab and Combination Chemotherapy in Treating Patients With Newly Diagnosed, HIV-Associated Burkitt's Lymphoma | lymphoma | Etoposide (NPC185498) | |
| NCT03043872 | Durvalumab ± Tremelimumab in Combination With Platinum Based Chemotherapy in Untreated Extensive-Stage Small Cell Lung Cancer (CASPIAN) | small cell lung carcinoma | Etoposide (NPC185498) |
❱❱❱ Associated Human Diseases and Detailed Association Evidence
How do we define the Plant-Targeted Human Disease Association?
Associated human diseases of an individual plant are summurized based on FOUR types of association evidence, these include:
❶ Association by Therapeutic Target: Bioactive protein targets of the plant were defined in "Molecular Targets" section, target-disease associations collected from TTD database were subsequently used to build the associations between the plant and its targeted human diseases.
❷ Association by Disease Gene Reversion: Plant and a specific disease will be associated when >= 1 plant target gene overlaped with disease's DEGs.
❸ Association by Clinical Trials of Plant: Plant and a specific disease will be associated when >= 1 clinical trial (the plant is the intervetion) can be matched in ClinicalTrials.gov database.
❹ Association by Clinical Trials of Plant Ingredients: Plant and a specific disease will be associated when >= 1 clinical trial (the plant ingredient is the intervetion) can be matched in ClinicalTrials.gov database.
Associated Disease of the Plant |
Association Type & Detailed Evidence |
|---|---|
LeukaemiaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A60-2B33 |
NCT00005977,NCT00004904,NCT00100477,NCT00005863,NCT00002514,NCT00002925,NCT00002588,NCT00002756,NCT00002567,NCT00002757,NCT00070174,NCT00764907,NCT00003816,NCT00400946,NCT00002719,NCT00052299,NCT02660762,NCT03273829,NCT00002532,NCT00109031,NCT00458848,NCT01900509,NCT00430118,NCT00002701,NCT01154439,NCT00003602,NCT00002674,NCT00006040,NCT00002912,NCT00002598,NCT00025402,NCT00002517,NCT02385110,NCT00002926,NCT01117441,NCT00427791,NCT00410423,NCT00002638,NCT00039130,NCT00002658,NCT00002471,NCT00002704,NCT00002816,NCT00005823,NCT00002549,NCT00006045,NCT00004128,NCT00613457,NCT00003783,NCT00002865,NCT00005802,NCT00372593,NCT00002868,NCT00003436,NCT00002531,NCT00003727,NCT00882076,NCT00040690,NCT00003243,NCT00002788,NCT00304018,NCT00576979,NCT00630565,NCT00005793,NCT00005946,NCT00002688,NCT00002945,NCT00124644,NCT00008190,NCT00536978,NCT00002494,NCT02512926,NCT00003728,NCT00006122,NCT00002785,NCT00002768,NCT02356159,NCT00002766,NCT00004086,NCT00002800,NCT00534469,NCT00350181,NCT00002961,NCT00004898,NCT00618501,NCT00274807,NCT00003402,NCT00004905,NCT00002805,NCT00018954
|
Unspecified malignant neoplasms of unspecified sitesDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D4Z |
NCT00002854,NCT02937116,NCT02574728,NCT02018861,NCT00881166,NCT00003597,NCT01604863,NCT04778410,NCT00109031
|
Burkitt lymphoma, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH4KA9 |
NCT01092182,NCT02199184,NCT05270057,NCT00058461,NCT00126191,NCT00057811,NCT00073918,NCT00002649,NCT05389423
|
Mature B-cell lymphomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A85 |
TOP2B
NCT03206671,NCT00968760,NCT02443077,NCT01300793,NCT00324467,NCT02405676,NCT00054665 |
Neuroendocrine carcinomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C34 |
NCT00094497,NCT05142865,NCT03168594,NCT03992911,NCT05076786,NCT00193531,NCT00001339,NCT00003558
|
Primary neoplasms of brainDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A00 |
NCT00003935,NCT00003625,NCT00027846,NCT00003141,NCT00085098,NCT00003573,NCT00078988,NCT02905110
|
Primary haemophagocytic lymphohistiocytosisDisease Category: 04.Diseases of the immune systemDisease ICD-11 Code: 4A01.23 |
NCT03742115,NCT01104025,NCT02631109,NCT04077905,NCT00426101,NCT01818908,NCT02912702,NCT04551131
|
Classical Hodgkin lymphomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B30.1 |
NCT05008224,NCT04044222,NCT03077828,NCT02166463,NCT02243436,NCT02661503,NCT00967369,NCT00025259
|
Solid tumour/cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A00-2F9Z |
CA14,CA9,CA1,TOP2B,CA2,CA12,NOS2
|
Peripheral T-cell lymphoma, not otherwise specifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A90.C |
NCT05458180,NCT03586999,NCT04061772,NCT03321890,NCT00970385,NCT00791947,NCT00632827
|
Carcinosarcoma of uterusDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C76.43 |
CA2,CA14,CASP3,CA9,NOS2,NOS1,CA7
|
