Collective Molecular Activities of the Plant: Amomum Tsao-Ko
Plant ID: NPO40539
Plant Latin Name: Amomum Tsao-Ko
Taxonomy Genus: Lanxangia
Taxonomy Family: Zingiberaceae
Plant External Links:
NCBI TaxonomyDB:
252867
Plant-of-the-World-Online:
n.a.
Overview of Ingredients
13 All known Ingredients in Total
Unique ingredients have been isolated from this plant.Plant-Ingredients Associations were manually curated from publications or collected from other databases.
7 Ingredients with Acceptable Bioavailablity
Unique ingredients exhibit acceptable human oral bioavailablity, according to the criteria of SwissADME [PMID: 28256516] and HobPre [PMID: 34991690]. The criteria details:SwissADME: six descriptors are used by SwissADME to evaluate the oral bioavailability of a natural product:
☑ LIPO(Lipophility): -0.7 < XLOGP3 < +5.0
☑ SIZE: 150g/mol < MW < 500g/mol
☑ POLAR(Polarity): 20Ų < TPSA < 130Ų
☑ INSOLU(Insolubility): -6 < Log S (ESOL) < 0
☑ INSATU(Insaturation): 0.25 < Fraction Csp3 < 1
☑ FLEX(Flexibility): 0 < Num. rotatable bonds < 9
If 6 descriptors of a natural plant satisfy the above rules, it will be labeled high HOB.
HobPre: A natural plant ingredient with HobPre score >0.5 is labeled high human oral availability (HOB)
12 Ingredients with experimental-derived Activity
Unique ingredients have activity data available.Ingredient Structrual Cards
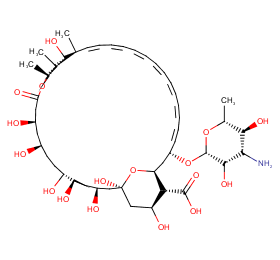
Ingredient ID: NPC67917
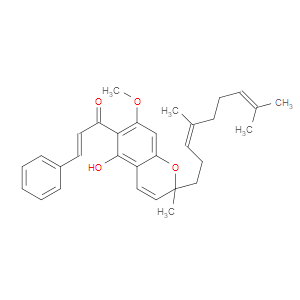
Ingredient ID: NPC488436
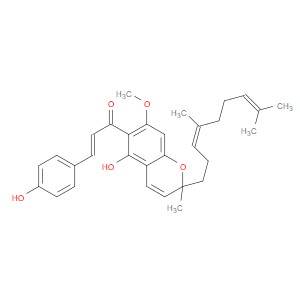
Ingredient ID: NPC488435
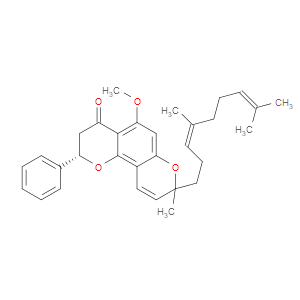
Ingredient ID: NPC488434
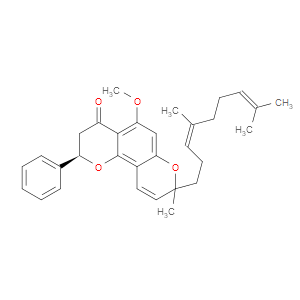
Ingredient ID: NPC488433
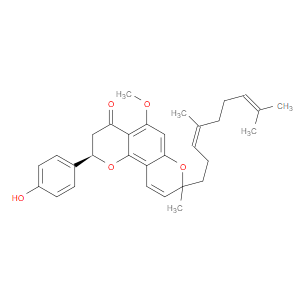
Ingredient ID: NPC488431
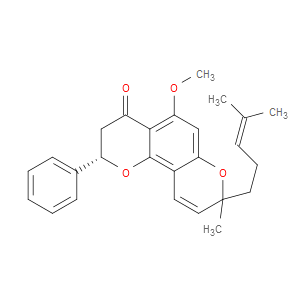
Ingredient ID: NPC488430
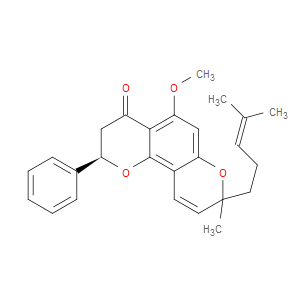
Ingredient ID: NPC488429
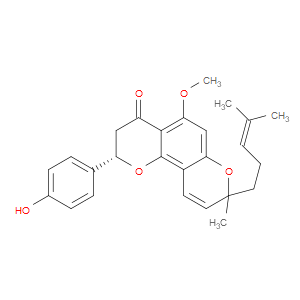
Ingredient ID: NPC488428
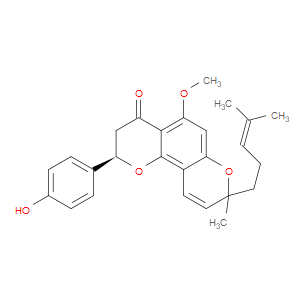
Ingredient ID: NPC488427

Ingredient ID: NPC482298
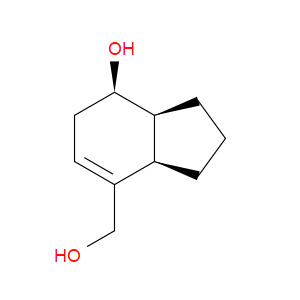
Ingredient ID: NPC479924
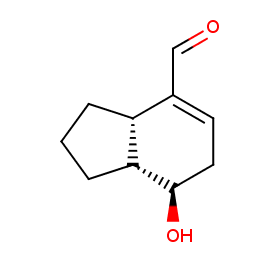
Ingredient ID: NPC242722
Classification of Human Proteins Collectively Targeted by the Plant
Detailed Information of Target Proteins
| Target Type | Protein Class | Gene ID | Protein Name | Uniprot ID | Target ChEMBL ID |
|---|---|---|---|---|---|
| Therapeutic Target | Structural protein | LMNA | Prelamin-A/C | P02545 | CHEMBL1293235 |
Clinical trials associated with plant from natural product (NP) & plant level:
| Clinical trials type | Number of clinical trials | |
|---|---|---|
| 69 | ||
| NCT ID | Title | Condition | Form in clinical use | Associated by plant or compound |
|---|---|---|---|---|
| NCT03945448 | Single Dose Liposomal Amphotericin for Asymptomatic Cryptococcal Antigenemia | cryptococcal meningitis | Amphotericin B (NPC67917) | |
| NCT00177710 | Pharmacokinetic Profile of Inhaled Liposomal Amphotericin B in Lung Transplant Recipients - Ambisome Study | fungal infectious disease | Amphotericin B (NPC67917) | |
