Collective Molecular Activities of the Plant: Garcinia Nigrolineata
Plant ID: NPO16861
Plant Latin Name: Garcinia Nigrolineata
Taxonomy Genus: Garcinia
Taxonomy Family: Clusiaceae
Plant External Links:
NCBI TaxonomyDB:
180110
Plant-of-the-World-Online:
n.a.
Overview of Ingredients
36 All known Ingredients in Total
Unique ingredients have been isolated from this plant.Plant-Ingredients Associations were manually curated from publications or collected from other databases.
34 Ingredients with Acceptable Bioavailablity
Unique ingredients exhibit acceptable human oral bioavailablity, according to the criteria of SwissADME [PMID: 28256516] and HobPre [PMID: 34991690]. The criteria details:SwissADME: six descriptors are used by SwissADME to evaluate the oral bioavailability of a natural product:
☑ LIPO(Lipophility): -0.7 < XLOGP3 < +5.0
☑ SIZE: 150g/mol < MW < 500g/mol
☑ POLAR(Polarity): 20Ų < TPSA < 130Ų
☑ INSOLU(Insolubility): -6 < Log S (ESOL) < 0
☑ INSATU(Insaturation): 0.25 < Fraction Csp3 < 1
☑ FLEX(Flexibility): 0 < Num. rotatable bonds < 9
If 6 descriptors of a natural plant satisfy the above rules, it will be labeled high HOB.
HobPre: A natural plant ingredient with HobPre score >0.5 is labeled high human oral availability (HOB)
7 Ingredients with experimental-derived Activity
Unique ingredients have activity data available.Ingredient Structrual Cards
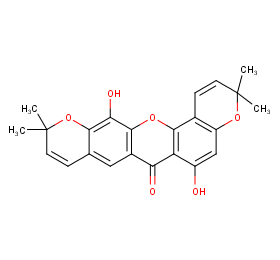
Ingredient ID: NPC90246
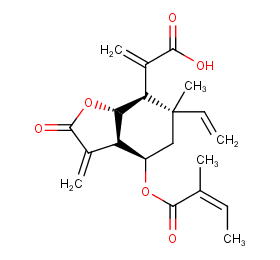
Ingredient ID: NPC87802
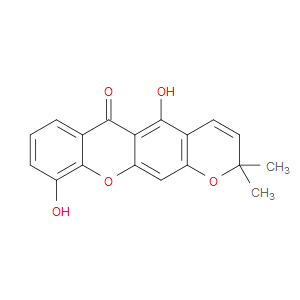
Ingredient ID: NPC6633
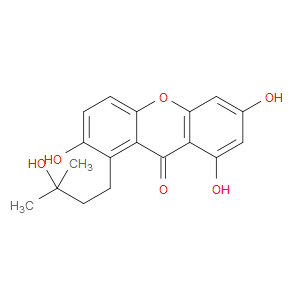
Ingredient ID: NPC66054
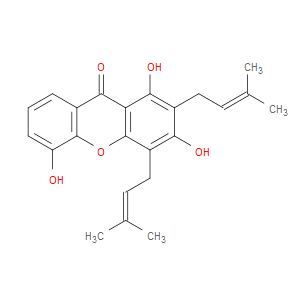
Ingredient ID: NPC33051
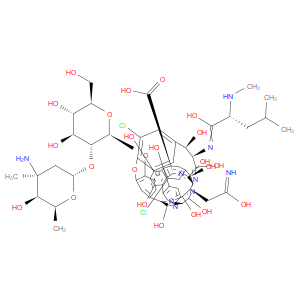
Ingredient ID: NPC314743
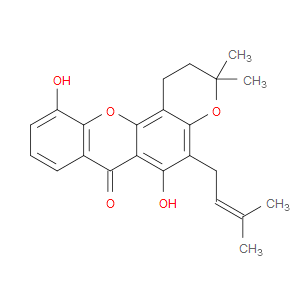
Ingredient ID: NPC3015
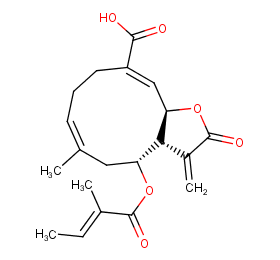
Ingredient ID: NPC288294
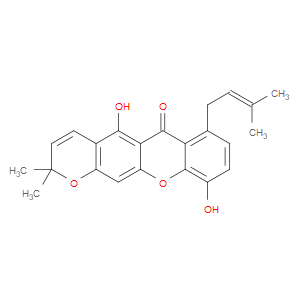
Ingredient ID: NPC276333
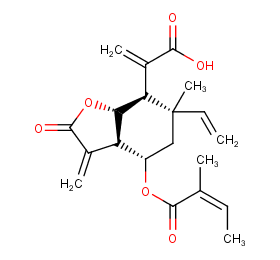
Ingredient ID: NPC275631

Ingredient ID: NPC271538
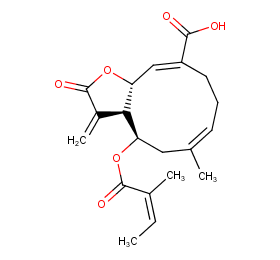
Ingredient ID: NPC268146
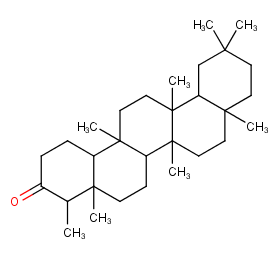
Ingredient ID: NPC266020
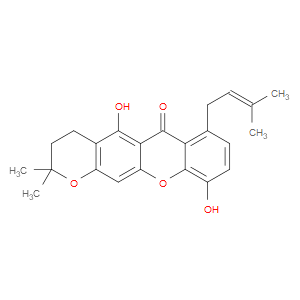
Ingredient ID: NPC250112
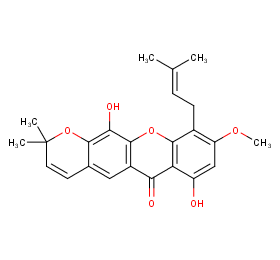
Ingredient ID: NPC242414
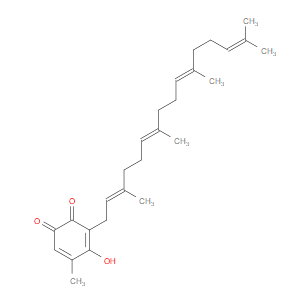
Ingredient ID: NPC231325

Ingredient ID: NPC223325
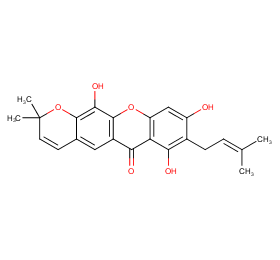
Ingredient ID: NPC218313
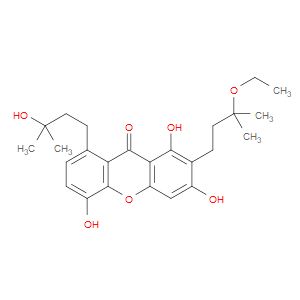
Ingredient ID: NPC218149
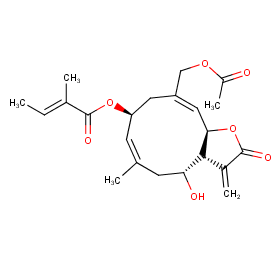
Ingredient ID: NPC209066
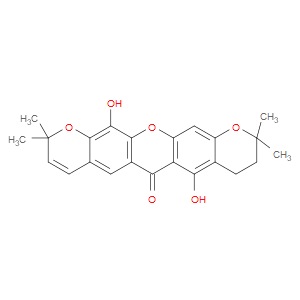
Ingredient ID: NPC207234
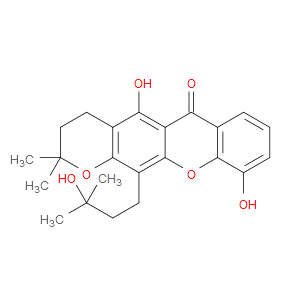
Ingredient ID: NPC204472
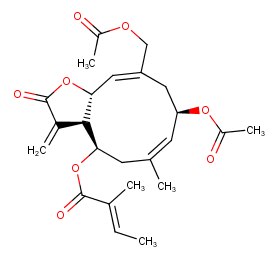
Ingredient ID: NPC204078
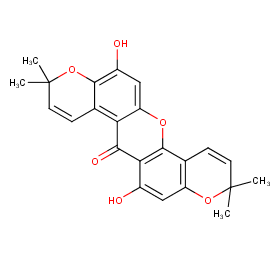
Ingredient ID: NPC195235
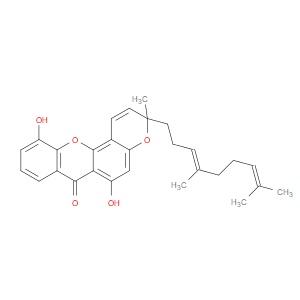
Ingredient ID: NPC185717
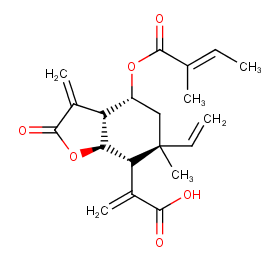
Ingredient ID: NPC171782
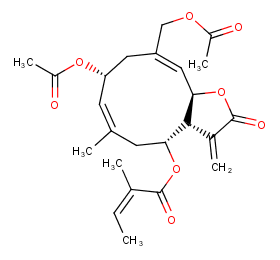
Ingredient ID: NPC164683
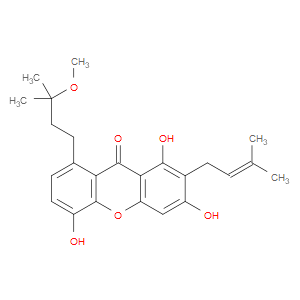
Ingredient ID: NPC159682
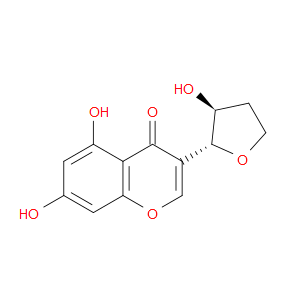
Ingredient ID: NPC153458
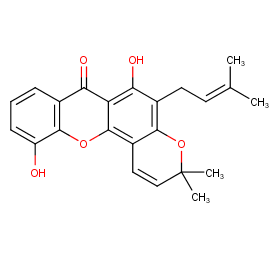
Ingredient ID: NPC14709
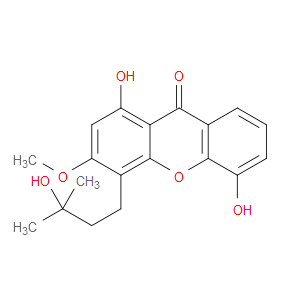
Ingredient ID: NPC145600
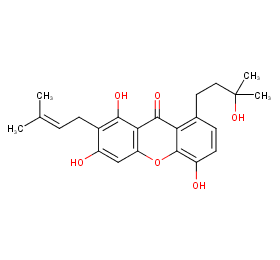
Ingredient ID: NPC136674
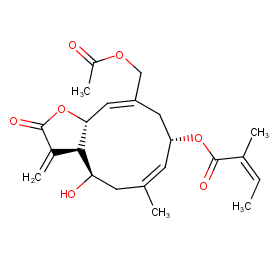
Ingredient ID: NPC129971
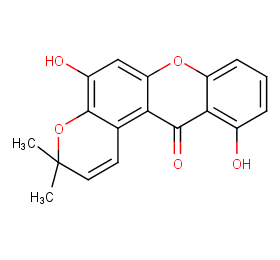
Ingredient ID: NPC129614

Ingredient ID: NPC125773

Ingredient ID: NPC120105
Classification of Human Proteins Collectively Targeted by the Plant
Detailed Information of Target Proteins
| Target Type | Protein Class | Gene ID | Protein Name | Uniprot ID | Target ChEMBL ID |
