Collective Molecular Activities of the Plant: Taxus Baccata
Country/Region:
Lebanon; IndiaTraditional Medicine System:
Medicinal Functions:
Anticonvulsant; Antispasmodic; Cancer; Cardiotonic; Diaphoretic; Emmenagogue; Expectorant; Homeopathy; Narcotic; Purgative
Turkey; Italy; India; France; Ireland; Norway; Belarus; Iran; Algeria; Germany; Belgium; Spain; Ukraine; Netherlands; Denmark; Poland; Finland; Morocco; Sweden; Switzerland; Russia; Bulgaria; Romania; Albania; Portugal; Lebanon; United Kingdom; Austria; Greece; Hungary
Overview of Ingredients
116 All known Ingredients in Total
Unique ingredients have been isolated from this plant.Plant-Ingredients Associations were manually curated from publications or collected from other databases.
18 Ingredients with Acceptable Bioavailablity
Unique ingredients exhibit acceptable human oral bioavailablity, according to the criteria of SwissADME [PMID: 28256516] and HobPre [PMID: 34991690]. The criteria details:SwissADME: six descriptors are used by SwissADME to evaluate the oral bioavailability of a natural product:
☑ LIPO(Lipophility): -0.7 < XLOGP3 < +5.0
☑ SIZE: 150g/mol < MW < 500g/mol
☑ POLAR(Polarity): 20Ų < TPSA < 130Ų
☑ INSOLU(Insolubility): -6 < Log S (ESOL) < 0
☑ INSATU(Insaturation): 0.25 < Fraction Csp3 < 1
☑ FLEX(Flexibility): 0 < Num. rotatable bonds < 9
If 6 descriptors of a natural plant satisfy the above rules, it will be labeled high HOB.
HobPre: A natural plant ingredient with HobPre score >0.5 is labeled high human oral availability (HOB)
28 Ingredients with experimental-derived Activity
Unique ingredients have activity data available.Ingredient Structrual Cards
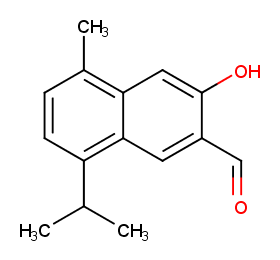
Ingredient ID: NPC98190

Ingredient ID: NPC9581
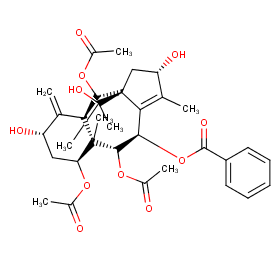
Ingredient ID: NPC87934
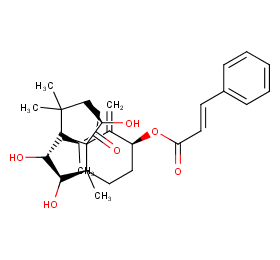
Ingredient ID: NPC86805

Ingredient ID: NPC85096
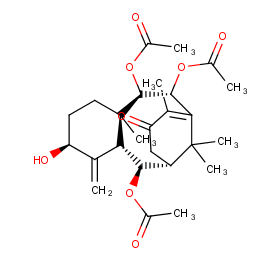
Ingredient ID: NPC8196
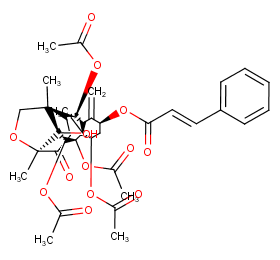
Ingredient ID: NPC80895
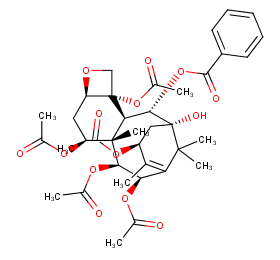
Ingredient ID: NPC79477
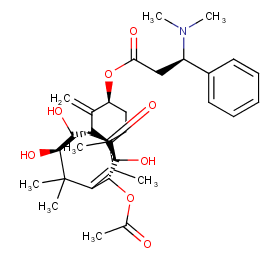
Ingredient ID: NPC73470

Ingredient ID: NPC73391
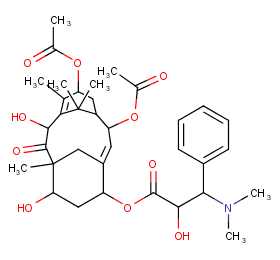
Ingredient ID: NPC67309
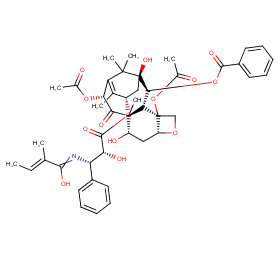
Ingredient ID: NPC67246

Ingredient ID: NPC59248
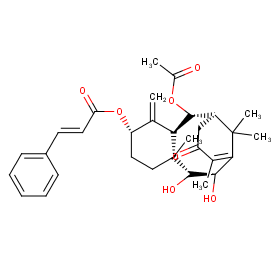
Ingredient ID: NPC58623
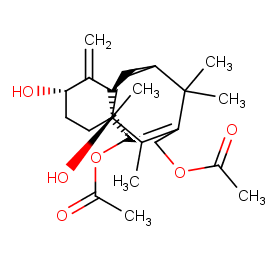
Ingredient ID: NPC57162
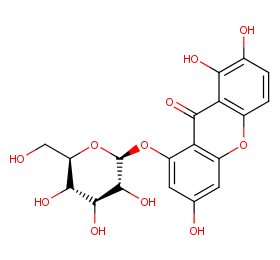
Ingredient ID: NPC55407
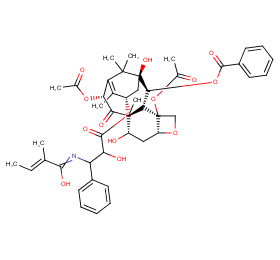
Ingredient ID: NPC52912
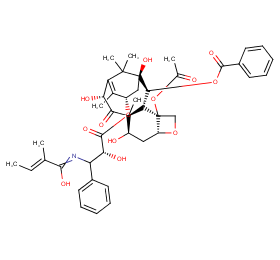
Ingredient ID: NPC49524

Ingredient ID: NPC48388

Ingredient ID: NPC471754

Ingredient ID: NPC471629
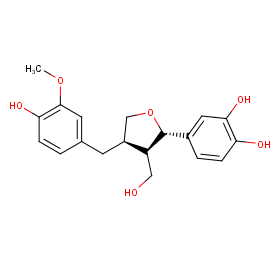
Ingredient ID: NPC42300
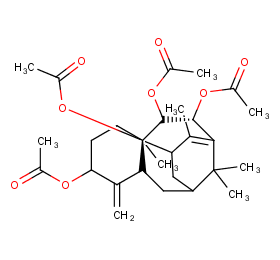
Ingredient ID: NPC41226
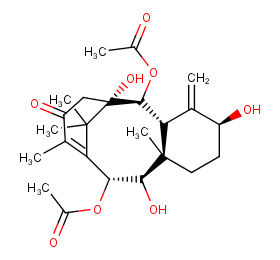
Ingredient ID: NPC4037
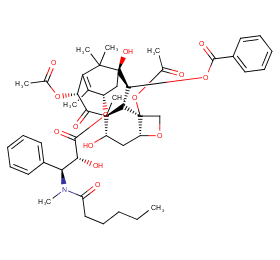
Ingredient ID: NPC36607