| NCT00936910 | Antifungal Locks to Treat Fungal-related Central Line Infections | fungal infectious disease | Amphotericin B (NPC67917) | |
| NCT00832208 | Open-Label, Sequential Step, Safety and Efficacy Study to Determine the Optimal Single Dose of Ambisome for Patients With VL | visceral Leishmaniasis | Amphotericin B (NPC67917) | |
| NCT04502381 | Combined Inhalational With Intravenous Amphotericin B Versus Intravenous Amphotericin B Alone for Pulmonary Mucormycosis | mucormycosis | Amphotericin B (NPC67917) | |
| NCT00235651 | Abelcet Radiotagging Protocol: Inhaled Lipid Complex Abelcet® in Lung Transplant Recipients | lung disease | Amphotericin B (NPC67917) | |
| NCT00012467 | Safety and Antifungal Activity of Recombinant Interferon-Gamma 1b (rIFN-Gamma 1b) Given With Standard Therapy in Patients With Cryptococcal Meningitis | cryptococcal meningitis | Amphotericin B (NPC67917) | |
| NCT02686853 | Efficacy of Intrathecal Administration of Liposomal Amphotericin B in Cryptococcal Meningitis | cryptococcal meningitis | Amphotericin B (NPC67917) | |
| NCT00467883 | Pilot Study of High Dose Liposomal Amphotericin B Efficacy in Initial Zygomycosis Treatment | mucormycosis | Amphotericin B (NPC67917) | |
| NCT00145249 | Amphotericin Alone or in Combination With Fluconazole for AIDS-Associated Meningitis | cryptococcal meningitis | Amphotericin B (NPC67917) | |
| NCT00370825 | Combination Chemotherapy for the Treatment of Indian Kala-Azar | visceral Leishmaniasis | Amphotericin B (NPC67917) | |
| NCT03636659 | Steady State Global Bioequivalence Study of Amphotericin B Liposome for Injection 50 mg/ Vial in Fed Condition | visceral Leishmaniasis | Amphotericin B (NPC67917) | |
| NCT00002159 | A Randomized, Open, Comparative Multicenter Study of Initial Treatment With Intravenous Itraconazole Versus Amphotericin B Followed by Consolidation Treatment With Itraconazole Capsules in Patients With Blastomycosis or Histoplasmosis | blastomycosis;histoplasmosis | Amphotericin B (NPC67917) | |
| NCT00451711 | Intermittent Liposomal Amphotericin B Primary Prophylaxis | acute myeloid leukemia | Amphotericin B (NPC67917) | |
| NCT00418951 | Liposomal Amphotericin B (Ambisome) Versus Oral Voriconazole for the Prevention of Invasive Fungal Infections | acute myeloid leukemia;myelodysplastic syndrome | Amphotericin B (NPC67917) | |
| NCT00624143 | Antifungal Prophylaxis in Pediatric Acute Leukemia | leukemia | Amphotericin B (NPC67917) | |
| NCT00696969 | Safety and Efficacy Study to Evaluate Different Combination Treatment Regimens for Visceral Leishmaniasis | visceral Leishmaniasis | Amphotericin B (NPC67917) | |
| NCT00986713 | Value of Amphotericin B Inhalation for Prophylaxis of Invasive Pulmonary Aspergillosis After Renal Transplantation | aspergillosis | Amphotericin B (NPC67917) | |
| NCT00002742 | Antifungal Therapy for Fever and Neutropenia in Patients Receiving Treatment for Hematologic Cancer | chronic myeloproliferative disorder;leukemia;multiple myeloma;myelodysplastic syndrome;infection | Amphotericin B (NPC67917) | |