|---|---|---|---|---|---|
| Therapeutic Target | Small molecule receptor (family A GPCR) | ADRA2A | Alpha-2a adrenergic receptor | P08913 | CHEMBL1867 |
Clinical trials associated with plant from natural product (NP) & plant level:
| Clinical trials type | Number of clinical trials | |
|---|---|---|
| 126 | ||
| NCT ID | Title | Condition | Form in clinical use | Associated by plant or compound |
|---|---|---|---|---|
| NCT04898348 | A Study to Investigate Efficacy and Safety of KBL697 in Patients With Mild to Moderate Active Ulcerative Colitis | ulcerative colitis | Vancomycin (NPC314743) | |
| NCT03827447 | Vancomycin for the Treatment of NAAT(+)/Toxin(-) C. Difficile | clostridium difficile infection | Vancomycin (NPC314743) | |
| NCT02600611 | Phase 3 Study to Evaluate Safety and Efficacy of Iclaprim Versus Vancomycin for ABSSSI: REVIVE-1 | infection | Vancomycin (NPC314743) | |
| NCT02765256 | Fundamental Modification of the Gut Microbiota in the Treatment of Refractory Crohn's Disease | Crohn's disease | Vancomycin (NPC314743) | |
| NCT01199783 | Application of Daptomycin in MRSA Infected Diabetic Foot in Comparison to Vancomycin Treatment | diabetic foot | Vancomycin (NPC314743) | |
| NCT01570192 | Clinical Trials to Reduce the Risk of Antimicrobial Resistance | bacterial pneumonia | Vancomycin (NPC314743) | |
| NCT02730962 | Interventional Bioremediation of Microbiota in Metabolic Syndrome | prediabetes syndrome | Vancomycin (NPC314743) | |
| NCT02504554 | Beneficial Bacteria Treatment for Autism | autism spectrum disorder | Vancomycin (NPC314743) | |
| NCT00442832 | TD-1792 in Gram-positive Complicated Skin and Skin Structure Infection | staphylococcal skin infections | Vancomycin (NPC314743) | |
| NCT02865616 | MET-2 Clinical Study for Recurrent Clostridium Difficile Infection (CDI) | clostridium difficile infection | Vancomycin (NPC314743) | |
| NCT00423657 | Comparative Study of Ceftaroline vs. Vancomycin Plus Aztreonam in Adult Subjects With Complicated Skin Infections | bacterial disease | Vancomycin (NPC314743) | |
| NCT01983683 | Efficacy and Safety of Cadazolid Versus Vancomycin in Subjects With Clostridium Difficile-associated Diarrhea | clostridium difficile infection | Vancomycin (NPC314743) | |
| NCT04968951 | Refined Fecal Microbiota Transplantation (FMT) for Ulcerative Colitis (UC) | ulcerative colitis | Vancomycin (NPC314743) | |
| NCT01598311 | A Study of CB-183,315 in Participants With Clostridium Difficile Associated Diarrhea (MK-4261-006) | clostridium difficile infection | Vancomycin (NPC314743) | |
| NCT00107978 | Comparison of Telavancin and Vancomycin for Complicated Skin and Skin Structure Infections With a Focus on Methicillin-resistant Staphylococcus Aureus | staphylococcal skin infections | Vancomycin (NPC314743) | |
| NCT02432807 | Safety and Efficacy of Vancomycin Ophthalmic Ointment in Patients With Moderate to Severe Bacterial Conjunctivitis | bacterial conjunctivitis | Vancomycin (NPC314743) | |
| NCT03817125 | Melanoma Checkpoint and Gut Microbiome Alteration With Microbiome Intervention | metastatic melanoma | Vancomycin (NPC314743) | |
| NCT02202135 | Evaluation of Ceftaroline Fosamil vs Vancomycin Plus Aztreonam in the Treatment of Patients With Skin Infections. | infection | Vancomycin (NPC314743) | |
| NCT02179658 | A Study to Compare Safety and Efficacy of OPT-80(Fidaxomicin) With Vancomycin in Subjects With Clostridium Difficile-associated Diarrhea (CDAD) | commensal Clostridium infectious disease | Vancomycin (NPC314743) | |
| NCT03105479 | Comparison of Cadazolid Versus Vancomycin in Children With Clostridium Difficile-associated Diarrhea (CDAD) | clostridium difficile infection | Vancomycin (NPC314743) | |