Ingredient ID: NPC33135
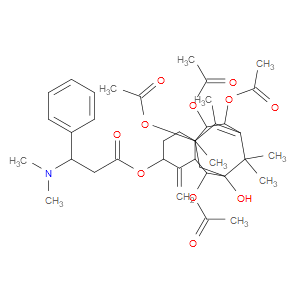
Ingredient ID: NPC328158
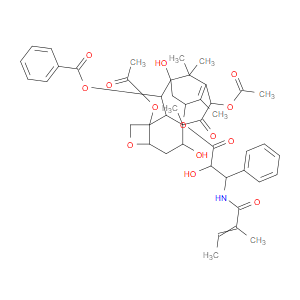
Ingredient ID: NPC327716

Ingredient ID: NPC327675
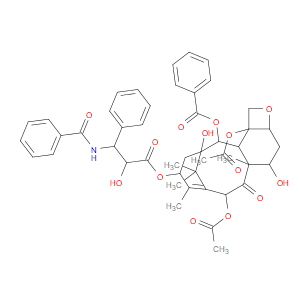
Ingredient ID: NPC327158

Ingredient ID: NPC327072
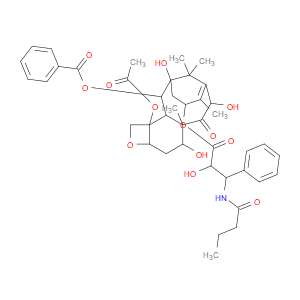
Ingredient ID: NPC327024
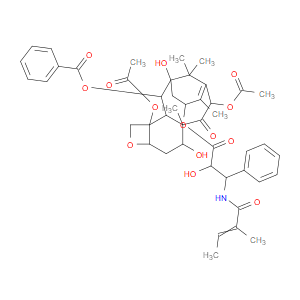
Ingredient ID: NPC326042
Ingredient ID: NPC324580
Ingredient ID: NPC324251

Ingredient ID: NPC324122

Ingredient ID: NPC323771

Ingredient ID: NPC323657

Ingredient ID: NPC321090

Ingredient ID: NPC321072

Ingredient ID: NPC320285
Ingredient ID: NPC320036
Ingredient ID: NPC319425
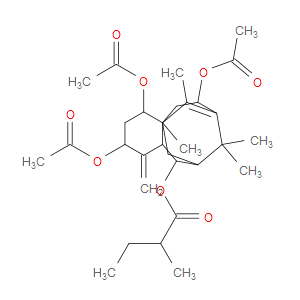
Ingredient ID: NPC316591
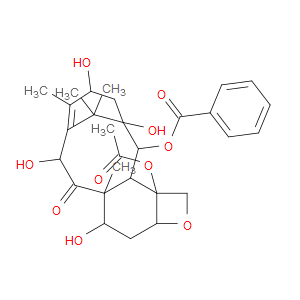
Ingredient ID: NPC316568
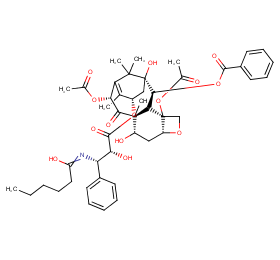
Ingredient ID: NPC306001
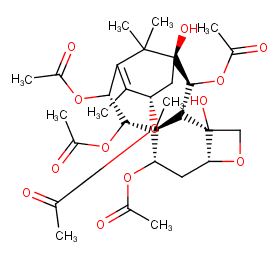
Ingredient ID: NPC303297
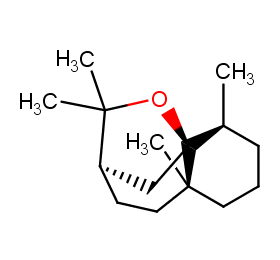
Ingredient ID: NPC301295
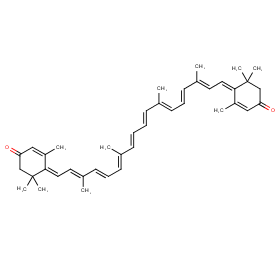
Ingredient ID: NPC29355

Ingredient ID: NPC289155

Ingredient ID: NPC284912

Ingredient ID: NPC284194
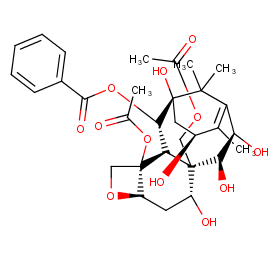
Ingredient ID: NPC281304

Ingredient ID: NPC280164
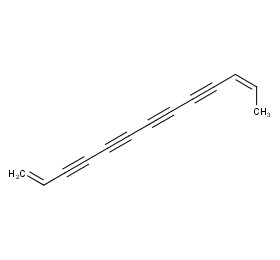
Ingredient ID: NPC275823
Ingredient ID: NPC275170

Ingredient ID: NPC274289

Ingredient ID: NPC271295
Ingredient ID: NPC263458

Ingredient ID: NPC259684
Ingredient ID: NPC25820
Ingredient ID: NPC257170
Ingredient ID: NPC256562
Ingredient ID: NPC247684
Ingredient ID: NPC246993
Ingredient ID: NPC242662
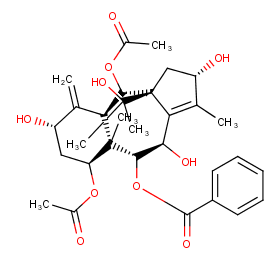
Ingredient ID: NPC241930

Ingredient ID: NPC241312
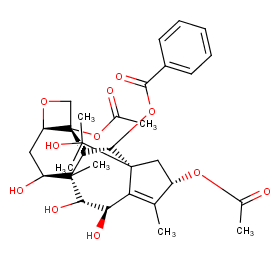
Ingredient ID: NPC238076

Ingredient ID: NPC237309
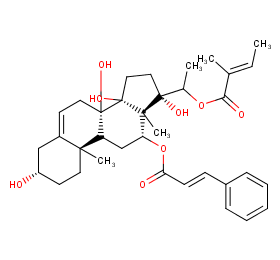
Ingredient ID: NPC237059
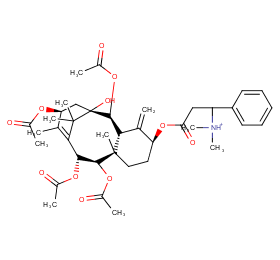
Ingredient ID: NPC236170

Ingredient ID: NPC235047
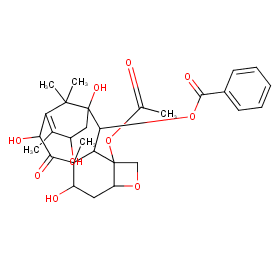
Ingredient ID: NPC229545