Diffuse large B-cell lymphomasDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A81 |
CA12,CASP3,CA9,NOS2,NOS3,NCOA3,TOP2B
|
Follicular lymphomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A80 |
TOP2B
NCT03019640,NCT00086944,NCT02592876,NCT00002649,NCT00073918 |
Malignant lymphoma, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH5FJ5 |
NCT00002524,NCT01775475,NCT00310128,NCT00002571,NCT00001563,NCT00049036
|
GerminomaDisease Category: X.Extension CodesDisease ICD-11 Code: XH1E13 |
NCT00002596,NCT02582697,NCT01873326,NCT02782754,NCT00864318,NCT00274950
|
Germ cell tumour of testisDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C80.2 |
CA12,CA14,NOS2,NCOA3,TOP2B
NCT00053352 |
OsteosarcomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B51 |
NCT04154189,NCT01258634,NCT00023998,NCT00134030,NCT00504140,NCT00180908
|
Adenocarcinoma of pancreasDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C10.0 |
CA2,CA14,CASP3,CA9,NOS2,CA7
|
Cytomegaloviral diseaseDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D82 |
CA2,CA1,CA14,NOS2,NOS3,NOS1
|
Diffuse large B-cell lymphomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A81 |
TOP2B
NCT01181271,NCT00073918,NCT05348213,NCT00137995,NCT01959698,NCT02471911,NCT03072771,NCT03064867,NCT05364424,NCT01415765,NCT00086944,NCT03293173,NCT01555541,NCT02219737,NCT01914718,NCT05466318,NCT04002947,NCT00556127,NCT02412267,NCT03367143,NCT00058461,NCT03019640,NCT00776802,NCT01481272,NCT02636322,NCT04139304,NCT03349346,NCT03736616,NCT00002649,NCT02570542,NCT04833114,NCT01165112,NCT05221645,NCT05389423,NCT01092182,NCT02329080,NCT00591630,NCT01197560,NCT02592876,NCT03229616,NCT01226849,NCT00482053,NCT02142049,NCT00057811,NCT03383406,NCT01030900,NCT00499018,NCT03213977,NCT03018626,NCT02366663,NCT00529503,NCT02228512,NCT00689169 |
Acute myeloid leukaemiaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A60 |
TOP2B
NCT03591510,NCT02400281,NCT00186966,NCT04326439,NCT02632708,NCT00315705,NCT03164057,NCT02306291,NCT00079482,NCT01411267,NCT00136084,NCT03839446,NCT03568994,NCT00880243,NCT00369317,NCT00003190,NCT01729845,NCT02421939,NCT01681537,NCT01180322,NCT01830777,NCT00512252,NCT00002798,NCT02070458,NCT02848183,NCT00660036,NCT03118466,NCT02773732,NCT00146120,NCT03504410,NCT03182244,NCT02349178,NCT00151242,NCT02039726,NCT01025778,NCT03860844,NCT00151255,NCT00780104,NCT01237808,NCT03926624,NCT02638428,NCT00939653,NCT01027923,NCT00774046,NCT00906945,NCT00006363,NCT02631252,NCT03793478,NCT01677949,NCT02626338,NCT04293562,NCT00003190,NCT00703820 |
Lung cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25 |
TOP2B
NCT00005022,NCT00061919,NCT00066222,NCT00002822,NCT00003053,NCT00003284,NCT00021112,NCT00006374,NCT00003158,NCT00049218,NCT00002642,NCT00045617,NCT00807755,NCT00083161,NCT00003696,NCT00072527,NCT00616785,NCT00869752,NCT00045162,NCT00025272,NCT00003583,NCT00011921,NCT01134861,NCT03811002,NCT00002858,NCT00003860,NCT00621361,NCT00589056,NCT00003606,NCT00006012,NCT03432598,NCT00144989,NCT00041015,NCT00004186,NCT03567642,NCT00632853,NCT00002823,NCT00002550,NCT00041054,NCT01579929,NCT00033696,NCT00006487,NCT00003299,NCT00554463,NCT00660504,NCT00984997,NCT00003364,NCT00387699,NCT00004176,NCT00003812,NCT00726986,NCT00002887,NCT00062439 |
Malignant neoplasms of stomachDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B72 |
CA1,NOS2,TOP2B
NCT02338518,NCT02949258 |
Anaplastic large cell lymphoma, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH1LC0 |
NCT00310128,NCT00354107,NCT01979536,NCT00057811,NCT00073918
|
HepatoblastomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C12.01 |
CA12,CA1,NOS2,NCOA3
NCT03017326 |
Serous cystadenoma,borderline malignancy of ovaryDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C73.4 |
CA12,CA2,CASP3,CA9,NOS1
|
Hepatocellular carcinoma of liverDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C12.02 |
CA12,NOS2,CA9,CA12,CA9
|
Glioblastoma of brainDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A00.00 |
CA12,CA2,CASP3,CA9,NOS2
|
Neuroblastoma, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH85Z0 |
NCT00004188,NCT00040872,NCT05338541,NCT03061656,NCT01857934,NCT00017368,NCT00070200,NCT00578864,NCT00135135,NCT00539500,NCT00025623,NCT02176967,NCT00793845,NCT00003141,NCT03786783,NCT00004110,NCT00017225,NCT00416676,NCT00030719,NCT00025610,NCT00002634,NCT00567567,NCT00025649,NCT02605421,NCT00808899,NCT00024193,NCT00365755,NCT00499616,NCT00003093,NCT00798148,NCT00002740,NCT01059071,NCT00002802,NCT00025428,NCT01704716,NCT00253435,NCT00025597,NCT02771743,NCT00003119,NCT00186849,NCT00005978,NCT04221035,NCT00276731