| NCT01975662 | Vancomycin Versus Daptomycin for the Treatment of Methicillin Resistant Staphylococcus Aureus (MRSA) Bacteremia | Staphylococcus aureus infection | Vancomycin (NPC314743) | |
| NCT01811732 | Delafloxacin Versus Vancomycin and Aztreonam for the Treatment of Acute Bacterial Skin and Skin Structure Infections | bacterial disease | Vancomycin (NPC314743) | |
| NCT05266807 | Fecal Microbiota Transplantation in Clostridioides Difficile Infection First Episode and First Recurrence | infection | Vancomycin (NPC314743) | |
| NCT01798537 | Simplified Selective Digestive Tract Decontamination for the Prevention of Intensive Care Unit Acquired Infections | bacteriemia;pneumonia | Vancomycin (NPC314743) | |
| NCT03030248 | Vancomycin for C Difficile NAAT+/EIA- Hematology Oncology Patients | clostridium difficile infection | Vancomycin (NPC314743) | |
| NCT04247542 | ACX-362E [Ibezapolstat] for Oral Treatment of Clostridioides Difficile Infection | clostridium difficile infection | Vancomycin (NPC314743) | |
| NCT02790996 | Neonatal Vancomycin Trial | Neonatal sepsis | Vancomycin (NPC314743) | |
| NCT02426918 | Study of Debio 1450 for Bacterial Skin Infections | bacterial disease | Vancomycin (NPC314743) | |
| NCT04775953 | DOTS: Dalbavancin as an Option for Treatment of Staphylococcus Aureus Bacteremia | bacteriemia | Vancomycin (NPC314743) | |
| NCT01987895 | Efficacy and Safety of Cadazolid Versus Vancomycin in Subjects With Clostridium Difficile - Associated Diarrhea | clostridium difficile infection | Vancomycin (NPC314743) | |
| NCT00050401 | Ventilator-Associated Pneumonia/Hospital-Acquired Pneumonia Requiring Mechanical Ventilatory Support | pneumonia | Vancomycin (NPC314743) | |
| NCT00037050 | Antibiotic Treatment for Infections of Short Term In-dwelling Vascular Catheters Due to Gram Positive Bacteria | bacteriemia;gram-positive bacterial infections | Vancomycin (NPC314743) | |
| NCT01764750 | Dose Escalation Trial of Intrasite Vancomycin Pharmacokinetics | infection | Vancomycin (NPC314743) | |
| NCT02227446 | Local Antibiotic Therapy to Reduce Infection After Operative Treatment of Fractures at High Risk of Infection: A Multicenter Randomized, Controlled Trial (VANCO) | infection | Vancomycin (NPC314743) | |
| NCT05370885 | VE202 in Patients With Mild-to-Moderate Ulcerative Colitis | ulcerative colitis | Vancomycin (NPC314743) | |
| NCT01515020 | Daptomycin Versus Vancomycin in the Treatment of Nosocomial or Healthcare-associated MRSA Bacteremia | nosocomial infection | Vancomycin (NPC314743) | |
| NCT02208063 | A Phase 3 Telavancin Staphylococcus Aureus (S. Aureus) Bacteremia Trial | bacteriemia | Vancomycin (NPC314743) | |
| NCT00428844 | Study of Daptomycin in Subjects Undergoing Surgery for Osteomyelitis Associated With an Infected Prosthetic Caused by Staphylococci | osteomyelitis | Vancomycin (NPC314743) | |
| NCT01283581 | A Study to Assess Objective Endpoint Measurements of Response in Bacterial Skin Infections | bacterial disease | Vancomycin (NPC314743) | |
| NCT01184872 | Study to Compare Efficacy and Safety of Daptomycin in Elderly Patients With Complicated Skin and Soft Tissue Infections | infection | Vancomycin (NPC314743) | |
| NCT01232595 | Safety and Efficacy of Multiple Daily Dosing of Oral LFF571 in Patients With Moderate Clostridium Difficile Infections | clostridium difficile infection | Vancomycin (NPC314743) | |
| NCT04017468 | Suprafascial Vancomycin Powder for Prevention of Surgical Site Infections After Instrumented Posterior Spinal Fusion | infection | Vancomycin (NPC314743) | |
| NCT00543608 | Clinical Efficacy of Intravenous Iclaprim Versus Vancomycin in the Treatment of Hospital-Acquired, Ventilator-Associated, or Health-Care-Associated Pneumonia | ventilator-associated pneumonia | Vancomycin (NPC314743) | |