Ingredient ID: NPC229395
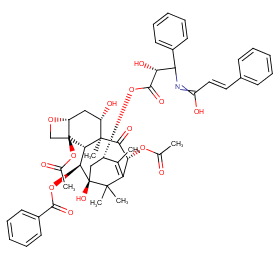
Ingredient ID: NPC22933
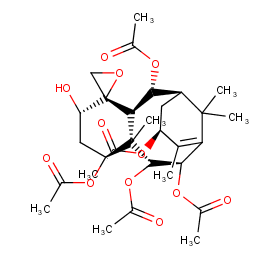
Ingredient ID: NPC229183
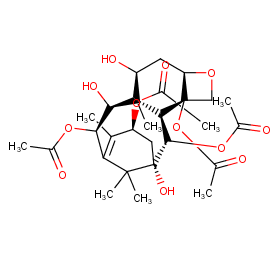
Ingredient ID: NPC228072
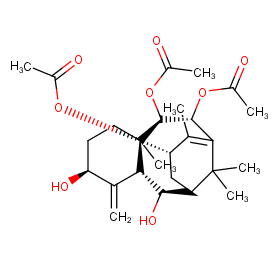
Ingredient ID: NPC225304
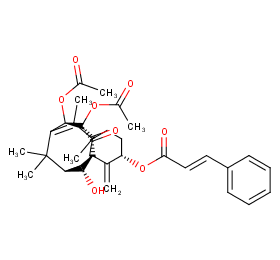
Ingredient ID: NPC225103
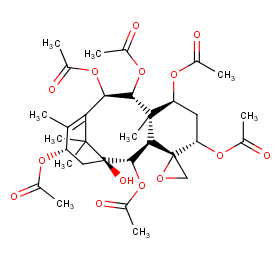
Ingredient ID: NPC223588
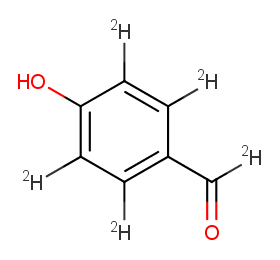
Ingredient ID: NPC216714
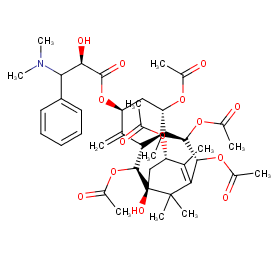
Ingredient ID: NPC216381
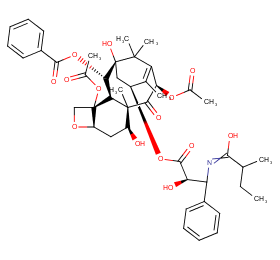
Ingredient ID: NPC215025
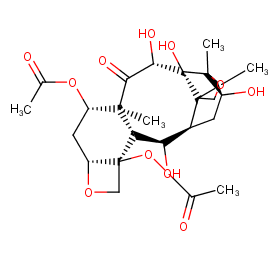
Ingredient ID: NPC212485

Ingredient ID: NPC211426

Ingredient ID: NPC209851
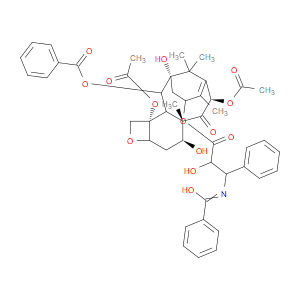
Ingredient ID: NPC208553
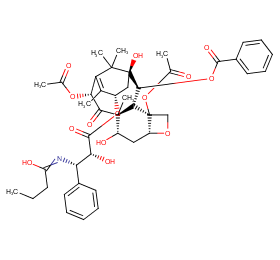
Ingredient ID: NPC206211
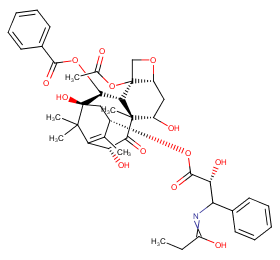
Ingredient ID: NPC200927
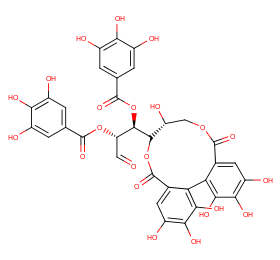
Ingredient ID: NPC200872

Ingredient ID: NPC200506
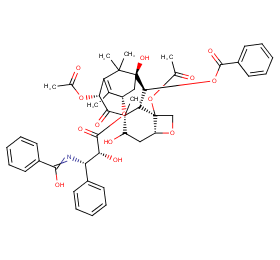
Ingredient ID: NPC184289
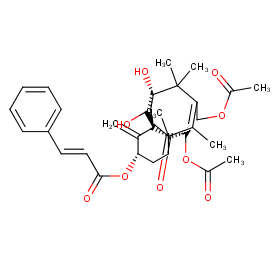
Ingredient ID: NPC181111
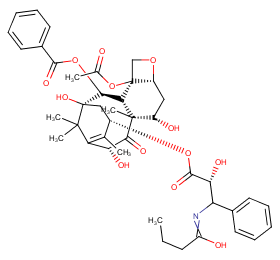
Ingredient ID: NPC179899
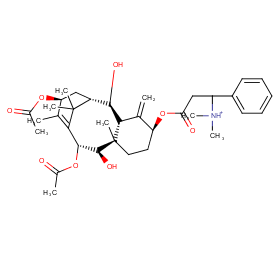
Ingredient ID: NPC175789
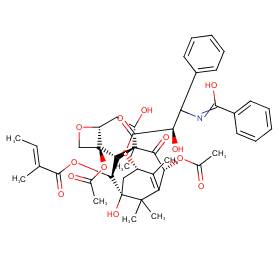
Ingredient ID: NPC166498
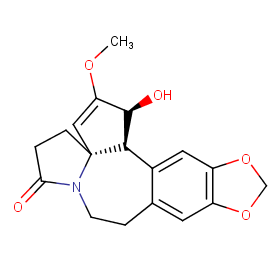
Ingredient ID: NPC16647

Ingredient ID: NPC163175
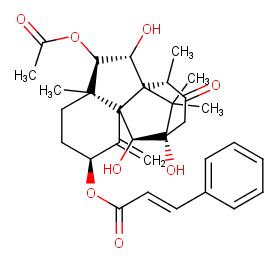
Ingredient ID: NPC161295

Ingredient ID: NPC157391
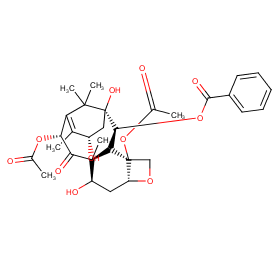
Ingredient ID: NPC155329

Ingredient ID: NPC155097
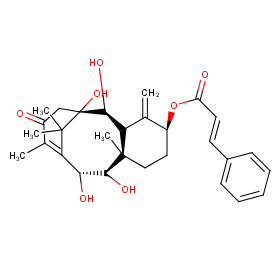
Ingredient ID: NPC15368
Ingredient ID: NPC151393
Ingredient ID: NPC14752

Ingredient ID: NPC147253

Ingredient ID: NPC146888
Ingredient ID: NPC138781
Ingredient ID: NPC123726
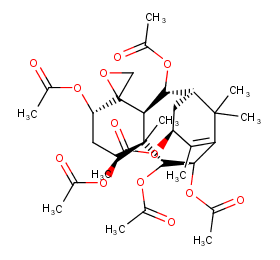
Ingredient ID: NPC122798