|
Precursor cell lymphoblastic leukaemia, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH5J37 |
NCT01411267,NCT00131027,NCT02349178,NCT00137111,NCT01331590,NCT00315705,NCT03991884,NCT02553460,NCT03117751,NCT00187161,NCT03020030,NCT01949129,NCT03860844,NCT03023046,NCT00022737,NCT03643276,NCT00098839,NCT00549848,NCT03553238,NCT02043587,NCT05192889,NCT02518750,NCT02003222,NCT02883049,NCT01025778,NCT00187005,NCT02101853,NCT02199184,NCT01677949,NCT02881086,NCT00476190,NCT02776605,NCT05453500,NCT00186875,NCT01423500,NCT00187083,NCT01423747,NCT00057811,NCT01457040,NCT00720109
|
Rheumatoid arthritisDisease Category: 15.Diseases of the musculoskeletal system or connective tissueDisease ICD-11 Code: FA20 |
NOS2,CA1,CA2,TRPV1
|
Bacterial infectionDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1A00-1C4Z |
TOP2B,CA12,CA14,CA2
|
Malignant haematopoietic neoplasmDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B33 |
TOP2B
NCT00051311,NCT03531281,NCT04177004 |
Acute monocytic leukaemiaDisease Category: X.Extension CodesDisease ICD-11 Code: XH9NE2 |
NCT00003190,NCT00006363,NCT00369317,NCT00002798
|
Acute erythroid leukaemiaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A60.35 |
NCT00003190,NCT00002798,NCT00006363,NCT00369317
|
Malignant neoplasms of kidney, except renal pelvisDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C90 |
NCT00025103,NCT00047138,NCT00002610,NCT00002611
|
Acute myelomonocytic leukaemiaDisease Category: X.Extension CodesDisease ICD-11 Code: XH78Y4 |
NCT00003190,NCT00002798,NCT00369317,NCT00006363
|
Acute megakaryoblastic leukaemiaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A60.36 |
NCT00002798,NCT00369317,NCT00003190,NCT00006363
|
Other specified malignant neoplasms of kidney, except renal pelvisDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C90.Y |
CA9,CA1,CA9
NCT04322318 |
PheochromocytomaDisease Category: X.Extension CodesDisease ICD-11 Code: XH3854 |
NOS1,NOS2,CA14,CA2
|
Squamous cell carcinoma of bronchus or lungDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25.2 |
CA12,CA9,NOS2,CA7
|
Mesothelioma of pleuraDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C26.0 |
CA12,CASP3,CA9,CA7
|
Malignant lymphoma, not elsewhere classifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B33.5 |
NCT02371161,NCT00367497,NCT00691210,NCT00481832,NCT03794167,NCT01900509,NCT01097057,NCT00504751,NCT02356159,NCT00350181,NCT00324467,NCT01300793,NCT02987244,NCT03458260,NCT01178645,NCT00193505,NCT02703272,NCT01840566,NCT00262210,NCT01178658,NCT03623087,NCT00100477,NCT03792815,NCT01746173,NCT03161054,NCT01458366,NCT00088530,NCT00250718,NCT02408042,NCT00809341,NCT02836639,NCT00712582
|
Non-small cell lung cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25 |
NCT02947113,NCT00776698,NCT01565772,NCT00924209,NCT01076231,NCT01993810,NCT03840902,NCT01733589,NCT02940990,NCT02768558,NCT04989283,NCT00240097,NCT04372927,NCT00682383,NCT02262325,NCT04203511,NCT00985855,NCT03519971,NCT04092283,NCT03663166,NCT02733107,NCT03944772,NCT00216125,NCT00686959,NCT00191139,NCT01822496,NCT04380636,NCT05298423,NCT00417248,NCT01222572,NCT02987998,NCT01411098
|
Hodgkin lymphomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B30 |
NCT02408042,NCT02686346,NCT00439361,NCT01181271,NCT01920932,NCT03407144,NCT01251107,NCT00667615,NCT01468740,NCT01165112,NCT02356159,NCT04638790,NCT03016871,NCT02227199,NCT00691210,NCT04871607,NCT03517137,NCT00388349,NCT02298283,NCT03755804,NCT04981899,NCT01900509,NCT01169636,NCT00225173,NCT03418038,NCT01476839,NCT01569204,NCT01030900,NCT01356680,NCT00026208,NCT00352027
|
Multiple myelomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A83 |
NCT03004287,NCT00003401,NCT00304018,NCT00572169,NCT02504359,NCT00004903,NCT00002552,NCT00871013,NCT00003399,NCT00083551,NCT01548573,NCT00083681,NCT00006184,NCT01653418,NCT00869232,NCT00005834,NCT03570983,NCT02356159,NCT03385096,NCT05294055,NCT00005987,NCT00349778,NCT00083915,NCT00574080,NCT01055301,NCT00083538,NCT00002657,NCT00004904,NCT00005946,NCT00734877
|
Seborrhoeic dermatitisDisease Category: 14.Diseases of the skinDisease ICD-11 Code: EA81 |
CA1,CA12,CA2
|
Colorectal cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B91 |
TOP2B
NCT03100955,NCT03769935 |
AsthmaDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CA23 |
TRPV1,TOP2B,NOS2
|
Brain cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A00 |
CASP3,TOP2B
NCT01014767 |