| NCT02276482 | Study of Tedizolid Phosphate in Adolescents With Complicated Skin and Soft Tissue Infection (cSSTI) (MK-1986-012) | skin disease | Vancomycin (NPC314743) | |
| NCT00210899 | Ceftobiprole in the Treatment of Resistant Staphylococcus Aureus Skin and Skin Structure Infections | staphylococcal skin infections;skin disease | Vancomycin (NPC314743) | |
| NCT00091819 | Comparison of Telavancin and Vancomycin for Complicated Skin and Skin Structure Infections With a Focus on Methicillin-resistant Staphylococcus Aureus | staphylococcal skin infections | Vancomycin (NPC314743) | |
| NCT01339091 | Efficacy and Safety of Dalbavancin for the Treatment of Acute Bacterial Skin and Skin Structure Infections | abscess;cellulitis;infection | Vancomycin (NPC314743) | |
| NCT02663596 | Safety and TDM of Continuous Infusion Vancomycin Through Continuous Renal Replacement Therapy Solution | kidney failure | Vancomycin (NPC314743) | |
| NCT00228410 | Study Comparing Tigecycline and Vancomycin With Aztreonam in Complicated Skin and Skin Structure Infections | skin disease | Vancomycin (NPC314743) | |
| NCT02344511 | Dalbavancin vs Comparator in Pediatric Subjects With Acute Hematogenous Osteomyelitis | osteomyelitis | Vancomycin (NPC314743) | |
| NCT00314951 | Fidaxomicin Versus Vancomycin for the Treatment of Clostridium Difficile-Associated Diarrhea (CDAD) (MK-5119-018) | commensal Clostridium infectious disease;Diarrhea | Vancomycin (NPC314743) | |
| NCT02774382 | Rectal Bacteriotherapy, Fecal Microbiota Transplantation or Oral Vancomycin Treatment of Recurrent Clostridium Difficile Infections | commensal Clostridium infectious disease | Vancomycin (NPC314743) | |
| NCT02037295 | Investigation of the Gut Microbiota in Regulating Nutrient Absorption in Humans | obesity | Vancomycin (NPC314743) | |
| NCT02857582 | Transplantation of Cultured Gut Microflora to Repeat Antibiotic-induced Diarrhea Due to Clostridium Difficile | commensal Clostridium infectious disease | Vancomycin (NPC314743) | |
| NCT03137173 | Ceftobiprole in the Treatment of Patients With Acute Bacterial Skin and Skin Structure Infections | infection | Vancomycin (NPC314743) | |
| NCT01400867 | Safety and Efficacy Study of Ceftaroline Versus a Comparator in Pediatric Subjects With Complicated Skin Infections | infection | Vancomycin (NPC314743) | |
| NCT01984684 | Delafloxacin vs Vancomycin and Aztreonam for the Treatment of Acute Bacterial Skin and Skin Structure Infections | infection;bacterial disease | Vancomycin (NPC314743) | |
| NCT03685747 | Vancomycin Pharmacokinetics in Patients on Peritoneal Dialysis | peritonitis | Vancomycin (NPC314743) | |
| NCT00150852 | Prevention of Gastrostomy-Related Wound Infection by Vancomycin in Carriers of Methicillin-Resistant Staphylococcus Aureus. | infection | Vancomycin (NPC314743) | |
| NCT02686645 | Fecal Microbiota Therapy for Recurrent Clostridium Difficile Infection | clostridium difficile infection | Vancomycin (NPC314743) | |
| NCT01925066 | Aerosolized Vancomycin in Methicillin-Resistant Staphylococcus Aureus Pneumonia Under Mechanical Ventilation | staphylococcal pneumonia;Staphylococcus aureus infection | Vancomycin (NPC314743) | |
| NCT02301221 | Effect of Fecal Microbiota Transplantation in Slow Transit Constipation | Constipation | Vancomycin (NPC314743) | |
| NCT00384527 | Study of Nitazoxanide in the Treatment of Clostridium Difficile-associated Disease | commensal Clostridium infectious disease | Vancomycin (NPC314743) | |
| NCT01922011 | Safety and Efficacy Study of Daptomycin Compared to Active Comparator in Pediatric Participants With Acute Hematogenous Osteomyelitis (AHO) (MK-3009-006) | osteomyelitis | Vancomycin (NPC314743) | |