Ingredient ID: NPC117774

Ingredient ID: NPC116862
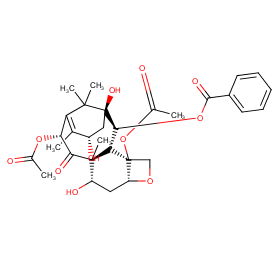
Ingredient ID: NPC114357
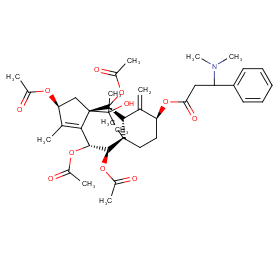
Ingredient ID: NPC10672
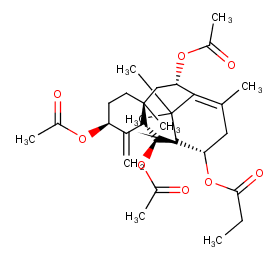
Ingredient ID: NPC102640
Classification of Human Proteins Collectively Targeted by the Plant
Detailed Information of Target Proteins
| Target Type | Protein Class | Gene ID | Protein Name | Uniprot ID | Target ChEMBL ID |
|---|---|---|---|---|---|
| Therapeutic Target | Hydrolase | RAB9A | Ras-related protein Rab-9A | P51151 | CHEMBL1293294 |
| Therapeutic Target | Nuclear hormone receptor subfamily 1 | THRB | Thyroid hormone receptor beta-1 | P10828 | CHEMBL1947 |
| Therapeutic Target | Protein Kinase | MTOR | Serine/threonine-protein kinase mTOR | P42345 | CHEMBL2842 |
| Drug Transporter | SLC superfamily of solute carriers | SLCO1B1 | Solute carrier organic anion transporter family member 1B1 | Q9Y6L6 | CHEMBL1697668 |
| Drug Transporter | SLC superfamily of solute carriers | SLCO1B3 | Solute carrier organic anion transporter family member 1B3 | Q9NPD5 | CHEMBL1743121 |
| Therapeutic Target | Structural protein | LMNA | Prelamin-A/C | P02545 | CHEMBL1293235 |
| Therapeutic Target | Structural protein | TUBB3 | Tubulin beta-3 chain | Q13509 | CHEMBL2597 |
| Therapeutic Target | Unclassified protein | PMP22 | Peripheral myelin protein 22 | Q01453 | CHEMBL1293298 |
| Therapeutic Target | Unclassified protein | GMNN | Geminin | O75496 | CHEMBL1293278 |
Clinical trials associated with plant from natural product (NP) & plant level:
| Clinical trials type | Number of clinical trials | |
|---|---|---|
| 2100 | ||
| NCT ID | Title | Condition | Form in clinical use | Associated by plant or compound |
|---|---|---|---|---|
| NCT04661696 | Dose Escalation of Lapatinib With Paclitaxel in Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT02483247 | A Safety, Tolerability, and Pharmacokinetics Study of MLN0128 as a Single Agent and in Combination With Paclitaxel in Adults With Advanced Nonhematologic Malignancies | cancer | Paclitaxel (NPC208553) | |
| NCT04929041 | Testing the Addition of Radiation Therapy to the Usual Treatment (Immunotherapy With or Without Chemotherapy) for Stage IV Non-Small Cell Lung Cancer Patients Who Are PD-L1 Negative | lung adenocarcinoma;squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT01376310 | GSK1120212 Rollover Study | cancer | Paclitaxel (NPC208553) | |
| NCT01090830 | Safety and Efficacy of Belinostat When Used With Standard of Care Chemotherapy for Untreated Non-small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00313768 | Randomized Ph 2 Trial Of Paclitaxel/Carboplatin /Bevacizumab + PF-3512676 And P/C/B Alone In Advanced Nonsquamous NSCLC | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00680940 | A Study of Mycobacterium w in Combination With Paclitaxel Plus Cisplatin in Advanced Non Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01333085 | Everolimus, Carboplatin, and Paclitaxel in Locally Advanced Head and Neck Cancer That Cannot Be Removed by Surgery | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT00052312 | Doxorubicin and Cisplatin With or Without Paclitaxel in Treating Patients With Locally Advanced, Metastatic, and/or Relapsed Endometrial Cancer | endometrial cancer | Paclitaxel (NPC208553) | |
| NCT01493505 | A Study of MM-121 With Paclitaxel in Platinum Resistant/ Refractory Advanced Ovarian Cancers | fallopian tube cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT03603184 | Atezolizumab Trial in Endometrial Cancer - AtTEnd | endometrial cancer | Paclitaxel (NPC208553) | |
| NCT00005046 | Paclitaxel in Treating Patients With Recurrent or Persistent Ovarian or Primary Peritoneal Cancer | ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT00505492 | Malignant Mixed Mesodermal Tumor (MMMT) - Early Stage With Postoperative XRT/Chemotherapy | uterine neoplasm | Paclitaxel (NPC208553) | |
| NCT01822756 | An Open-Label Study of Ruxolitinib Given With Chemotherapy in Patients With Advanced Solid Tumors | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT02054884 | F16IL2 Plus Paclitaxel in Metastatic Merkel Cell Carcinoma | Merkel cell skin cancer | Paclitaxel (NPC208553) | |
| NCT04814485 | Efficacy and Safety of SHR-1020 Combined With Albumin-bound Paclitaxel in the Second-line Treatment of Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT01171170 | Paclitaxel-Carboplatin-Bevacizumab +/- Nitroglycerin in Metastatic Non-Squamous-Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00030654 | Hormone Therapy Plus Chemotherapy in Treating Patients With Prostate Cancer | prostate cancer | Paclitaxel (NPC208553) | |
| NCT00001498 | A Pilot Trial of Sequential Chemotherapy With Antimetabolite Induction, High-Dose Alkylating Agent Consolidation With Peripheral Blood Progenitor Cell Support, and Intensification With Paclitaxel and Doxorubicin for Patients With High-Risk Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT01688791 | Weekly Paclitaxel, 5FU+Irinotecan or Carboplatin+Paclitaxel in Subjects With Advanced / Metastatic Solid Tumors | cancer | Paclitaxel (NPC208553) | |
| NCT01566435 | Induction Chemotherapy With ACF Followed by Chemoradiation Therapy for Adv. Head & Neck Cancer | upper aerodigestive tract neoplasm | Paclitaxel (NPC208553) | |
| NCT00806286 | Study of Carboplatin/Paclitaxel With or Without Investigational Drug (CS-7017) in Subjects With Metastatic Non-small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02486601 | NAB-PACLITAXEL Plus FOLFOX as Perioperative Chemotherapy in Patients With Operable Oesogastric Adenocarcinoma | stomach neoplasm | Paclitaxel (NPC208553) | |
| NCT02044601 | Biomarker-Integrated Approach of Targeted Therapy for Lung Cancer Elimination Plus External Beam Radiation Therapy (BATTLE-XRT) | lung cancer | Paclitaxel (NPC208553) | |
| NCT00111007 | A Treatment Combination for Patients With Unresectable Stage III or Stage IV Melanoma | melanoma | Paclitaxel (NPC208553) | |
| NCT00077129 | Paclitaxel and Carboplatin in Treating Patients With Locally Advanced or Metastatic Renal Cell Cancer | kidney cancer | Paclitaxel (NPC208553) | |
| NCT00003449 | Combination Chemotherapy in Treating Patients With Platinum-Resistant Recurrent Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT02468557 | Study of Single Agent Idelalisib Followed by Idelalisib in Combination With Chemotherapy in Adults With Metastatic Pancreatic Ductal Adenocarcinoma | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT01166542 | Efficacy Study of REOLYSIN® in Combination With Paclitaxel and Carboplatin in Platinum-Refractory Head and Neck Cancers | squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT03013218 | A Study of Evorpacept (ALX148) in Patients With Advanced Solid Tumors and Lymphoma (ASPEN-01) | non-Hodgkins lymphoma | Paclitaxel (NPC208553) | |
| NCT01797900 | The Role of Induction Chemotherapy for High-risk Locally Advanced Nasopharyngeal Carcinoma in the Era of IMRT | nasopharyngeal neoplasm | Paclitaxel (NPC208553) | |
| NCT02713386 | Ruxolitinib Phosphate, Paclitaxel, and Carboplatin in Treating Patients With Stage III-IV Epithelial Ovarian, Fallopian Tube, or Primary Peritoneal Cancer | endometrioid carcinoma | Paclitaxel (NPC208553) | |