| NCT03187691 | Safety and PK of Oral Encochleated Amphotericin B (CAMB/MAT2203) for Antifungal Prophylaxis in Patients Undergoing Induction Chemotherapy for Acute Myelogenous and Lymphoblastic Leukaemia | acute lymphoblastic leukemia;acute myeloid leukemia | Amphotericin B (NPC67917) | |
| NCT00419770 | The Deferasirox-AmBisome Therapy for Mucormycosis (DEFEAT Mucor) Study | mucormycosis | Amphotericin B (NPC67917) | |
| NCT04031833 | Encochleated Oral Amphotericin for Cryptococcal Meningitis Trial (EnACT) | cryptococcal meningitis | Amphotericin B (NPC67917) | |
| NCT00008359 | Caspofungin Acetate Compared With Amphotericin B Liposomal in Treating Patients With Persistent Fever and Neutropenia Following Cancer Treatment | cancer | Amphotericin B (NPC67917) | |
| NCT00076869 | MK0991 in Combination With Standard Antifungal Agent(s) for the Treatment of Salvage Invasive Aspergillosis (0991-037) | aspergillosis | Amphotericin B (NPC67917) | |
| NCT00263315 | Inhalation of Liposomal Amphotericin B to Prevent Invasive Aspergillosis | aspergillosis | Amphotericin B (NPC67917) | |
| NCT00177684 | Pharmacokinetic Profiles of Inhaled Lipid Complex Amphotericin B (Abelcet ®) | fungal infectious disease | Amphotericin B (NPC67917) | |
| NCT00001107 | Comparison of Two Treatments to Prevent Invasive Fungal Infections in Patients Who Have Received Liver Transplants | candidiasis | Amphotericin B (NPC67917) | |
| NCT04267497 | Nebulised Liposomal Amphotericin for Invasive Pulmonary Aspergillosis (NAIFI01 Study) | aspergillosis | Amphotericin B (NPC67917) | |
| NCT00003315 | Liposomal Amphotericin B With or Without Sargramostim in Treating Patients With Invasive Fungal Infection | infection | Amphotericin B (NPC67917) | |
| NCT00002277 | A Comparison of Fluconazole and Amphotericin B in the Treatment of Fungal Infections | fungal infectious disease | Amphotericin B (NPC67917) | |
| NCT01945281 | Safety, Tolerability, and Efficacy of Caspofungin Versus Amphotericin B Deoxycholate in the Treatment of Invasive Candidiasis in Neonates and Infants (MK-0991-064) | Candidiasis, Invasive | Amphotericin B (NPC67917) | |
| NCT00815516 | Study to Compare the Efficacy and Safety of Micafungin Versus Conventional Amphotericin B for the Treatment of Neonatal Candidiasis | candidiasis | Amphotericin B (NPC67917) | |
| NCT02656797 | Topical Liposomal Amphotericin B Gel Treatment for Cutaneous Leishmaniasis | Leishmaniasis | Amphotericin B (NPC67917) | |
| NCT00003031 | Comparison of Voriconazole and Amphotericin B in Treating Patients With Aspergillosis | aspergillosis | Amphotericin B (NPC67917) | |
| NCT00001017 | Comparison of Fluconazole and Amphotericin B in the Treatment of Brain Infections in Patients With AIDS | cryptococcal meningitis | Amphotericin B (NPC67917) | |
| NCT00158730 | Study Safety/Efficacy of AmBisome Loading Dose Regimen Versus Standard AmBisome Regimen for Initial Treatment | aspergillosis | Amphotericin B (NPC67917) | |
| NCT00628719 | Single Infusion of Liposomal Amphotericin B in Indian Visceral Leishmaniasis | visceral Leishmaniasis | Amphotericin B (NPC67917) | |
| NCT00497601 | A Phase II Study To Assess Safety and Efficacy Of Short-Course Regimens Of Amphotericin B Emulsion In Kala-Azar | visceral Leishmaniasis | Amphotericin B (NPC67917) | |
