Collective Molecular Activities of the Plant: Onychium Lucidum
Overview of Ingredients
3 All known Ingredients in Total
Unique ingredients have been isolated from this plant.Plant-Ingredients Associations were manually curated from publications or collected from other databases.
1 Ingredients with Acceptable Bioavailablity
Unique ingredients exhibit acceptable human oral bioavailablity, according to the criteria of SwissADME [PMID: 28256516] and HobPre [PMID: 34991690]. The criteria details:SwissADME: six descriptors are used by SwissADME to evaluate the oral bioavailability of a natural product:
☑ LIPO(Lipophility): -0.7 < XLOGP3 < +5.0
☑ SIZE: 150g/mol < MW < 500g/mol
☑ POLAR(Polarity): 20Ų < TPSA < 130Ų
☑ INSOLU(Insolubility): -6 < Log S (ESOL) < 0
☑ INSATU(Insaturation): 0.25 < Fraction Csp3 < 1
☑ FLEX(Flexibility): 0 < Num. rotatable bonds < 9
If 6 descriptors of a natural plant satisfy the above rules, it will be labeled high HOB.
HobPre: A natural plant ingredient with HobPre score >0.5 is labeled high human oral availability (HOB)
2 Ingredients with experimental-derived Activity
Unique ingredients have activity data available.Ingredient Structrual Cards
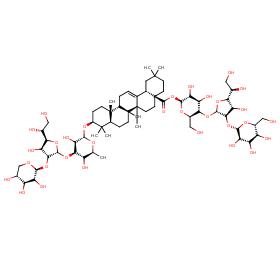
Ingredient ID: NPC59727
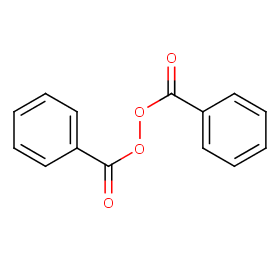
Ingredient ID: NPC261181
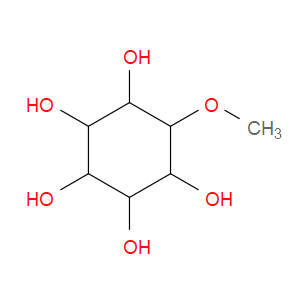
Ingredient ID: NPC207656
Classification of Human Proteins Collectively Targeted by the Plant
Detailed Information of Target Proteins
| Target Type | Protein Class | Gene ID | Protein Name | Uniprot ID | Target ChEMBL ID |
|---|---|---|---|---|---|
| Therapeutic Target | Structural protein | LMNA | Prelamin-A/C | P02545 | CHEMBL1293235 |
Clinical trials associated with plant from natural product (NP) & plant level:
| Clinical trials type | Number of clinical trials | |
|---|---|---|
| 97 | ||
| NCT ID | Title | Condition | Form in clinical use | Associated by plant or compound |
|---|---|---|---|---|
| NCT00377000 | A Pilot, Multi-Center, Patient Preference Study Comparing Two Clindamycin/Benzoyl Peroxide Gels. | acne | Benzoyl Peroxide (NPC261181) | |
| NCT04855266 | Iron Sucrose in Patients With Iron Deficiency and POTS | orthostatic hypotension | Sucrose (NPC130683) | |
| NCT01238471 | Oral Propranolol Versus Placebo for Early Stages of Retinopathy of Prematurity: A Randomized and Prospective Study | retinopathy of prematurity | Sucrose (NPC130683) | |
| NCT00877409 | Safety and Efficacy Study of Acnase Creme in the Treatment of Acne Vulgaris I and II | acne | Benzoyl Peroxide (NPC261181) | |
| NCT02990364 | Combination Analgesia for Neonatal Circumcision | pain | Sucrose (NPC130683) | |
| NCT02005666 | To Compare the Efficacy and Safety of Clindamycin Phosphate 1.2% / Benzoyl Peroxide 5% Gel of CHL Versus DUAC® Gel | acne | Benzoyl Peroxide (NPC261181) | |
| NCT01138514 | Clinical Study Between Two Clindamycin 1%/Benzoyl Peroxide 5% Topical Gels | acne | Benzoyl Peroxide (NPC261181) | |
| NCT03257202 | Topical Treatment and Prevalence of P. Acnes | infection | Benzoyl Peroxide (NPC261181) | |
| NCT02595034 | A Study CLBG and Benzoyl Peroxide Gel 1%/5% to BenzaClin® Gel in the Treatment of Acne Vulgaris | acne | Benzoyl Peroxide (NPC261181) | |
| NCT02073448 | Efficacy and Safety Study of GK530G Versus CD0271 0.1% Gel and CD1579 2.5% Gel in the Treatment of Acne Vulgaris. | acne | Benzoyl Peroxide (NPC261181) | |