Soft tissue disorderDisease Category: 15.Diseases of the musculoskeletal system or connective tissueDisease ICD-11 Code: FB56 |
NCT02784015,NCT01710176,NCT00876031
|
CNS embryonal tumour, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH8SH6 |
NCT00006461,NCT00936156,NCT00336024
|
Seminoma, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH9FM4 |
NCT02341989,NCT01887340,NCT03937843
|
Extranodal NK/T-cell lymphoma, nasal typeDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A90.6 |
NCT04917250,NCT02825147,NCT02631239
|
Adrenal cortical carcinomaDisease Category: X.Extension CodesDisease ICD-11 Code: XH4KH2 |
NCT03723941,NCT00304070,NCT03583710
|
GliosarcomaDisease Category: X.Extension CodesDisease ICD-11 Code: XH9RC8 |
NCT02502708,NCT00612430,NCT00613223
|
Ewing sarcomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B52 |
NCT01864109,NCT02063022,NCT00568464
|
Adult T-cell lymphoma or leukaemia, human T-cell lymphotropic virus type 1-associatedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A90.5 |
NCT01000285,NCT02538926,NCT03007147
|
AdenocarcinomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D40 |
NCT00042835,NCT01958372,NCT00020709
|
Squamous cell carcinomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B60-2D01 |
NCT00020709,NCT00042835,NCT01947062
|
Chronic lymphocytic leukaemia or small lymphocytic lymphomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A82.0 |
NCT01181271,NCT00275015,NCT00276809
|
Myeloproliferative neoplasmDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A20 |
NCT00005863,NCT00006040,NCT00005823
|
Rhabdomyosarcoma, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH0GA1 |
NCT01871766,NCT00025363,NCT00162695
|
Adenocarcinoma of bronchus or lungDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25.0 |
CA12,CA9,CA7
|
Malignant neoplasms of adrenal glandDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D11 |
CA12,CA9,CA7
|
Breast cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C60-2C6Y |
CA12,TOP2B,CA14,CA9,CA1,CA2
NCT01589159,NCT00006032,NCT01492556,NCT00002509,NCT00020722,NCT00002836,NCT00004906,NCT00012311,NCT00693719,NCT00004900,NCT00810017,NCT04452370,NCT00006260,NCT03923179,NCT03535961 |
Pleomorphic sarcomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B54 |
NCT00354744,NCT01294670,NCT00470223,NCT00006734,NCT00379457,NCT00003141,NCT00043979,NCT00061893,NCT00077285,NCT00002936,NCT00003937,NCT00014313,NCT00204646,NCT00003052,NCT00334867,NCT00002516,NCT00072280,NCT00002974,NCT00742924,NCT00020566
|
Coronary atherosclerosisDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA80 |
NOS3,NOS2
|
Kaposi sarcomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B57 |
TOP2B
NCT00000660 |
PsoriasisDisease Category: 14.Diseases of the skinDisease ICD-11 Code: EA90 |
TOP2B
NOS2 |
GlaucomaDisease Category: 09.Diseases of the visual systemDisease ICD-11 Code: 9C61 |
CA1,CA2
|
Pulmonary hypertensionDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BB01 |
NOS3,NOS3
|
MigraineDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8A80 |
TRPV1,NOS1
|
OsteoarthritisDisease Category: 15.Diseases of the musculoskeletal system or connective tissueDisease ICD-11 Code: FA00-FA05 |
TRPV1,NOS2
|
Pain, unspecifiedDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: MG3Z |
NOS2,TRPV1
|
Duodenal ulcerDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DA63 |
CA2,CA1
|
Graft-versus-host diseaseDisease Category: 04.Diseases of the immune systemDisease ICD-11 Code: 4B24 |
NCT00350181,NCT00004904
|
Malignant neoplasms of retroperitoneumDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C50 |
NCT00003380,NCT00003967
|
Castlemans diseaseDisease Category: 04.Diseases of the immune systemDisease ICD-11 Code: 4B2Y |
NCT02228512,NCT04585893
|
Glioblastoma, primary, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH0MB1 |
NCT02372409,NCT04765098
|
GanglioneuroblastomaDisease Category: X.Extension CodesDisease ICD-11 Code: XH77W7 |
NCT03786783,NCT02176967
|
Malignant neoplasms of thymusDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C27 |
CA9
NCT02633514 |
Other specified gliomas of brainDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A00.0Y |
NCT02265770,NCT01032070
|
Teratoma, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH83G5 |
NCT00053352,NCT00104676
|
Fallopian tube cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C74 |
NCT00003967,NCT00003380
|
Acute basophilic leukaemiaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A60.37 |
NCT00006363,NCT00369317