| NCT01845727 | Topical 3% Amphotericin B Cream for the Treatment of Cutaneous Leishmaniasis in Colombia | cutaneous Leishmaniasis | Amphotericin B (NPC67917) | |
| NCT03196921 | Efficacy and Safety of Oral Encochleated Amphotericin B for the Treatment of Cryptococcal Infection | infection | Amphotericin B (NPC67917) | |
| NCT00002316 | The Safety and Effectiveness of RMP-7 Plus Amphotericin B in Patients With HIV and Cryptococcal Meningitis | cryptococcal meningitis | Amphotericin B (NPC67917) | |
| NCT00876824 | To Study the Effect Of Single Infusions Of Amphotericin B Lipid Preparations in Treatment of Patients Of Kala Azar | Leishmaniasis | Amphotericin B (NPC67917) | |
| NCT00847678 | Efficacy and Safety of Mycograb as Adjunctive Therapy for Cryptococcal Meningitis in Patients With AIDS | cryptococcal meningitis | Amphotericin B (NPC67917) | |
| NCT00163111 | A Clinical Study Intended To Compare Treatment With Voriconazole To Treatment With Amphotericin Followed By Fluconazole In Patients With Candidemia, A Serious Fungus Infection Of The Blood. | candidiasis | Amphotericin B (NPC67917) | |
| NCT01310738 | Efficacy and Safety Study of Drugs for Treatment of Visceral Leishmaniasis in Brazil | visceral Leishmaniasis | Amphotericin B (NPC67917) | |
| NCT01293643 | A Study to Compare the Efficacy and Safety of a Combination of Ketoconazole and Clindamycin Compared to Tetracycline/Amphotericin B Combination Cream for the Treatment of Bacterial Vaginosis and Vaginal Candidiasis (P08077) | bacterial vaginosis;vulvovaginal candidiasis | Amphotericin B (NPC67917) | |
| NCT00001646 | Voriconazole vs. Amphotericin B in the Treatment of Invasive Aspergillosis | aspergillosis | Amphotericin B (NPC67917) | |
| NCT01032187 | Amphotericin B to Treat Visceral Leishmaniasis in Brazilian Children | visceral Leishmaniasis | Amphotericin B (NPC67917) | |
| NCT02025491 | Liposomal Amphotericin in Disseminated Leishmaniasis | Leishmaniasis | Amphotericin B (NPC67917) | |
| NCT01566552 | Single Dose Liposomal Amphotericin B for Visceral Leishmaniasis | visceral Leishmaniasis | Amphotericin B (NPC67917) | |
| NCT00000776 | Dexamethasone in Cryptococcal Meningitis | intracranial hypertension;cryptococcal meningitis | Amphotericin B (NPC67917) | |
| NCT00106288 | Micafungin Versus AmBisome in Invasive Candidiasis and Candidemia | candidiasis | Amphotericin B (NPC67917) | |
| NCT03327727 | VL-2397 Compared to Standard First-Line Treatment for Invasive Aspergillosis (IA) in Adults | aspergillosis | Amphotericin B (NPC67917) | |
| NCT02011958 | Efficacy Trial of Ambisome Given Alone and Ambisome Given in Combination With Miltefosine for the Treatment of VL HIV Positive Ethiopian Patients. | visceral Leishmaniasis | Amphotericin B (NPC67917) | |
| NCT01122771 | Phase III, Study of Three Short Course Combo (Ambisome®, Miltefosine, Paromomycin) Compared With AmBisome for the Treatment of VL in Bangladesh | visceral Leishmaniasis | Amphotericin B (NPC67917) | |
| NCT01058174 | Liver Transplant European Study Into the Prevention of Fungal Infection | fungal infectious disease | Amphotericin B (NPC67917) | |