| NCT01209949 | Assessment of Subjects' Efficacy and Experiences Using Adapalene BPO Gel in the Treatment of Acne Vulgaris | acne | Benzoyl Peroxide (NPC261181) | |
| NCT01016977 | A Phase 4, Single-Blind, Randomized, Study to Compare the Tolerability and Efficacy of 0.1% Tazorac Cream When Used in Combination With Either Duac Gel or Acanya Gel for the Treatment of Facial Acne Vulgaris | acne | Benzoyl Peroxide (NPC261181) | |
| NCT01201954 | Pain Relief of Newborn Preterm Infants During Endotracheal Suctioning | pain | Sucrose (NPC130683) | |
| NCT01880320 | Multi-center, Randomized, Double-blind Efficacy and Safety of CD0271 0.3%/CD1579 2.5 % Topical Gel in Acne Vulgaris | acne | Benzoyl Peroxide (NPC261181) | |
| NCT00891982 | A Clinical Study to Evaluate the Tolerability of a Topical Antibiotic and Retinoid Used in a Combined Regimen With a BPO Wash | acne | Benzoyl Peroxide (NPC261181) | |
| NCT04300010 | Blue Light Therapy of C. Acnes | acne | Benzoyl Peroxide (NPC261181) | |
| NCT03457636 | The Use of Oracea and Epiduo Forte in Severe Acne Patients | acne | Benzoyl Peroxide (NPC261181) | |
| NCT01561547 | Trial of Repeated Analgesia With Kangaroo Care | pain | Sucrose (NPC130683) | |
| NCT00664248 | A Clinical Study Evaluating the Safety and Efficacy of IDP-110 in Patients With Acne Vulgaris | acne | Benzoyl Peroxide (NPC261181) | |
| NCT01788384 | Evaluate Therapeutic Equivalence and Safety of Two Clindamycin Phosphate and Benzoyl Peroxide Gels in Acne Vulgaris | acne | Benzoyl Peroxide (NPC261181) | |
| NCT01106807 | Exploratory Study to Evaluate the Efficacy and Safety of CD07223 Gel in Subjects With Acne | acne | Benzoyl Peroxide (NPC261181) | |
| NCT01504204 | Effect of Samples on Acne Treatment With Epiduo® Gel | acne | Benzoyl Peroxide (NPC261181) | |
| NCT00869492 | Comparison of Nadifloxacin Cream Alone and With Benzoyl Peroxide Solution in the Treatment of Acne | acne | Benzoyl Peroxide (NPC261181) | |
| NCT00913185 | Comparison Study of Topical Acne Regimens | acne | Benzoyl Peroxide (NPC261181) | |
| NCT03745963 | The Influence of Skin-to-skin Contact on Cortical Activity During Painful Procedures on Preterm Infants in the NICU | pain | Sucrose (NPC130683) | |
| NCT00671749 | Combination Therapy With Differin® Gel 0.3% and Duac® (Clindamycin/Benzoyl Peroxide Gel) in Subjects With Acne Vulgaris | acne | Benzoyl Peroxide (NPC261181) | |
| NCT03082001 | Sucrose to Reduce Pain During Initiation of Venipuncture in Extremely Low Birth Weight Babies | pain | Sucrose (NPC130683) | |
| NCT01428466 | Japanese Phase 1 Study of GSK2585823 | acne | Benzoyl Peroxide (NPC261181) | |
| NCT01206348 | Combination Treatment for Moderate to Severe Acne | acne | Benzoyl Peroxide (NPC261181) | |
| NCT01188538 | Anti Propionibacterium(P.) Acnes Activity of Epiduo® Gel Compared to Benzoyl Peroxide (BPO) 2.5% Gel | acne | Benzoyl Peroxide (NPC261181) | |
| NCT00687908 | Adapalene-Benzoy Peroxide (BPO) Gel in the Treatment of Acne Vulgaris as a 6-month Maintenance | acne | Benzoyl Peroxide (NPC261181) | |
| NCT00422240 | Study to Demonstrate the Efficacy and Safety of Adapalene/Benzoyl Peroxide Topical Gel in Subjects With Acne Vulgaris | acne | Benzoyl Peroxide (NPC261181) | |
| NCT01522456 | Split-face Tolerability Comparison Between Adapalene-Benzoyl Peroxide Gel Versus Tretinoin Gel | acne | Benzoyl Peroxide (NPC261181) | |
| NCT03272594 | The Influence of Breastfeeding on Cortical Activity During Procedures | pain | Sucrose (NPC130683) | |