| NCT01572038 | LUX-Breast 2 | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT02302807 | A Study of Atezolizumab Compared With Chemotherapy in Participants With Locally Advanced or Metastatic Urothelial Bladder Cancer [IMvigor211] | urinary bladder cancer | Paclitaxel (NPC208553) | |
| NCT00550784 | Combination Chemotherapy and Autologous Peripheral Stem Cell Transplant in Treating Patients With Stage III, Stage IV, or Recurrent Ovarian Epithelial Cancer, Primary Peritoneal Cancer, or Fallopian Tube Cancer | fallopian tube cancer;ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT04835896 | Study of M7824 and Paclitaxel Combination as a Second-line Treatment in Patients With Recurrent/Metastatic Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00585052 | A Phase II Study of Interaction of Lovastatin and Paclitaxel For Patients With Refractory or Relapsed Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT01444547 | A Study on Predictive Value of ERCC1 in Esophageal Cancer Patients Treated With Paclitaxel and Cisplatin | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00368875 | Phase I-II Study of Vorinostat, Paclitaxel, and Bevacizumab in Metastatic Breast Cancer | male breast carcinoma | Paclitaxel (NPC208553) | |
| NCT01938833 | Romidepsin and Abraxane in Treating Patients With Metastatic Inflammatory Breast Cancer | inflammatory breast carcinoma;male breast carcinoma | Paclitaxel (NPC208553) | |
| NCT00003284 | High-Dose Combination Chemotherapy Followed by Peripheral Stem Cell Transplantation in Treating Patients With Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00005646 | Paclitaxel in Treating Patients With Extensive-Stage Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00139633 | Selective Dose Escalation for Esophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00003957 | Combination Chemotherapy Plus Peripheral Stem Cell Transplantation in Treating Patients With Relapsed Non-Hodgkin's Lymphoma | lymphoma | Paclitaxel (NPC208553) | |
| NCT00030368 | Bortezomib and Paclitaxel in Treating Patients With Locally Advanced or Metastatic Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT04781413 | Nab-PTX Plus S-1 and Sintilimab as Adjuvant Therapy in Patients With Stage IIIC Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT01234038 | Safety and Tolerability Study of ISIS EIF4E Rx in Combination With Carboplatin and Paclitaxel | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02033538 | Neoadjuvant Chemotherapy of Nanoparticle Albumin-bound Paclitaxel in Squamous Cell Carcinoma of Esophagus | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT05357846 | PD-1 Inhibitor Combined With Neoadjuvant Chemoradiotherapy Plus Surgery for Locally Advanced ESCC | esophageal carcinoma | Paclitaxel (NPC208553) | |
| NCT00610714 | AZD0530 Phase II Study in Patients With Advanced Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00960960 | A Study of PI3-Kinase Inhibitor GDC-0941 in Combination With Paclitaxel, With and Without Bevacizumab or Trastuzumab, and With Letrozole, in Participants With Locally Recurrent or Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT01650376 | Phase Ib Study of Olaparib Plus Weekly Carboplatin and Paclitaxel in Relapsed Ovarian Cancer | uterine cancer | Paclitaxel (NPC208553) | |
| NCT00452803 | Pre-operative Chemotherapy Versus Concurrent Chemoradiotherapy in N2 Positive IIIA Non Small Cell Lung Cancer (NSCLC) | lung cancer | Paclitaxel (NPC208553) | |
| NCT00230451 | Pre-operative Chemotherapy and Radiation Therapy for Esophageal Carcinoma | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT04278092 | Paclitaxel Plus Cetuximab After First-line Checkpoint Inhibitor Failure | head and neck squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT04251533 | A Study of Novel Anti-cancer Agents in Patients With Metastatic Triple Negative Breast Cancer | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT01851174 | Study to Evaluate Bi-weekly Dosing of Gemcitabine Plus Nab-Paclitaxel to Treat Metastatic Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT02124317 | Nab-paclitaxel Plus S-1 in Patients With Advanced Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00028938 | Chemotherapy and Radiation Therapy With or Without Epoetin Alfa in Treating Patients With Stage IIIA or Stage IIIB Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT02340117 | Study of Combined SGT-53 Plus Gemcitabine/Nab-Paclitaxel for Metastatic Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT01174238 | A Two Arm Trial of Axitinib and Carboplatin/Paclitaxel in Melanoma | melanoma | Paclitaxel (NPC208553) | |
| NCT00864318 | Dose Intensification Study in Refractory Germ Cell Tumors With Relapse and Bad Prognosis | germ cell tumor | Paclitaxel (NPC208553) | |
| NCT01187199 | Phase I Trial of Bevacizumab and Temsirolimus in Combination With 1) Carboplatin, 2) Paclitaxel, 3) Sorafenib for the Treatment of Advanced Cancer | cancer | Paclitaxel (NPC208553) | |
| NCT01868022 | Study to Evaluate GSK3052230 in Combination With Paclitaxel and Carboplatin, or Docetaxel or as Single Agent in Subjects With Solid Malignancies and Deregulated Fibroblast Growth Factor (FGF) Pathway Signaling | neoplasm | Paclitaxel (NPC208553) | |
| NCT04015661 | Induction Chemotherapy Followed by Concurrent Chemoradiotherapy in Patients With LA-NPC | nasopharyngeal neoplasm | Paclitaxel (NPC208553) | |
| NCT01593306 | Concurrent Chemoradiotherapy With Weekly Cisplatin Versus Concurrent Chemoradiotherapy With Weekly Cisplatin and Paclitaxel in Locally Advanced Carcinoma Cervix | carcinoma | Paclitaxel (NPC208553) | |
| NCT01301716 | A Study of the Safety and Pharmacology of GDC-0980 in Combination With Either Paclitaxel and Carboplatin (With or Without Bevacizumab) or Pemetrexed and Cisplatin in Patients With Solid Tumors | cancer | Paclitaxel (NPC208553) | |
| NCT00236899 | Phase III Study of Two Different Schedules (Weekly and Tri-weekly) of Combination of Gemcitabine and Two Taxanes in MBC | breast cancer | Paclitaxel (NPC208553) | |
| NCT00003127 | S9720 Combination Chemotherapy in Treating Patients With Metastatic, Recurrent, or Refractory Endometrial Cancer | endometrial cancer | Paclitaxel (NPC208553) | |
| NCT00001569 | Phase I Trial of Continuous Hyperthermic Peritoneal Perfusion (CHPP) With Cisplatin Plus Early Postoperative Intraperitoneal Paclitaxel and 5-FU for Peritoneal Carcinomatosis | peritoneal neoplasm;carcinoma | Paclitaxel (NPC208553) | |
| NCT01263145 | MK2206 and Paclitaxel in Treating Patients With Locally Advanced or Metastatic Solid Tumors or Metastatic Breast Cancer | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT03740165 | A Study of the Efficacy and Safety of Bevacizumab in Chinese Women With Newly Diagnosed, Previously Untreated Stage III or Stage IV Epithelial Ovarian, Fallopian Tube, or Primary Peritoneal Cancer | fallopian tube cancer | Paclitaxel (NPC208553) | |
| NCT02243007 | Phase II Study of Preoperative FOLFIRINOX Versus Gemcitabine/Nab-Paclitaxel in Patients With Resectable Pancreatic Cancer | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00639522 | Dose Escalation Study of Liposomal Paclitaxel With/Without Capecitabine in Patients With Advanced Gastric Carcinoma | gastric carcinoma | Paclitaxel (NPC208553) | |