| NCT00003805 | Prevention of Infection in Patients With Hematologic Cancer and Persistent Fever Caused by a Low White Blood Cell Count | infection | Vancomycin (NPC314743) | |
| NCT01757236 | Efficacy and Safety Study of Antibiotic Treatment to Treat Hip Prosthetic Joint Infection | infection | Vancomycin (NPC314743) | |
| NCT01191840 | Treatment Algorithm to Reduce the Use of Vancomycin in Adults With Blood Stream Infection | bacteriemia | Vancomycin (NPC314743) | |
| NCT00124020 | Comparison of Telavancin and Vancomycin for Hospital-acquired Pneumonia Due to Methicillin-resistant Staphylococcus Aureus | bacterial pneumonia | Vancomycin (NPC314743) | |
| NCT02618187 | A Study to Evaluate the Safety, Tolerability and Microbiome Dynamics of SER-287 in Subjects With Mild-to-Moderate Ulcerative Colitis | ulcerative colitis | Vancomycin (NPC314743) | |
| NCT00424190 | Comparative Study of Ceftaroline vs. Vancomycin Plus Aztreonam in Adult Subjects With Complicated Skin Infections | bacterial disease | Vancomycin (NPC314743) | |
| NCT01802073 | Primary Sclerosing Cholangitis With Oral Vancomycin by the Study of Its Antimicrobial and Immunomodulating Effects | sclerosing cholangitis | Vancomycin (NPC314743) | |
| NCT00695903 | Phase 2 Study of Safety, Efficacy, and Pharmacokinetics of Higher Doses of Daptomycin and Vancomycin in MRSA Bacteremia | infective endocarditis | Vancomycin (NPC314743) | |
| NCT03595566 | Comparison of Ridinilazole Versus Vancomycin Treatment for Clostridium Difficile Infection | infection | Vancomycin (NPC314743) | |
| NCT00426933 | Phase 2 Safety and Efficacy Study of Daptomycin in Complicated Skin and Skin Structure Infections | infection | Vancomycin (NPC314743) | |
| NCT03546829 | Randomized Pilot Trial of Antimicrobial Therapy and Stereotactic Body Radiation Therapy in Early-stage Non-small Cell Lung Cancer | non-small cell lung carcinoma | Vancomycin (NPC314743) | |
| NCT00430937 | Efficacy and Safety of Daptomycin Versus Vancomycin or Teicoplanin for Treatment of Complicated Skin and Soft Tissue Infections | infection;skin disease | Vancomycin (NPC314743) | |
| NCT03759041 | A Study to Assess Efficacy and Safety of SER-287 in Adults With Active Mild-to-Moderate Ulcerative Colitis | ulcerative colitis | Vancomycin (NPC314743) | |
| NCT00871104 | Fosfomycin Plus Imipenem for Treatment of Infective Endocarditis | infective endocarditis | Vancomycin (NPC314743) | |
| NCT03452189 | The Effect of an Antibiotic on the Production of Uremic Toxins by the Gut Microbiome | chronic kidney disease | Vancomycin (NPC314743) | |
| NCT01597505 | Study of CB-183,315 in Participants With Clostridium Difficile Associated Diarrhea (MK-4261-005) | clostridium difficile infection | Vancomycin (NPC314743) | |
| NCT00062647 | Telavancin for Treatment of Uncomplicated Staphylococcus Aureus Bacteremia | gram-positive bacterial infections | Vancomycin (NPC314743) | |
| NCT05076955 | Evaluating the Healing of Diabetic Foot Ulcers With Compounded Anti-Infective Irrigation Therapy | diabetic foot | Vancomycin (NPC314743) | |
| NCT03476317 | Pilot Study of Fundamental Modification of the Gut Microbiota in the Treatment of Refractory Crohn's Disease | Crohn's disease | Vancomycin (NPC314743) | |
| NCT02374853 | Topical Use of Vancomycin in Reducing Sternal Wound Infection in Cardiac Surgery (SWI Trial) | infection | Vancomycin (NPC314743) | |
| NCT03595553 | Comparison of Ridinilazole Versus Vancomycin Treatment for Clostridium Difficile Infection | infection | Vancomycin (NPC314743) | |
| NCT02148601 | Fecal Microbiota Transplantation by Colonoscopy for Recurrent C. Difficile Infection | commensal Clostridium infectious disease | Vancomycin (NPC314743) | |
| NCT04678154 | Evaluation of a New Strategy for Protocolized Antibiotic Care for Severe Open Fractures: SEXTANT | infection | Vancomycin (NPC314743) | |