| NCT01769664 | A Study Comparing Clindamycin 1%/Benzoyl Peroxide 5% Topical Gel to Duac® Topical Gel in the Treatment of Acne Vulgaris | acne | Benzoyl Peroxide (NPC261181) | |
| NCT00949104 | Role of Oral Sucrose in Reducing the Pain to Orogastric Tube Insertion in Preterm Neonates | pain | Sucrose (NPC130683) | |
| NCT01448733 | Safety and Tolerability of Acanya Gel in Combination With Atralin Gel for Acne Vulgaris | acne | Benzoyl Peroxide (NPC261181) | |
| NCT00964366 | Study to Determine and Compare the Tolerance and Irritation Potential of Topical Acne Medications | acne | Benzoyl Peroxide (NPC261181) | |
| NCT00787059 | AC6 Gene Transfer for CHF | congestive heart failure | Sucrose (NPC130683) | |
| NCT01138735 | Epiduo Pediatric Acne Study | acne | Benzoyl Peroxide (NPC261181) | |
| NCT01769235 | Study Comparing Clindamycin Phosphate and Benzoyl Peroxide Gel to Acanya® Gel and Both to a Vehicle Control in the Treatment of Acne Vulgaris | acne | Benzoyl Peroxide (NPC261181) | |
| NCT00919191 | Evaluation of Irritation by Two Facial Gels Applied to Opposite Sides of the Face | acne | Benzoyl Peroxide (NPC261181) | |
| NCT02899000 | A Treatment for Severe Inflammatory Acne Subjects | acne | Benzoyl Peroxide (NPC261181) | |
| NCT02515305 | Comparative Safety and Efficacy of Two Treatments in the Treatment of Acne Vulgaris | acne | Benzoyl Peroxide (NPC261181) | |
| NCT02616614 | Double-blind Placebo-controlled Trial of Generic Clindamycin/Benzoyl Peroxide Gel Versus Onexton Gel in Acne Vulgaris | acne | Benzoyl Peroxide (NPC261181) | |
| NCT03034460 | Efficacy and Safety of CD5024 1% in Acne Vulgaris | acne | Benzoyl Peroxide (NPC261181) | |
| NCT01015638 | Compare the Tolerance of Clindamycin 1% /Benzoyl Peroxide (BPO) 5% Gel to Clindamycin 1.2%/ BPO 2.5% Topical Medications | acne | Benzoyl Peroxide (NPC261181) | |
| NCT01706263 | U0289-402: An Open Label, 8 Week Study to Evaluate the Efficacy and Tolerability of MAXCLARITY II in Subjects With Acne | acne | Benzoyl Peroxide (NPC261181) | |
| NCT00663286 | A Clinical Trial Evaluating the Safety and Efficacy of IDP-110 in Patients With Acne Vulgaris | acne | Benzoyl Peroxide (NPC261181) | |
| NCT02174705 | Rotavirus vs. Sucrose for Vaccine Injection Pain | pain | Sucrose (NPC130683) | |
| NCT01422785 | A Study Comparing Combination Clindamycin Phosphate/Tretinoin Gel Alone Versus With Benzoyl Peroxide Foaming Cloths for Facial Acne | acne | Benzoyl Peroxide (NPC261181) | |
| NCT00713609 | Safety and Efficacy Study of Clindamycin/Benzoyl Peroxide/Tazarotene Cream in Subjects With Acne | acne | Benzoyl Peroxide (NPC261181) | |
| NCT00446043 | A Long-term Study to Demonstrate the Efficacy and Safety of Adapalene/Benzoyl Peroxide Topical Gel in Subjects With Acne Vulgaris | acne | Benzoyl Peroxide (NPC261181) | |
| NCT01742637 | Study Comparing Adapalene and Benzoyl Peroxide Gel 0.1%/2.5% to Epiduo® and Both to a Placebo Control in the Treatment of Acne Vulgaris | acne | Benzoyl Peroxide (NPC261181) | |
| NCT02219776 | Decreasing Infection In Arthroscopic Shoulder Surgery | infection | Benzoyl Peroxide (NPC261181) | |
| NCT03122457 | Perimenstrual Acne With Clindamycin Phosphate and Benzoyl Peroxide | actinic keratosis | Benzoyl Peroxide (NPC261181) | |
| NCT01260571 | Clinical Study to Evaluate the Efficacy of Topical Medications Containing Benzoyl Peroxide and Sulfur in the Regression in the Short Time (24 Hours) of Moderate Acne Vulgaris Grade II (Pimples) | acne | Benzoyl Peroxide (NPC261181) | |