|
Lymphoid leukaemia, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH7Q12 |
NCT03328104,NCT02523976
|
Refractory anaemiaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A30 |
NCT00002798,NCT00209833
|
Nephroblastoma, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH5QN3 |
NCT05384821,NCT00379340
|
Myeloproliferative neoplasm, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH5HH7 |
NCT00003816,NCT00004904
|
Choroid plexus tumoursDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A00.22 |
NCT00500890,NCT01014767
|
Malignant neoplasms of biliary tract, distal bile ductDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C15 |
NOS2,CA9
|
Other specified malignant neoplasms of bronchus or lungDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25.Y |
CA12,CA9
|
Urothelial carcinoma of bladderDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C94.2 |
CA9,CA2
|
Chronic rhinosinusitisDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CA0A |
NOS2,CA9
|
Malignant neoplasms of thyroid gland, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D10.Z |
CA12,CA7
|
Malignant neoplasms of corpus uteri, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C76.Z |
CA9,CA2
|
Invasive carcinoma of breastDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C61 |
CA12,CA9
|
Renal cell carcinoma, chromophobe typeDisease Category: X.Extension CodesDisease ICD-11 Code: XH6153 |
CA12,CA2
|
Myelodysplastic syndromeDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A37 |
NCT00003816,NCT00004904,NCT00002517,NCT00002926,NCT00304018,NCT00005823,NCT03164057,NCT00003436,NCT01729845,NCT00003602,NCT00369317,NCT00005863,NCT00774046,NCT00006363,NCT00003243,NCT00006040,NCT02356159,NCT00002800,NCT00005946
|
NeoplasmsDisease Category: 02.NeoplasmsDisease ICD-11 Code: 02 |
NCT02794571,NCT04469530,NCT00573690,NCT00811993,NCT00193596,NCT00009815,NCT00006047,NCT00027898,NCT02964013,NCT00878449,NCT00653068,NCT02157792,NCT03628131,NCT04047862,NCT00003194,NCT00551005,NCT03617432
|
Mature T-cell lymphomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A90 |
CA9,TOP2B
NCT00003541,NCT00070187,NCT00079105,NCT00004010,NCT00598169,NCT00002488,NCT00472056,NCT00005964,NCT00032019,NCT00002766,NCT00039130,NCT00002987,NCT00908180,NCT00920153,NCT00002481,NCT00012051,NCT00004031,NCT00040690,NCT00007852,NCT00004086,NCT00002905,NCT00002714,NCT00002691,NCT00450801,NCT00117598,NCT00948090,NCT00003815,NCT00049439,NCT00829205,NCT00641381,NCT00005589,NCT00255723,NCT00577629,NCT00002941,NCT00499343,NCT00028665,NCT00060385,NCT00244946,NCT00005985,NCT01329900,NCT00002982,NCT00025064,NCT00398554,NCT05367856,NCT00521014,NCT00983944,NCT00002565,NCT00005613,NCT00004907,NCT02359162,NCT00006708,NCT00032149,NCT05006664,NCT00002510,NCT00217503,NCT00003243,NCT00392834,NCT00007865,NCT00264953,NCT00004898,NCT01046825,NCT00003421,NCT00003114,NCT00003632,NCT00002757,NCT00039195,NCT00098774,NCT00723658,NCT01516567,NCT00002936,NCT00004169,NCT00003389,NCT00058292,NCT00005631,NCT00002835,NCT00006669,NCT00002522,NCT01390584,NCT00003423,NCT00002557,NCT00416832,NCT00003816,NCT00003957,NCT00568607,NCT00002521,NCT00003402,NCT00002461,NCT00003631,NCT00003397,NCT00003728,NCT00002494,NCT00242996,NCT00822120,NCT00002827,NCT00002590,NCT00069238,NCT00003215,NCT00003578,NCT00801918,NCT00732498,NCT00005578,NCT00233987,NCT00016887,NCT00006695,NCT00005998,NCT00002471,NCT00439556,NCT00577993,NCT00006373,NCT02436707,NCT00274924,NCT00562978,NCT00720447,NCT00515138,NCT00002697,NCT00003262,NCT00695409,NCT00005977,NCT00933673,NCT00052923,NCT00109928,NCT00118209,NCT01063439,NCT00041327,NCT00053768,NCT00536978,NCT03188198,NCT00006455,NCT00569985,NCT00018954,NCT00020943,NCT00265031,NCT00041210,NCT00080886,NCT00005090,NCT00002829,NCT00003113,NCT01511562,NCT00666484,NCT00678327,NCT00049595,NCT00003650,NCT00559104,NCT00002715,NCT00265889,NCT00003575,NCT00109031,NCT00345865,NCT00005867,NCT00025636 |
RetinoblastomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D02.2 |
NCT02870907,NCT01783535,NCT01899066,NCT04681417,NCT00072384,NCT00335738,NCT00110110,NCT00186888,NCT00179920,NCT00004006,NCT02116959,NCT01906814,NCT00002675,NCT02137928,NCT00554788
|
Central nervous system diseaseDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8A04-8D87 |
NCT00278278,NCT00278252,NCT00003309,NCT00084838,NCT00003859,NCT00006258,NCT00392886,NCT00025324,NCT05124951,NCT00003621,NCT00004224,NCT00003996,NCT00047281,NCT00002876
|
Small cell carcinoma of bronchus or lungDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25.1 |