| NCT03617445 | Fecal Microbiota Transplantation for C. Difficile Infection in Solid Organ Transplant Recipients | clostridium difficile infection | Vancomycin (NPC314743) | |
| NCT05320068 | Oral Vancomycin vs Placebo in the Prevention of Recurrence of Clostridioides Difficile's Infection | infection | Vancomycin (NPC314743) | |
| NCT02607618 | Phase 3 Study to Evaluate Safety and Efficacy of Iclaprim Versus Vancomycin for ABSSSI: REVIVE-2 | infection | Vancomycin (NPC314743) | |
| NCT03710122 | Vancomycin for Primary Sclerosing Cholangitis | sclerosing cholangitis | Vancomycin (NPC314743) | |
| NCT03462459 | Efficacy of Oral Vancomycin Prophylaxis for Prevention of Recurrent Clostridium Difficile Infection | clostridium difficile infection;Diarrhea | Vancomycin (NPC314743) | |
| NCT03964493 | TNP-2092 to Treat Acute Bacterial Skin and Skin Structure Infection | gram-positive bacterial infections | Vancomycin (NPC314743) | |
| NCT02099240 | Patients Response to Early Switch To Oral:Osteomyelitis Study | osteomyelitis | Vancomycin (NPC314743) | |
| NCT01594827 | Persistent Methicillin Resistant Staphylococcus Aureus Eradication Protocol (PMEP) | cystic fibrosis | Vancomycin (NPC314743) | |
| NCT00077675 | Phase 2 Trial of TD-6424 (Telavancin) Versus Standard Therapy for Complicated Gram Positive Skin and Skin Structure Infections (Gram Positive cSSSI) | infection | Vancomycin (NPC314743) | |
| NCT02466698 | Intestinal Lavage for the Treatment of Severe C. Difficile Infections | commensal Clostridium infectious disease | Vancomycin (NPC314743) | |
| NCT02641236 | Gut Decontamination In Pediatric Allogeneic Hematopoietic | acute graft vs. host disease | Vancomycin (NPC314743) | |
| NCT04816968 | Antibiotics Continuous Infusion at Home | bacterial disease | Vancomycin (NPC314743) | |
| NCT01792804 | Staphylococcus Aureus Bacteremia Antibiotic Treatment Options | Staphylococcus aureus infection | Vancomycin (NPC314743) | |
| NCT05256693 | Prevention of C.Difficile Infections With Oral Vancomycine in Patients Undergoing Allogeneic Hematopoietic Stem Cell Transplant | commensal Clostridium infectious disease | Vancomycin (NPC314743) | |
| NCT01756924 | Safety and Efficacy of CEM-102 With Rifampin Compared to Standard Therapy in Patients With Prosthetic Joint Infections or Spacer Infection | infection | Vancomycin (NPC314743) | |
| NCT01499277 | Evaluation of Ceftaroline Fosamil Versus Vancomycin Plus Aztreonam in the Treatment of Patients With Skin Infections | infection | Vancomycin (NPC314743) | |
| NCT01222702 | Efficacy, Safety and Tolerability of Cadazolid in Subjects With Clostridium Difficile Associated Diarrhea (CDAD) | clostridium difficile infection | Vancomycin (NPC314743) | |
| NCT00770341 | A Study of MK-3009 in Japanese Patients With Skin or Blood Stream Infections Caused by Methicillin-resistant Staphylococcus Aureus (MK-3009-002) | Staphylococcus aureus infection | Vancomycin (NPC314743) | |
| NCT04138706 | Initial Vancomycin Taper for the Prevention of Recurrent Clostridium Difficile Infection | clostridium difficile infection | Vancomycin (NPC314743) | |
| NCT00945152 | Effects of MRSA Bactericidal Gel To Promote Healing and Eliminate MRSA in cSSTI Vancogel(TM) | bacterial disease | Vancomycin (NPC314743) | |
| NCT04983901 | PHASE II SINGLE-CENTER, RANDOMIZED, OPEN-LABEL, PROSPECTIVE, STUDY TO DETERMINE THE IMPACT OF SERIAL PROCALCITONIN | hematopoietic and lymphoid cell neoplasm | Vancomycin (NPC314743) | |
| NCT04025567 | Vancomycin in Patients With Unresectable Fibrolamellar Hepatocellular Carcinoma (FLC) Oral | hepatocellular carcinoma | Vancomycin (NPC314743) | |
| NCT04948814 | Application of Fecal Microbiota Transplantation in Children With ASD | autism spectrum disorder | Vancomycin (NPC314743) | |