| NCT00151541 | A Phase 3 Study to Compare the Safety and Efficacy of 5% Dapsone Topical Gel, (DTG) Twice Daily in Combination With Once Daily Vehicle Control, Adapalene Gel 0.1% or Benzoyl Peroxide Gel 4% | acne | Benzoyl Peroxide (NPC261181) | |
| NCT00757523 | Evaluation of the Effectiveness, Safety, and Tolerability of Duac Akne Gel and Epiduo Gel in the Treatment of Facial Acne Vulgaris | acne | Benzoyl Peroxide (NPC261181) | |
| NCT00952523 | Evaluation of Irritation That Potentially Could be Caused by Two Facial Gels Applied to Opposite Sides of the Face | acne | Benzoyl Peroxide (NPC261181) | |
| NCT00218946 | Nasogastric Tube in Premature Babies - Pain and Treatment | premature birth | Sucrose (NPC130683) | |
| NCT05096312 | Effects of Oral Zinc Gluconate Among Acne Vulgaris Patients | acne | Benzoyl Peroxide (NPC261181) | |
| NCT01014689 | Comparison of Epiduo Associated With Lymecycline Versus Epiduo Vehicle Associated With Lymecycline in Acne Vulgaris | acne | Benzoyl Peroxide (NPC261181) | |
| NCT01796665 | A Study to Compare Clindamycin Phosphate and Benzoyl Peroxide Topical Gel 1.2%/2.5% to Acanya® Topical Gel in the Treatment of Acne Vulgaris | acne | Benzoyl Peroxide (NPC261181) | |
| NCT02465632 | To Study Generic Clindamycin 1%/Benzoyl Peroxide 5% Topical Gel (Glenmark Generics, Ltd) in the Treatment of Acne Vulgaris. | acne | Benzoyl Peroxide (NPC261181) | |
| NCT01132443 | W0261-101: A Phase 1, Single Center, Randomized, Open-Label Study to Evaluate the Bioavailability of Clindamycin From Clindamycin 1%-Benzoyl Peroxide 3% Gel, Topical Gel (Clindamycin 1%- Benzoyl Peroxide 5%), and Once Daily Gel (Clindamycin 1%-Benzoyl Peroxide 5%) in Subjects With Acne | acne | Benzoyl Peroxide (NPC261181) | |
| NCT02073461 | Efficacy and Safety Study of 2 Different Concentrations of CD1579 Gels Versus Vehicle in the Treatment of Acne Vulgaris. | acne | Benzoyl Peroxide (NPC261181) | |
| NCT00441415 | Efficacy and Safety of Fixed Combination Adapalene 0.1% / Benzoyl Peroxide 2.5% Gel in the Treatment of Acne Vulgaris | acne | Benzoyl Peroxide (NPC261181) | |
| NCT00807014 | Evaluation of Quality of Life, Efficacy, and Tolerance of Duac® Gel Compared to Differin® Gel in the Treatment of Acne | acne | Benzoyl Peroxide (NPC261181) | |
| NCT00837213 | Comparative Efficacy and Safety of Benzoyl Peroxide Used in Combination With Clindamycin vs. Benzoyl Peroxide Used in Combination With Clindamycin and Doxycycline in the Treatment of Moderate Acne | acne | Benzoyl Peroxide (NPC261181) | |
| NCT00519207 | Evaluation of Topical Liposomal Lidocaine and Oral Sucrose for Treatment of Pain in Newborns Undergoing Venipuncture | pain | Sucrose (NPC130683) | |
| NCT01190995 | Role of Repeated Painful Procedures in Preterm Neonates on Short Term Neurobehavioural Outcome | pain | Sucrose (NPC130683) | |
| NCT00907257 | A Study of Different Use Regimens Using Two Acne Treatments | acne | Benzoyl Peroxide (NPC261181) | |
| NCT01231334 | A Study Comparing Aczone® Plus Differin® Versus Duac® Plus Differin® in Patients With Severe Facial Acne | acne | Benzoyl Peroxide (NPC261181) | |
| NCT01527123 | A Study to Evaluate the Pharmacokinetics of Benzoic Acid and Hippuric Acid After Topical Administration of GSK2585823 in Japanese Subjects With Acne Vulgaris | acne | Benzoyl Peroxide (NPC261181) | |