| NCT02317991 | Study of Nab-Paclitaxel and Ramucirumab as Second-line Treatment for Patients With Metastatic Gastroesophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00062010 | Interferon Alfa, Isotretinoin, and Paclitaxel in Treating Patients With Recurrent Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00003120 | S9701 Paclitaxel in Treating Patients With Advanced Ovarian, Fallopian Tube, or Primary Peritoneal Cancer in Remission | fallopian tube cancer;ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT00584857 | A Phase II Study of Therapy With Paclitaxel, Carboplatin and Megesterol Acetate for the Management of Uterine Cancer | uterine cancer | Paclitaxel (NPC208553) | |
| NCT02603679 | Neoadjuvant Response-guided Treatment of Luminal B-type Tumors and Luminal A-type Tumors With Node Metastases | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT01366131 | Study Evaluating the Safety and Efficacy of MEGF0444A in Combination With Carboplatin, Paclitaxel and Bevacizumab in Patients With Advanced or Recurrent Non-Squamous Non-Small Cell Lung Cancer Who Have Not Received Prior Chemotherapy for Advanced Disease (NILE) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02834975 | Adavosertib Plus Chemotherapy in Platinum-Resistant Epithelial Ovarian, Fallopian Tube, or Primary Peritoneal Cancer | peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT00894569 | Paclitaxel/Carboplatin With or Without Cetuximab in CUP | carcinoma | Paclitaxel (NPC208553) | |
| NCT01276496 | Weekly Doses of Cilengitide and Paclitaxel in Treating Patients With Advanced Solid Tumors That Cannot Be Removed by Surgery | male breast carcinoma | Paclitaxel (NPC208553) | |
| NCT00168883 | Study for Patients With Non Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01160601 | Study of Paclitaxel/Carboplatin With or Without Bavituximab in Previously Untreated Non Small-Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00006110 | Multimodality Treatment for Women With Stage II, Stage III, or Stage IV Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT03083470 | Study of SOR007 Ointment for Actinic Keratosis | actinic keratosis | Paclitaxel (NPC208553) | |
| NCT00004173 | Oxaliplatin and Paclitaxel in Treating Patients With Metastatic or Unresectable Cancer | neoplasm | Paclitaxel (NPC208553) | |
| NCT01527864 | Paclitaxel-Carboplatin Alone or With M2ES for Non-Small-Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00280150 | Combination Chemotherapy, Bev, RT, and Erlotinib in Treating Patients With Stage III Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00128856 | Gemcitabine, Doxorubicin and Paclitaxel (GAT) as Neoadjuvant Treatment of Breast Cancer Patients | breast cancer | Paclitaxel (NPC208553) | |
| NCT00675259 | Phase II NCT (Neoadjuvant Chemotherapy) w/ Weekly Abraxane in Combination With Carboplatin & Bevacizumab in Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00004097 | Combination Chemotherapy Plus Radiation Therapy in Treating Patients With Advanced and/or Recurrent Head and Neck Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT02366949 | A Study MLN2480 in Combination With MLN0128 or Alisertib, or Paclitaxel, or Cetuximab, or Irinotecan in Adult Participants With Advanced Nonhematologic Malignancies | cancer | Paclitaxel (NPC208553) | |
| NCT00005635 | Trastuzumab Plus Paclitaxel in Treating Women With Metastatic Breast Cancer That Overexpresses HER2 | breast cancer | Paclitaxel (NPC208553) | |
| NCT00976677 | Carboplatin, Paclitaxel, and Bevacizumab With or Without Erlotinib Hydrochloride in Treating Non-Smokers With Advanced Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00063258 | Tarceva Surgery for Resectable Stage IIIA(N2) and IIIB (T4 N2) Non-Small-Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00307255 | Paclitaxel Albumin-Stabilized Nanoparticle Formulation and Gemcitabine in Treating Patients With Advanced Metastatic Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT03219762 | Study of Nab-paclitaxel in Sensitive and Refractory Relapsed SCLC | small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00311636 | Triptorelin in Preventing Early Menopause in Premenopausal Women Who Are Receiving Chemotherapy for Stage I, Stage II, or Stage III Breast Cancer That Has Been Removed By Surgery | breast cancer | Paclitaxel (NPC208553) | |
| NCT00176267 | Paclitaxel, Carboplatin And Low Dose Radiation As Induction Therapy In Locally Advanced Head And Neck Cancer | squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT03588039 | Study of Oraxol and Pembrolizumab in Subjects With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT05254171 | Study of Nab-Paclitaxel and Gemcitabine With or Without SBP-101 in Pancreatic Cancer | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT01583426 | Nanoparticle-based Paclitaxel vs Solvent-based Paclitaxel as Part of Neoadjuvant Chemotherapy for Early Breast Cancer (GeparSepto) | inflammatory breast carcinoma | Paclitaxel (NPC208553) | |
| NCT00016874 | 3-AP Plus Cisplatin and Paclitaxel in Treating Patients With Advanced or Metastatic Cancer | neoplasm | Paclitaxel (NPC208553) | |
| NCT02073968 | PET-Adjusted Intensity Modulated Radiation Therapy and Combination Chemotherapy in Treating Patients With Stage II-IV Non-small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00002854 | High-Dose Combination Chemotherapy Plus Peripheral Stem Cell Transplantation in Treating Patients With Advanced Cancer | cancer | Paclitaxel (NPC208553) | |
| NCT00001442 | A Pilot Study of Paclitaxel With Radiation Therapy for Locally Advanced Head and Neck Cancer | squamous cell carcinoma;upper aerodigestive tract neoplasm | Paclitaxel (NPC208553) | |
| NCT02784288 | Phase II Treatment Stratification Trial Using Neck Dissection-Driven Selection to Improve Quality of Life for Low Risk Patients With HPV+ Oropharyngeal Squamous Cell Cancer | squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT05190445 | Cinrebafusp Alfa in Combination With Ramucirumab and Paclitaxel in HER2-High Gastric or GEJ Adenocarcinoma and in Combination With Tucatinib in HER2-Low Gastric or GEJ Andenocarinoma | gastric cancer | Paclitaxel (NPC208553) | |
| NCT01591135 | A Phase III Study of Comparing Paclitaxel Plus 5-Fluorouracil Versus Cisplatin Plus 5-Fluorouracil in Chemoradiotherapy for Locally Advanced Esophageal Carcinoma | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT04314895 | Trial of NanoPac Intratumoral Injection in Lung Cancer | small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02106546 | Study Comparing Veliparib Plus Carboplatin and Paclitaxel Versus Placebo Plus Carboplatin and Paclitaxel in Previously Untreated Advanced or Metastatic Squamous Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02934464 | Assessment of Ramucirumab Plus Paclitaxel as Switch MANteInance Versus Continuation of First-line Chemotherapy in Patients With Advanced HER-2 Negative Gastric or Gastroesophageal Junction Cancers | stomach neoplasm | Paclitaxel (NPC208553) | |
| NCT02187991 | Study to Compare Alisertib With Paclitaxel vs. Paclitaxel Alone in Metastatic or Locally Recurrent Breast Cancer | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT01891357 | Phase II Trial to Validate Markers for a Response Evaluation of a Combined Therapy in Patients With HER2+ Breast Cancer | breast ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00945191 | A Study of First Line Treatment With Avastin (Bevacizumab) in Combination With Carboplatin and Weekly Paclitaxel in Patients With Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00010257 | Carboplatin Combined With Paclitaxel in Treating Patients With Advanced Thymoma | Thymic Carcinoma;Thymoma | Paclitaxel (NPC208553) | |