| NCT04042077 | Delafloxacin IV and OS Administration Compared to Best Available Therapy in Patients With Surgical Site Infections | infection | Vancomycin (NPC314743) | |
| NCT01252732 | Oritavancin Versus IV Vancomycin for the Treatment of Patients With Acute Bacterial Skin and Skin Structure Infection | abscess;cellulitis;infection;inflammation | Vancomycin (NPC314743) | |
| NCT01226992 | Oral Vancomycin Followed by Fecal Transplant Versus Tapering Oral Vancomycin | clostridium difficile infection | Vancomycin (NPC314743) | |
| NCT02743234 | Fecal Microbiota Transplantation for Relapsing Clostridium Difficile Infection | clostridium difficile infection | Vancomycin (NPC314743) | |
| NCT03506347 | Regional Prophylactic Vancomycin With Restricted Tourniquet Time in Primary Total Knee Replacement | Arthropathy | Vancomycin (NPC314743) | |
| NCT02092935 | A Study of SMT19969 Compared With Vancomycin for the Treatment of Clostridium Difficile-Associated Diarrhoea (CDAD) | clostridium difficile infection | Vancomycin (NPC314743) | |
| NCT03796650 | Fecal Transplantation for Primary Clostridium Difficile Infection | clostridium difficile infection | Vancomycin (NPC314743) | |
| NCT00269399 | A Trial to Compare Xifaxan to Vancomycin for the Treatment of Clostridium Difficile-Associated Diarrhea (CDAD) | commensal Clostridium infectious disease;Diarrhea | Vancomycin (NPC314743) | |
| NCT00772447 | China Registration Study in Patients With Skin Infections | skin disease | Vancomycin (NPC314743) | |
| NCT02801656 | Fecal Microbiota Transplantation for Primary Clostridium Difficile Diarrhea | commensal Clostridium infectious disease | Vancomycin (NPC314743) | |
| NCT04075526 | Vancomycin Powder and Dilute Povidone Iodine Lavage for Infection Prophylaxis in High Risk Total Joint Arthroplasty | osteoarthritis | Vancomycin (NPC314743) | |
| NCT04802837 | Safety, Tolerability and the Pharmacokinetics of Ridinilazole in Adolescent Subjects | infection | Vancomycin (NPC314743) | |
| NCT03673956 | Topical Antibiotics in Chronic Rhinosinusitis | sinusitis | Vancomycin (NPC314743) | |
| NCT01085760 | A Pilot Study of Vancomycin or Metronidazole in Patients With Primary Sclerosing Cholangitis | sclerosing cholangitis | Vancomycin (NPC314743) | |
| NCT00107952 | Comparison of Telavancin and Vancomycin for Hospital-acquired Pneumonia Due to Methicillin-resistant Staphylococcus Aureus | bacterial pneumonia | Vancomycin (NPC314743) | |
| NCT00108433 | Linezolid in the Treatment of Hemodialysis Patients With Catheter-Related Gram-Positive Bloodstream Infections | bacteriemia;gram-positive bacterial infections | Vancomycin (NPC314743) |
❱❱❱ Associated Human Diseases and Detailed Association Evidence
How do we define the Plant-Targeted Human Disease Association?
Associated human diseases of an individual plant are summurized based on FOUR types of association evidence, these include:
❶ Association by Therapeutic Target: Bioactive protein targets of the plant were defined in "Molecular Targets" section, target-disease associations collected from TTD database were subsequently used to build the associations between the plant and its targeted human diseases.
❷ Association by Disease Gene Reversion: Plant and a specific disease will be associated when >= 1 plant target gene overlaped with disease's DEGs.
❸ Association by Clinical Trials of Plant: Plant and a specific disease will be associated when >= 1 clinical trial (the plant is the intervetion) can be matched in ClinicalTrials.gov database.
❹ Association by Clinical Trials of Plant Ingredients: Plant and a specific disease will be associated when >= 1 clinical trial (the plant ingredient is the intervetion) can be matched in ClinicalTrials.gov database.