| NCT02086864 | A Placebo Controlled Study of Homeopathic Treatment of Children and Youth With ADHD | attention deficit hyperactivity disorder | Sucrose (NPC130683) | |
| NCT03718507 | Study on the Effects of Different Premedication for LISA on Stress and Cerebral Tissue Oxygenation in Preterm Infants | acute respiratory distress syndrome | Sucrose (NPC130683) | |
| NCT02524665 | 8 Week Study to Evaluate and Compare the Efficacy and Tolerability of MAXCLARITY II and MURAD To Treat Acne | acne | Benzoyl Peroxide (NPC261181) | |
| NCT02525549 | Comparative Safety and Bioequivalence of Two Treatments in the Treatment of Acne Vulgaris | acne | Benzoyl Peroxide (NPC261181) | |
| NCT01706250 | U0289-401: Eight Week, Split-face, Study to Determine and Compare the Efficacy and Tolerability of MAXCLARITY™ II to PROACTIV™ | acne | Benzoyl Peroxide (NPC261181) | |
| NCT00482560 | Analgesic Effect of Breast Feeding vs Sucrose in Neonatal Screening | pain | Sucrose (NPC130683) | |
| NCT02249104 | A Treatment Regimen for Student Athletes With Mild to Moderate Acne Vulgaris | acne | Benzoyl Peroxide (NPC261181) | |
| NCT00926367 | Two-week Study to Compare the Tolerance and Irritation Potential of Two Combination Topical Gel Acne Medications | acne | Benzoyl Peroxide (NPC261181) | |
| NCT01400932 | Study STF115288, a Clinical Confirmation Study of GI148512 in the Treatment of Acne Vulgaris in Japanese Subjects | acne | Benzoyl Peroxide (NPC261181) | |
| NCT01445301 | Study STF115287, a Clinical Confirmation Study of GSK2585823 in the Treatment of Acne Vulgaris in Japanese Subjects | acne | Benzoyl Peroxide (NPC261181) | |
| NCT01503060 | Effectiveness of Parent-Led Interventions in Reducing Infant Hypersensitivity to Pain | pain | Sucrose (NPC130683) | |
| NCT02578043 | A Study Comparing Clindamycin and Benzoyl Peroxide Gel 1.2%/3.75% to Onexton™ Gel in the Treatment of Acne Vulgaris | acne | Benzoyl Peroxide (NPC261181) | |
| NCT00841776 | Comparative Antimicrobial Efficacy of Two Topical Acne Therapies for the Treatment of Facial Acne | acne | Benzoyl Peroxide (NPC261181) | |
| NCT03563365 | The Functional and Emotional Benefits of Replenix Power of Three With Resveratrol | acne | Benzoyl Peroxide (NPC261181) | |
| NCT04548349 | Profiling the Skin Microbiome in Response to Altreno in Acne Patients | acne | Benzoyl Peroxide (NPC261181) | |
| NCT02651220 | Clinical End Point Study of Generic Adapalene and Benzoyl Peroxide Gel Versus Epiduo® Forte Gel in Treatment of Acne Vulgaris | acne | Benzoyl Peroxide (NPC261181) | |
| NCT02589405 | Benzac 5% Gel in Combination With Cosmetic Products in Acne Vulgaris | acne | Benzoyl Peroxide (NPC261181) | |
| NCT01910064 | A Long Term Study of GK530G in Subjects With Acne Vulgaris | acne | Benzoyl Peroxide (NPC261181) | |
| NCT03393494 | Bioequivalence Study of Two Treatments in the Treatment of Acne Vulgaris on the Face | acne | Benzoyl Peroxide (NPC261181) | |
| NCT00776919 | Trial of Clindamycin / Benzoyl Peroxide Gel in Subjects With Acne | acne | Benzoyl Peroxide (NPC261181) | |
| NCT00887484 | A Comparative Study of the Tolerability of Two Combination Therapies for the Treatment of Acne | acne | Benzoyl Peroxide (NPC261181) | |
| NCT00421993 | A Study to Demonstrate the Efficacy and Safety of Adapalene/Benzoyl Peroxide Topical Gel in Subjects With Acne Vulgaris | acne | Benzoyl Peroxide (NPC261181) |
❱❱❱ Associated Human Diseases and Detailed Association Evidence
How do we define the Plant-Targeted Human Disease Association?