NCT05092412,NCT00658580,NCT04699838,NCT03059667,NCT03850067,NCT00927875,NCT04665856,NCT05384015,NCT04256421,NCT01859741,NCT02580994,NCT04702880,NCT03781869,NCT00320359,NCT03613597,NCT03066778,NCT02722369,NCT04660097,NCT00958022,NCT05091567,NCT02337712,NCT04740021,NCT03711305,NCT00916669,NCT02763579,NCT00066742,NCT04363255,NCT04730999,NCT00079040,NCT03135977,NCT05403723,NCT01555710,NCT00702962,NCT04624204,NCT04422210,NCT02402920,NCT02748889,NCT01064466,NCT05116007,NCT03041311,NCT04675697,NCT05468489,NCT00363415,NCT00042978,NCT04346914,NCT00388960,NCT04189094,NCT00613626,NCT00453154,NCT04996771,NCT00826644,NCT00401609,NCT00544596,NCT05001412,NCT04878016,NCT05245994,NCT04728230,NCT03467360,NCT01563601,NCT05354700,NCT05353257,NCT00926640,NCT02934503,NCT01722292,NCT03700359,NCT04543890,NCT02499770,NCT03699956,NCT04560972,NCT04012606,NCT01573338,NCT00043927,NCT02179528,NCT02949895,NCT05224141,NCT03043872,NCT04923776,NCT00537511,NCT02899728,NCT04622228,NCT01450761,NCT05026593,NCT04712903,NCT00887159,NCT04696575,NCT00017251,NCT04234607,NCT04005716,NCT02738346,NCT00977561,NCT04951115,NCT03116971,NCT02289690,NCT04745689,NCT04101357,NCT04254471,NCT02819999,NCT01439568,NCT04028050,NCT04696939,NCT04683198,NCT00143455,NCT05142696,NCT02194049,NCT01237678,NCT00791154,NCT00912392,NCT00812266,NCT04490421,NCT04684017,NCT05309629,NCT05361395,NCT02815592,NCT05484583,NCT03389087,NCT04542369,NCT01987232,NCT04472949,NCT00717938,NCT02875457,NCT00682981,NCT05161533,NCT00526396,NCT04774380,NCT00168896,NCT03382561,NCT04924101,NCT00057837,NCT04063163,NCT02323737
|
Ovarian cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C73 |
TOP2B
NCT04520074,NCT00003380,NCT02562365,NCT05422183,NCT04000295,NCT03298074,NCT03901118,NCT00053352,NCT02867956,NCT00005612,NCT00274950,NCT00003967 |
Precursor B-lymphoblastic neoplasmDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A70 |
NCT03328104,NCT02881086,NCT02845882,NCT00073918,NCT02043587,NCT01451515,NCT03023046,NCT00058461,NCT03564704,NCT00195871,NCT03991884
|
Prostate cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C82 |
NCT00627354,NCT00514540,NCT00004054,NCT00028769,NCT00003084,NCT00151060,NCT02861573,NCT00483561,NCT00176605,NCT00973882
|
Mantle cell lymphomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A85.5 |
NCT00878254,NCT00114738,NCT01181271,NCT00073918,NCT00209222,NCT00571493,NCT02896582,NCT00086944,NCT00005780,NCT03019640
|
Medulloblastoma, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH8P29 |
NCT02502708,NCT04501718,NCT01857453,NCT00749723,NCT01356290,NCT02025881,NCT00180791,NCT00031590,NCT00006461,NCT00336024
|
Testicular cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C80 |
NCT00002566,NCT00053352,NCT00936936,NCT02341989,NCT00104676,NCT00231582,NCT00003941,NCT00324298,NCT03937843,NCT00002596
|
Lower limb mononeuropathyDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8C11 |
TRPV1
|
Heart failureDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BD10-BD1Z |
CA2
|
SyphilisDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1A61-1A6Z |
TOP2B
|
Myocardial infarctionDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA41-BA43 |
NOS3
|
Alzheimer diseaseDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8A20 |
TOP2B
|
Angina pectorisDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA40 |
NOS3
|
Anogenital wartsDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1A95 |
TOP2B
|
Visual system diseaseDisease Category: 09.Diseases of the visual systemDisease ICD-11 Code: 9E1Z |
TRPV1
|
General pain disorderDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8E43 |
TRPV1
|
Atopic eczemaDisease Category: 14.Diseases of the skinDisease ICD-11 Code: EA80 |
TRPV1
|
Pancreatic cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C10 |
TOP2B
|
Respiratory system diseaseDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CB40-CB7Z |
TOP2B
|
HypotensionDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA20-BA21 |
NOS3
|
Chronic painDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: MG30 |
TRPV1
|
Liver cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C12 |
TOP2B
|
Otitis externaDisease Category: 10.Diseases of the ear or mastoid processDisease ICD-11 Code: AA00-AA13 |
TOP2B
|
HIV-infected patients with tuberculosisDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1B10-1B14 |
TOP2B
|
Urinary tract infectionDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GC08 |
TOP2B
|