| NCT00046527 | Study of ABI-007 and Taxol in Patients With Metastatic Breast Cancer | metastasis;breast neoplasm | Paclitaxel (NPC208553) | |
| NCT00578149 | Bevacizumab and Carboplatin/Paclitaxel and Radiation in Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00486668 | A Study of AC Followed by a Combination of Paclitaxel Plus Trastuzumab or Lapatinib or Both Given Before Surgery to Patients With Operable HER2 Positive Invasive Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT03635489 | Carboplatin-Paclitaxel-Bevacizumab vs Carbo-Pacli-Beva-Rucaparib vs Carbo-Pacli-Ruca, Selected According to HRD Status, in Patients With Advanced Ovarian, Primary Peritoneal and Fallopian Tube Cancer, Preceded by a Phase I Dose Escalation Study on Ruca-Beva Combination | fallopian tube cancer | Paclitaxel (NPC208553) | |
| NCT04753879 | Multi-agent Low Dose Chemotherapy GAX-CI Followed by Olaparib and Pembro in Metastatic Pancreatic Ductal Cancer. | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00003862 | Chemotherapy and Radiation Therapy in Treating Patients With Stomach Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT01581476 | Adolescent Type 1 Diabetes Cardio-Renal Intervention Trial | type 1 diabetes mellitus | Paclitaxel (NPC208553) | |
| NCT00008138 | S0009 Combination Chemo and Surgery in Stage III or Stage IV Ovarian Cancer | fallopian tube cancer;ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT03740165 | Study of Chemotherapy With Pembrolizumab (MK-3475) Followed by Maintenance With Olaparib (MK-7339) for the First-Line Treatment of Women With BRCA Non-mutated Advanced Epithelial Ovarian Cancer (EOC) (MK-7339-001/KEYLYNK-001/ENGOT-ov43/GOG-3036) | peritoneal neoplasm | Paclitaxel (NPC208553) | |
| NCT01220154 | Neoadjuvant Chemotherapy IV Carboplatin With Weekly Paclitaxel Bevacizumab for Primary Ovarian | fallopian tube cancer;ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00054210 | Polyglutamate Paclitaxel Plus Carboplatin Compared With Paclitaxel Plus Carboplatin in Treating Patients With Advanced or Recurrent Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT03467178 | Pembrolizumab With Chemotherapy in Front Line Advanced Ovarian, Primary Peritoneal and Fallopian Tube Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT05244993 | Study of Pembrolizumab (MK-3475) Plus Chemotherapy Versus Placebo Plus Chemotherapy for HR+/HER2- Locally Recurrent Inoperable or Metastatic Breast Cancer (MK-3475-B49/KEYNOTE-B49) | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT04821284 | Sonoporation and Chemotherapy for the Treatment of Pancreatic Cancer | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00707707 | Phase I/II Study of AZD2281 Given in Combination With Paclitaxel in Metastatic Triple Negative Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT03762564 | Study With Paclitaxel +/- Ramucirumab in Patients With Squamous-cell Carcinoma of the Esophagus After Prior Therapy | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT02193633 | Phase I Dose Escalation of Oral BAY1161909 in Combination With Intravenous Paclitaxel | cancer | Paclitaxel (NPC208553) | |
| NCT00553358 | Phase II Trial to Compare the Safety of Two Chemotherapy Plus Trastuzumab Regimens as Adjuvant Therapy for HER2-positive Breast Cancer (Study P05048) | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT00154882 | Paclitaxel (Phyxol) and Cisplatin as First-line Chemotherapy for Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00039039 | Combination Chemotherapy Followed by Radiation Therapy With or Without Paclitaxel in Treating Patients With Unresectable Stage III Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT05083247 | Preoperative mFOLFIRINOX (or Gem-Nab-P) +/- Isotoxic High-dose SBRT for Borderline Resectable Pancreatic Adenocarcinoma | pancreatic neoplasm | Paclitaxel (NPC208553) | |
| NCT00022191 | Cisplatin Plus Gemcitabine With or Without Paclitaxel in Treating Patients With Stage IV Urinary Tract Cancer | urethra cancer;urinary bladder cancer | Paclitaxel (NPC208553) | |
| NCT00193531 | Paclitaxel, Carboplatin, and Oral Etoposide Followed by Weekly Paclitaxel in High Grade Neuroendocrine Carcinoma | neuroendocrine carcinoma | Paclitaxel (NPC208553) | |
| NCT00259675 | Alternating Cycles of Carboplatin/Gemcitabine and Carboplatin/Taxol for Advanced Stage NSCLC | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00314678 | Cisplatin Induction With Paclitaxel Consolidation for Stage III-IV Epithelial Ovarian and Primary Peritoneal Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT01969955 | Nab-paclitaxel as Second-line Therapy in Locally Advanced or Metastatic Squamous Lung Cancer | squamous cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00392392 | Preoperative Bevacizumab and Trastuzumab With ABI-007 and Carboplatin in HER2+ Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT01507428 | Study of Positron Emission Tomography and Computed Tomography in Guiding Radiation Therapy in Patients With Stage III Non-small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT04150575 | A Clinical Study to Evaluate Efficacy and Safety of HLX10 Combined With Albumin-Bound Paclitaxel in Patients With Advanced Cervical Cancer Who Have Progressive Disease or Intolerable Toxicity After First-Line Standard Chemotherapy | cervical cancer | Paclitaxel (NPC208553) | |
| NCT03718624 | A Study of Intraperitoneal and Intravenous Paclitaxel Plus Apatinib and S-1 Conversion Therapy for Gastric Cancer With Positive Exfoliative Cancer Cells | gastric cancer | Paclitaxel (NPC208553) | |
| NCT03047265 | Concurrent Chemoradiotherapy in Patients With Locoregionally Advanced Nasopharyngeal Carcinoma | nasopharyngeal neoplasm | Paclitaxel (NPC208553) | |
| NCT01964534 | Efficacy of ABI-007 Plus Gemcitabine or sLV5FU2 as First-line Therapy in Patients With Metastatic Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT01558492 | Carboplatin and Paclitaxel in Patients With Metastatic, Castrate-Resistant Prostate Cancer | prostate cancer | Paclitaxel (NPC208553) | |
| NCT02598687 | Testing TH-302, in Combination With Preoperative Chemoradiotherapy, in Esophageal Cancer. | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00611962 | Phase II Study of Oxaliplatine-Paclitaxel in Patients With Metastatic Germ Cell Tumor Refractory to Cisplatin | germ cell tumor | Paclitaxel (NPC208553) | |
| NCT03901118 | Efficacy and Safety of Paclitaxel for Injection (Albumin-bound) for First-line Chemotherapy of Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT05179239 | A Study of SHR-1701 Plus Platinum-containing Chemotherapy With or Without BP102 (Bevacizumab) as First-line Treatment in Cervical Cancer | cervical cancer | Paclitaxel (NPC208553) | |
| NCT01770301 | Efficacy and Safety of Bevacizumab (Avastin®) Combined to Weekly Paclitaxel Followed by Bevacizumab (Avastin®) Alone in Patients With Relapsed Ovarian Sex-cord Stromal Tumours (ALIENOR) | sex cord-gonadal stromal tumor | Paclitaxel (NPC208553) | |