Associated Disease of the Plant |
Association Type & Detailed Evidence |
|---|---|
Bacterial infectionDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1A00-1C4Z |
NCT02426918,NCT04816968,NCT01984684,NCT00945152,NCT01811732,NCT00423657,NCT00424190,NCT01283581
|
BacteraemiaDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: MA15.0 |
NCT01798537,NCT04775953,NCT02208063,NCT01191840,NCT00108433,NCT00037050
|
Ulcerative colitisDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DD71 |
NCT05370885,NCT04898348,NCT04968951,NCT03759041,NCT02618187
|
Skin diseaseDisease Category: 14.Diseases of the skinDisease ICD-11 Code: EA00-EM0Z |
NCT00228410,NCT00430937,NCT02276482,NCT00772447,NCT00210899
|
Osteomyelitis or osteitisDisease Category: 15.Diseases of the musculoskeletal system or connective tissueDisease ICD-11 Code: FB84 |
NCT01922011,NCT00428844,NCT02344511,NCT02099240
|
Staphylococcal/streptococcal diseaseDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1B5Y |
NCT00091819,NCT00210899,NCT00107978,NCT00442832
|
Gram-positive bacterial infectionDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1B74-1F40 |
NCT00108433,NCT00062647,NCT00037050,NCT03964493
|
Other specified pyogenic bacterial infection of skin and subcutaneous tissueDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1B7Y |
NCT01925066,NCT00770341,NCT01792804,NCT01975662
|
DiarrhoeaDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: ME05.1 |
NCT03462459,NCT00314951,NCT00269399
|
Primary sclerosing cholangitisDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DB96.2 |
NCT01802073,NCT01085760,NCT03710122
|
Bacterial pneumoniaDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CA40.0 |
NCT01570192,NCT00107952,NCT00124020
|
Clostridium difficile enterocolitisDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1A04 |
NCT05256693,NCT02092935,NCT01983683,NCT03617445,NCT03462459,NCT01222702,NCT03827447,NCT02466698,NCT02148601,NCT01597505,NCT03796650,NCT00314951,NCT02179658,NCT03105479,NCT02857582,NCT04247542,NCT02686645,NCT02801656,NCT00384527,NCT01598311,NCT01226992,NCT02743234,NCT02865616,NCT01987895,NCT03030248,NCT01232595,NCT04138706,NCT00269399,NCT02774382
|
Herpes simplex infectionDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1F00 |
NCT00150852,NCT01339091,NCT02202135,NCT01184872,NCT01764750,NCT04802837,NCT01757236,NCT02607618,NCT00430937,NCT01984684,NCT01252732,NCT03595566,NCT01756924,NCT00077675,NCT02374853,NCT04678154,NCT00426933,NCT05320068,NCT02227446,NCT03595553,NCT04017468,NCT00003805,NCT01499277,NCT04042077,NCT01400867,NCT03137173,NCT02600611,NCT05266807
|
Diabetic foot ulcerDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BD54 |
NCT01199783,NCT05076955
|
Pyogenic abscess of the skinDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1B75.3 |
NCT01339091,NCT01252732
|
PneumoniaDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CA40 |
NCT01798537,NCT00050401
|
Autism spectrum disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6A02 |
NCT04948814,NCT02504554
|
Crohn diseaseDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DD70 |
NCT02765256,NCT03476317
|
EndocarditisDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BB40-BA42 |
NCT00871104,NCT00695903
|
Bacterial cellulitis, erysipelas or lymphangitisDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1B70 |
NCT01339091,NCT01252732
|
DepressionDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6A70-6A7Z |
ADRA2A
|
Substance abuseDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6C40 |
ADRA2A
|
Attention deficit hyperactivity disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6A05 |
ADRA2A
|
Opioid use disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6C43 |
ADRA2A
|
Pain, unspecifiedDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: MG3Z |
ADRA2A
|
Other specified conjunctivitisDisease Category: 09.Diseases of the visual systemDisease ICD-11 Code: 9A60.Y |
NCT02432807
|
NosocomialDisease Category: X.Extension CodesDisease ICD-11 Code: XB25 |
NCT01515020
|
Pneumonia due to StaphylococcusDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CA40.06 |
NCT01925066
|
Ventilation associated with injury or harm in therapeutic useDisease Category: 23.External causes of morbidity or mortalityDisease ICD-11 Code: PK81.0 |
NCT00543608
|
Chronic rhinosinusitisDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CA0A |
NCT03673956
|
Cystic fibrosisDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CA25 |
NCT01594827
|
Non-small cell lung cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25 |
NCT03546829
|
Inflammatory arthropathyDisease Category: 15.Diseases of the musculoskeletal system or connective tissueDisease ICD-11 Code: FA2Z |
NCT03506347
|
Kidney failureDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GB60-GB6Z |
NCT02663596
|
OsteoarthritisDisease Category: 15.Diseases of the musculoskeletal system or connective tissueDisease ICD-11 Code: FA00-FA05 |
NCT04075526
|
Malignant haematopoietic neoplasmDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B33 |
NCT04983901
|
PeritonitisDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DC50 |
NCT03685747
|
Graft-versus-host diseaseDisease Category: 04.Diseases of the immune systemDisease ICD-11 Code: 4B24 |
NCT02641236
|
Chronic kidney diseaseDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GB61 |
NCT03452189
|
NADisease Category: NADisease ICD-11 Code: NA |
NCT01252732
|
Hepatocellular carcinoma, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH4W48 |
NCT04025567
|
Malignant neoplasm metastasis, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2E2Z |
NCT03817125
|
Sepsis of fetus or newbornDisease Category: 19.Certain conditions originating in the perinatal periodDisease ICD-11 Code: KA60 |
NCT02790996
|
ObesityDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5B80-5B81 |
NCT02037295
|
ConstipationDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: ME05.0 |
NCT02301221
|
Intermediate hyperglycaemia, unspecifiedDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A40.Z |
NCT02730962
|
PheochromocytomaDisease Category: X.Extension CodesDisease ICD-11 Code: XH3854 |
ADRA2A
|
Malignant neoplasms of biliary tract, distal bile ductDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C15 |
ADRA2A
|
Diffuse large B-cell lymphomasDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A81 |
ADRA2A
|
Myotonic dystrophyDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8C71.0 |
ADRA2A
|
Mesothelioma of pleuraDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C26.0 |
ADRA2A
|
Superficial ovarian endometriosisDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GA10.B4 |
ADRA2A
|
Adenocarcinoma of bronchus or lungDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25.0 |
ADRA2A
|
Malignant neoplasms of adrenal glandDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D11 |
ADRA2A
|