Low bone mass disorderDisease Category: 15.Diseases of the musculoskeletal system or connective tissueDisease ICD-11 Code: FB83 |
CA2
|
Stomach cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B72 |
TOP2B
|
SepsisDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1G40-1G41 |
NOS2
|
Common wartsDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1E80 |
TOP2B
|
Chronic kidney diseaseDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GB61 |
NOS2
|
Intracranial injuryDisease Category: 22.Injury, poisoning or certain other consequences of external causesDisease ICD-11 Code: NA07 |
NOS3
|
InsomniaDisease Category: 07.Sleep-wake disordersDisease ICD-11 Code: 7A00-7A0Z |
CA2
|
Inborn metabolism deficiencyDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5C50-5C59 |
CA14
|
ThrombosisDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DB61-GB90 |
TOP2B
|
Cardiovascular diseaseDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA00-BE2Z |
NOS2
|
Renal cell carcinomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C90 |
CA9
|
Multiple sclerosisDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8A40 |
TOP2B
|
Insulin-resistance syndromeDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A44 |
NOS2
|
Knee osteoarthritisDisease Category: 15.Diseases of the musculoskeletal system or connective tissueDisease ICD-11 Code: FA01 |
TRPV1
|
Functional bladder disorderDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GC50 |
TRPV1
|
RetinopathyDisease Category: 09.Diseases of the visual systemDisease ICD-11 Code: 9B71 |
NOS2
|
Bowel habit changeDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: ME05 |
NOS2
|
Tension-type headacheDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8A81 |
NOS1
|
Otitis mediaDisease Category: 10.Diseases of the ear or mastoid processDisease ICD-11 Code: AA80-AB0Z |
TOP2B
|
ChemoprotectionDisease Category: NADisease ICD-11 Code: N.A. |
TOP2B
|
Bladder cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C94 |
TOP2B
|
Gonococcal infectionDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1A70-1A7Z |
TOP2B
|
Essential hypertensionDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA00 |
CA2
|
Chronic inflammatory demyelinating polyneuropathyDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8C01.3 |
NCT00716066
|
Neuroendocrine neoplasms of pancreasDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C10.1 |
NCT02595424
|
Embryonal carcinoma, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH8MB9 |
NCT00053352
|
Astrocytoma, anaplasticDisease Category: X.Extension CodesDisease ICD-11 Code: XH96C7 |
NCT02372409
|
Mature B-cell neoplasms, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A8Z |
NCT02911142
|
Inappropriate saccadesDisease Category: 09.Diseases of the visual systemDisease ICD-11 Code: 9C85.02 |
NCT00716066
|
Myeloproliferative and myelodysplastic disease, unclassifiableDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A44 |
NCT00003243
|
Metastatic digestive system neoplasmDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D8Y |
NCT03980925
|
Congenital human immunodeficiency virus infectionDisease Category: 19.Certain conditions originating in the perinatal periodDisease ICD-11 Code: KA62.6 |
NCT02199184
|
Myeloid sarcomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A60.39 |
NCT01371981
|
Waldenstrom macroglobulinemiaDisease Category: X.Extension CodesDisease ICD-11 Code: XH8GW4 |
NCT00073918
|
OligoastrocytomaDisease Category: X.Extension CodesDisease ICD-11 Code: XH6F49 |
NCT02372409
|
Human immunodeficiency virus type 1Disease Category: X.Extension CodesDisease ICD-11 Code: XN8LD |
NCT01435018
|
T lymphoblastic leukaemia/lymphomaDisease Category: X.Extension CodesDisease ICD-11 Code: XH50W7 |
NCT02538926
|
Oligodendroglioma, anaplasticDisease Category: X.Extension CodesDisease ICD-11 Code: XH8844 |
NCT00303849
|
Adult T-cell leukaemia/lymphoma (HTLV-1 positive)Disease Category: X.Extension CodesDisease ICD-11 Code: XH6TE2 |
NCT00145002
|
Optic nerve disorderDisease Category: 09.Diseases of the visual systemDisease ICD-11 Code: 9C40 |
NCT02372409
|
Oligodendroglioma, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH7W59 |
NCT02372409
|
Malignant neoplasm of pancreas, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C10.Z |
NCT00202800
|
Glioma, malignantDisease Category: X.Extension CodesDisease ICD-11 Code: XH4RQ3 |
NCT01756989
|
Chronic myeloid leukaemia, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH4XG8 |
NCT00780104
|