| NCT04018417 | Evaluation of Amphotericin B in Optisol-GS for Prevention of Post-Keratoplasty Fungal Infections. | Fuchs endothelial corneal dystrophy | Amphotericin B (NPC67917) | |
| NCT00523965 | Combination Therapy in Indian Visceral Leishmaniasis | Leishmaniasis | Amphotericin B (NPC67917) | |
| NCT00003938 | Liposomal Amphotericin B in Treating Granulocytopenia and Persistent Unexplained Fever in Cancer Patients | cancer | Amphotericin B (NPC67917) | |
| NCT05468372 | Amphotericin Versus Posaconazole for Pulmonary Mucormycosis | mucormycosis | Amphotericin B (NPC67917) | |
| NCT00047827 | Trial of Micafungin (FK463) in Combination With Liposomal Amphotericin B (AmBisome) for Aspergillosis | aspergillosis | Amphotericin B (NPC67917) | |
| NCT01857479 | A Randomized Controlled Trial of Inhaled Amphotericin B for Maintaining Remission in Allergic Bronchopulmonary Aspergillosis | allergic bronchopulmonary aspergillosis | Amphotericin B (NPC67917) | |
| NCT00371995 | Short Course of Miltefosine and Liposomal Amphotericin B for Kala-azar | visceral Leishmaniasis | Amphotericin B (NPC67917) | |
| NCT01437020 | SCH708980 With and Without AmBisome for Visceral Leishmaniasis | Leishmaniasis | Amphotericin B (NPC67917) | |
| NCT00216346 | Safety and Efficacy Study of Paromomycin to Treat Visceral Leishmaniasis | visceral Leishmaniasis | Amphotericin B (NPC67917) | |
| NCT01067443 | Clinical Trial to Assess the Safety and Efficacy of Sodium Stibogluconate (SSG) and AmBisome® Combination, Miltefosine and AmBisome® and Miltefosine Alone for the Treatment Visceral Leishmaniasis in Eastern Africa | visceral Leishmaniasis | Amphotericin B (NPC67917) | |
| NCT00334412 | COMBISTRAT: AmBisome® in Combination With Caspofungin for the Treatment of Invasive Aspergillosis | aspergillosis | Amphotericin B (NPC67917) | |
| NCT04799236 | Treatment of Mucosal Bolivian Leishmaniasis | mucocutaneous Leishmaniasis | Amphotericin B (NPC67917) | |
| NCT02629419 | CAMB/MAT2203 in Patients With Mucocutaneous Candidiasis | Chronic mucocutaneous candidosis | Amphotericin B (NPC67917) |
❱❱❱ Associated Human Diseases and Detailed Association Evidence
How do we define the Plant-Targeted Human Disease Association?
Associated human diseases of an individual plant are summurized based on FOUR types of association evidence, these include:
❶ Association by Therapeutic Target: Bioactive protein targets of the plant were defined in "Molecular Targets" section, target-disease associations collected from TTD database were subsequently used to build the associations between the plant and its targeted human diseases.
❷ Association by Disease Gene Reversion: Plant and a specific disease will be associated when >= 1 plant target gene overlaped with disease's DEGs.
❸ Association by Clinical Trials of Plant: Plant and a specific disease will be associated when >= 1 clinical trial (the plant is the intervetion) can be matched in ClinicalTrials.gov database.
❹ Association by Clinical Trials of Plant Ingredients: Plant and a specific disease will be associated when >= 1 clinical trial (the plant ingredient is the intervetion) can be matched in ClinicalTrials.gov database.