Associated human diseases of an individual plant are summurized based on FOUR types of association evidence, these include:
❶ Association by Therapeutic Target: Bioactive protein targets of the plant were defined in "Molecular Targets" section, target-disease associations collected from TTD database were subsequently used to build the associations between the plant and its targeted human diseases.
❷ Association by Disease Gene Reversion: Plant and a specific disease will be associated when >= 1 plant target gene overlaped with disease's DEGs.
❸ Association by Clinical Trials of Plant: Plant and a specific disease will be associated when >= 1 clinical trial (the plant is the intervetion) can be matched in ClinicalTrials.gov database.
❹ Association by Clinical Trials of Plant Ingredients: Plant and a specific disease will be associated when >= 1 clinical trial (the plant ingredient is the intervetion) can be matched in ClinicalTrials.gov database.
Associated Disease of the Plant |
Association Type & Detailed Evidence |
|---|---|
Acne vulgarisDisease Category: 14.Diseases of the skinDisease ICD-11 Code: ED80 |
NCT02005666,NCT02249104,NCT00964366,NCT04548349,NCT01910064,NCT01769235,NCT01209949,NCT01231334,NCT00952523,NCT01788384,NCT02073461,NCT00926367,NCT02073448,NCT01706263,NCT00776919,NCT01260571,NCT01016977,NCT01796665,NCT05096312,NCT03457636,NCT01106807,NCT00446043,NCT00658112,NCT02616614,NCT04300010,NCT01880320,NCT01015638,NCT00807014,NCT00919191,NCT01769664,NCT00663286,NCT00441415,NCT00887484,NCT02899000,NCT00913185,NCT03393494,NCT02525549,NCT00837213,NCT02595034,NCT00664248,NCT01422785,NCT01445301,NCT01706250,NCT01014689,NCT01522456,NCT02465632,NCT02515305,NCT02589405,NCT00151541,NCT01448733,NCT03034460,NCT02651220,NCT03563365,NCT00421993,NCT01132443,NCT01138514,NCT01428466,NCT00841776,NCT00869492,NCT00687908,NCT00757523,NCT01527123,NCT00671749,NCT00422240,NCT01206348,NCT01504204,NCT00377000,NCT00713609,NCT02524665,NCT01742637,NCT01138735,NCT02578043,NCT00907257,NCT01188538,NCT00877409,NCT01400932,NCT00891982
|
Herpes simplex infectionDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1F00 |
NCT02219776,NCT03257202
|
Pain, unspecifiedDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: MG3Z |
NCT02174705,NCT01201954,NCT01190995,NCT03745963,NCT00482560,NCT03272594,NCT00519207,NCT03082001,NCT02990364,NCT01503060,NCT00949104,NCT01561547
|
Acute respiratory distress syndromeDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CB00 |
NCT03718507
|
Attention deficit hyperactivity disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6A05 |
NCT02086864
|
Actinic keratosisDisease Category: X.Extension CodesDisease ICD-11 Code: XH36H6 |
NCT03122457
|
Preterm newborn, unspecifiedDisease Category: 19.Certain conditions originating in the perinatal periodDisease ICD-11 Code: KA21.4Z |
NCT00218946
|
Orthostatic hypotensionDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA21 |
NCT04855266
|
Retinopathy of prematurityDisease Category: 09.Diseases of the visual systemDisease ICD-11 Code: 9B71.3 |
NCT01238471
|
Congestive heart failureDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BD10 |
NCT00787059
|
Malignant neoplasms of biliary tract, distal bile ductDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C15 |
LMNA
|
Diffuse large B-cell lymphomasDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A81 |
LMNA
|
Adenocarcinoma of pancreasDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C10.0 |
LMNA
|
Mesothelioma of pleuraDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C26.0 |
LMNA
|
Superficial ovarian endometriosisDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GA10.B4 |
LMNA
|
Chikungunya virus diseaseDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D40 |
LMNA
|
Glioblastoma of brainDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A00.00 |
LMNA
|