Paraneoplastic or autoimmune disorders of the central nervous system, brain or spinal cordDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8E4A.0 |
NCT00716066
|
NeutropeniaDisease Category: 04.Diseases of the immune systemDisease ICD-11 Code: 4B00 |
NCT00002800
|
Neoplasms of unknown behaviour of urinary organsDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2F98 |
NCT03669783
|
Skin cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C30-2C37 |
NCT01013779
|
Relapsing-remitting multiple sclerosisDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8A40.0 |
NCT00288626
|
Embryonal rhabdomyosarcoma, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH83G1 |
NCT01055314
|
Pilocytic astrocytomaDisease Category: X.Extension CodesDisease ICD-11 Code: XH12D2 |
NCT02372409
|
Refractory anaemia with excess of blastsDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A35 |
NCT00002798
|
Tumours of neuroepithelial tissue of brainDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A00.2 |
NCT02653196
|
Malignant neoplasms of oropharynxDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B6A |
NCT01525927
|
Small cell carcinoma, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH0YB0 |
NCT05058651
|
ThymomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C27 |
NCT00387868
|
Malignant trophoblastic neoplasms of placentaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C75.0 |
NCT02639650
|
Diseases of the blood or blood-forming organs, unspecifiedDisease Category: 03.Diseases of the blood or blood-forming organsDisease ICD-11 Code: 3C0Z |
NCT01220297
|
COVID-19Disease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D6Y |
NCT04356690
|
Alveolar rhabdomyosarcomaDisease Category: X.Extension CodesDisease ICD-11 Code: XH7099 |
NCT01055314
|
Malignant neoplasm metastasis in mediastinumDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D71 |
NCT00003941
|
Ependymoma, anaplasticDisease Category: X.Extension CodesDisease ICD-11 Code: XH6922 |
NCT01096368
|
Hepatocellular carcinoma, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH4W48 |
NCT00351195
|
Chronic myelomonocytic leukaemiaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A40 |
NCT00002798
|
Other specified malignant neoplasms of the ovaryDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C73.Y |
NCT02429700
|
Myasthenia gravisDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8C6Y |
NCT00716066
|
Mature B-cell leukaemiaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A82 |
NCT02504359
|
Extranodal marginal zone B-cell lymphoma of mucosa-associated lymphoid tissue of stomachDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A85.1 |
NCT00073918
|
Neuromyelitis opticaDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8A43 |
NCT00716066
|
Ependymoma, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH1511 |
NCT02502708
|
Crohn diseaseDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DD70 |
NOS2
|
Inflammatory bowel diseases, unspecifiedDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DD7Z |
NOS2
|
Osteoarthritis, unspecifiedDisease Category: 15.Diseases of the musculoskeletal system or connective tissueDisease ICD-11 Code: FA0Z |
CA12
|
Atrial fibrillationDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BC81.3 |
CA7
|
Malignant neoplasm metastasis in lymph nodes of head, face or neckDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D60.0 |
CA9
|
Intrauterine growth restrictionDisease Category: 19.Certain conditions originating in the perinatal periodDisease ICD-11 Code: KA20.1 |
NOS2
|
Pre-eclampsiaDisease Category: 18.Pregnancy, childbirth or the puerperiumDisease ICD-11 Code: JA24 |
CA1
|
Tetralogy of FallotDisease Category: 20.Developmental anomaliesDisease ICD-11 Code: LA88.2 |
CA14
|
Other specified malignant neoplasms ofcolonDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B90.Y |
CA9
|
Melanoma of skinDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C30 |
NOS2
|
Superficial ovarian endometriosisDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GA10.B4 |
CA2
|
Malignant neoplasms of oesophagusDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B70 |
NOS2
|
Systemic lupus erythematosusDisease Category: 04.Diseases of the immune systemDisease ICD-11 Code: 4A40.0 |
NOS2
|
Chikungunya virus diseaseDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D40 |
NOS1
|
Carpal tunnel syndromeDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8C10.0 |
NOS2
|
Lyme borreliosisDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1C1G |
CA1
|