Associated Disease of the Plant |
Association Type & Detailed Evidence |
|---|---|
Meningitis due to Cryptococcus neoformansDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D01.10 |
NCT02686853,NCT04031833,NCT00145249,NCT00000776,NCT03945448,NCT00002316,NCT00012467,NCT00001017,NCT00847678
|
Skin fungal infection disorderDisease Category: 14.Diseases of the skinDisease ICD-11 Code: EA60 |
NCT00936910,NCT00177710,NCT01058174,NCT00177684,NCT00002277
|
CandidosisDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1F23 |
NCT00815516,NCT00001107,NCT00106288,NCT00163111,NCT01945281
|
MucormycosisDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1F2C |
NCT04502381,NCT05468372,NCT00419770,NCT00467883
|
Acute myeloid leukaemiaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A60 |
NCT03187691,NCT00451711,NCT00418951
|
Herpes simplex infectionDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1F00 |
NCT03196921,NCT00002742,NCT00003315
|
LeishmaniasisDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1F54 |
NCT01122771,NCT02656797,NCT00696969,NCT01032187,NCT01566552,NCT00523965,NCT00628719,NCT00216346,NCT00497601,NCT00371995,NCT03636659,NCT04799236,NCT00832208,NCT00370825,NCT00876824,NCT01437020,NCT01845727,NCT01067443,NCT01310738,NCT02011958,NCT02025491
|
Unspecified malignant neoplasms of unspecified sitesDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D4Z |
NCT00008359,NCT00003938
|
Vaginal dischargeDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: MF3A |
NCT01293643,NCT01293643
|
Myelodysplastic syndromeDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A37 |
NCT00418951,NCT00002742
|
LeukaemiaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A60-2B33 |
NCT00624143,NCT00002742
|
Mesothelioma of pleuraDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C26.0 |
LMNA,AOC1
|
Glioblastoma of brainDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A00.00 |
LMNA,AOC1
|
AspergillosisDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1F20 |
NCT00003031,NCT00001646,NCT00263315,NCT00076869,NCT04267497,NCT00986713,NCT03327727,NCT00158730,NCT00334412,NCT00047827
|
HistoplasmosisDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1F2A |
NCT00002159
|
NADisease Category: NADisease ICD-11 Code: NA |
NCT00235651
|
Endothelial corneal dystrophyDisease Category: 09.Diseases of the visual systemDisease ICD-11 Code: 9A70.0 |
NCT04018417
|
Aspergillus-induced allergic or hypersensitivity conditionsDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CA82.4 |
NCT01857479
|
Multiple myelomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A83 |
NCT00002742
|
Myeloproliferative neoplasm, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH5HH7 |
NCT00002742
|
Chronic mucocutaneous candidosisDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1F23.14 |
NCT02629419
|
BlastomycosisDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1F22 |
NCT00002159
|
Other specified increased intracranial pressureDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8D60.Y |
NCT00000776
|
Precursor cell lymphoblastic leukaemia, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH5J37 |
NCT03187691
|
Carcinosarcoma of uterusDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C76.43 |
AOC1
|
Serous cystadenoma,borderline malignancy of ovaryDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C73.4 |
AOC1
|
PheochromocytomaDisease Category: X.Extension CodesDisease ICD-11 Code: XH3854 |
AOC1
|
Malignant neoplasms of biliary tract, distal bile ductDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C15 |
LMNA
|
Idiopathic pulmonary fibrosisDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CB03.4 |
AOC1
|
Diffuse large B-cell lymphomasDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A81 |
LMNA
|
Adenocarcinoma of pancreasDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C10.0 |
LMNA
|
Other specified malignant neoplasms of bronchus or lungDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25.Y |
AOC1
|
Superficial ovarian endometriosisDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GA10.B4 |
LMNA
|
Chronic rhinosinusitisDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CA0A |
AOC1
|
Malignant neoplasms of thyroid gland, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D10.Z |
AOC1
|
Adenocarcinoma of bronchus or lungDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25.0 |
AOC1
|
Malignant neoplasms of corpus uteri, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C76.Z |
AOC1
|
Chikungunya virus diseaseDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D40 |
LMNA
|
Malignant neoplasms of thymusDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C27 |
AOC1
|
Invasive carcinoma of breastDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C61 |
AOC1
|
Malignant neoplasms of adrenal glandDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D11 |
AOC1
|