| NCT00511459 | Phase 2 Study of AMG 386 Plus Paclitaxel With or Without Bevacizumab as First Line Therapy in Her2-Negative Breast Cancer Patients | breast cancer | Paclitaxel (NPC208553) | |
| NCT00815308 | Erbitux Combined With Chemo-radiotherapy in Esophageal Squamous Cell Carcinoma | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00002972 | Paclitaxel in Treating Patients With Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00326456 | MITO-2: A Study Comparing 2 Chemotherapy Regimens (Carboplatin/Liposomal Doxorubicin vs Carboplatin/Paclitaxel) in Patients With Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT02283372 | Nab-Paclitaxel Plus Gemcitabine With Concurrent MR-Guided IMRT in Patients With Locally Advanced Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00193362 | Study of Paclitaxel, Carboplatin, and Gemcitabine Versus Gemcitabine and Vinorelbine for Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT03940001 | A Study of Sintilimab Plus Chemoradiation Before Surgery for Esophageal Cancer | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00795899 | Taxol Epirubicin Cyclophosphamide Herceptin Neoadjuvant (TECHNO) | breast cancer | Paclitaxel (NPC208553) | |
| NCT02513563 | AZD1775 Plus Carboplatin-Paclitaxel in Squamous Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00811993 | A Study of R1507 in Combination With Multiple Standard Chemotherapy Treatments in Patients With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT05455918 | Paclitaxel/Ifosfamide/Cisplatin Chemotherapy for High Risk Pediatric Germ Cell Tumor | childhood germ cell tumor | Paclitaxel (NPC208553) | |
| NCT00028990 | Paclitaxel With or Without Bevacizumab in Treating Patients With Locally Recurrent or Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT03278015 | Phase 2 Trial of Gemcitabine vs S-1 vs Gemcitabine Plus Nab-paclitaxel as Adjuvant Chemotherapy of Post-operative Pancreatic Cancer Patients | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00373217 | Vaccine Therapy, Paclitaxel, and Carboplatin in Treating Patients Who Are Undergoing Surgery for Stage III or Stage IV Ovarian Cancer, Primary Peritoneal Cancer, or Fallopian Tube Cancer | fallopian tube cancer;ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT03577704 | The Safety,Efficacy of Anti-EGFR Humanized Monoclonal Antibody Combined With Chemotherapy in Advanced Solid Tumors. | neoplasm | Paclitaxel (NPC208553) | |
| NCT02017015 | Safety and Efficacy Study of Nab-paclitaxel Plus Gemcitabine in Chinese Patients With Metastatic Pancreatic Cancer | pancreatic neoplasm | Paclitaxel (NPC208553) | |
| NCT01447706 | Pazopanib Hydrochloride, Paclitaxel, and Carboplatin in Treating Patients With Refractory or Resistant Ovarian Epithelial Cancer, Fallopian Tube Cancer, or Peritoneal Cancer | fallopian tube cancer;ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT05125055 | Neoadjuvant Anti-PD-1 and TP Versus TPF on Pathological Response in OSCC | oral squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00274456 | Phase II Trial Comparing ABI-007 (Abraxane®, Nab®-Paclitaxel) to Taxotere in First Line Therapy of Patients With Stage IV Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT04081389 | Chemokine Modulation Therapy and Standard Chemotherapy Before Surgery for the Treatment of Early Stage Triple Negative Breast Cancer | breast carcinoma in situ | Paclitaxel (NPC208553) | |
| NCT00607048 | Dose Finding Study Of CP-870,893, An Immune System Stimulating Antibody, In Combination With Paclitaxel And Carboplatin For Patients With Metastatic Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT00003591 | Radiation Therapy Plus Paclitaxel in Treating Patients With Nonmetastatic, Unresectable Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT01778803 | A Study of Bevacizumab (Avastin) in Neoadjuvant Therapy in Participants With International Federation of Gynecology and Obstetrics (FIGO) Stage IIIC/IV Ovarian, Tubal, or Peritoneal Cancer, Initially Unresectable | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00886717 | Imatinib Mesylate (Gleevec) and Paclitaxel in Recurrent Patients of Ovarian and Other Cancers of Mullerian Origin | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT03704077 | An Investigational Immuno-therapy Study of Relatlimab Plus Nivolumab Compared to Various Standard-of-Care Therapies in Previously Treated Participants With Recurrent, Advanced or Metastatic Gastric Cancer or Gastroesophageal Junction Adenocarcinoma | gastric cancer | Paclitaxel (NPC208553) | |
| NCT01649947 | Modulation of Autophagy in Patients With Advanced/Recurrent Non-small Cell Lung Cancer - Phase II | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02630823 | MK-3475 Immunotherapy in Endometrial Carcinoma | endometrial carcinoma | Paclitaxel (NPC208553) | |
| NCT00003972 | Combination Chemotherapy and Peripheral Stem Cell Transplantation in Treating Patients With Stage II or Stage IIIA Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00653952 | CAELYX Versus Paclitaxel HCl in Patients With Epithelial Ovarian Carcinoma Following Failure of First-Line, Platinum-Based Chemotherapy | ovarian neoplasm | Paclitaxel (NPC208553) | |
| NCT00369551 | Bevacizumab, Paclitaxel, Carboplatin, and Radiation Therapy to the Chest in Treating Patients With Locally Advanced Non-Small Cell Lung Cancer | lung adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00068757 | Lonafarnib, Trastuzumab, and Paclitaxel in Treating Patients With HER2/Neu-Overexpressing Stage IIIB, Stage IIIC, or Stage IV Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00003004 | Combination Chemotherapy in Treating Patients With Refractory or Recurrent Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT03344172 | Pre-Operative Trial (PGHA vs. PGH) for Resectable Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00194792 | Hormone Therapy and Combination Chemotherapy Before and After Surgery in Treating Patients With Stage I-IIIA Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT01967043 | A Study to Evaluate Safety, Tolerability, Pharmacokinetics and Activity of Oraxol in Subjects With Advanced Malignancies | neoplasm | Paclitaxel (NPC208553) | |
| NCT00024388 | Chemotherapy in Treating Patients With Metastatic Kidney Cancer | kidney cancer | Paclitaxel (NPC208553) | |
| NCT00003896 | S9912 Combination Chemo in Stage III Ovarian Cancer, | fallopian tube cancer;ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT01818063 | Carboplatin and Combination Chemotherapy With or Without Veliparib in Treating Patients With Stage IIB-IIIC Breast Cancer | triple-negative breast cancer | Paclitaxel (NPC208553) | |
| NCT03731442 | Salvage Chemoradiation Therapy for Recurrence After Radical Surgery or Palliative Surgery in Esophageal Cancer Patients | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00217607 | Paclitaxel in Treating Patients With Locally Advanced or Metastatic Soft Tissue Angiosarcoma or Lymphangiosarcoma That Cannot Be Removed By Surgery | sarcoma | Paclitaxel (NPC208553) | |
| NCT00343291 | A Study of Cetuximab and Bevacizumab in Combination With Paclitaxel and Carboplatin in Stage IIIb/IV NSCLC | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00057928 | S0227 Cisplatin With Either Paclitaxel or Gemcitabine in Recurrent, Persistent, or Metastatic Cervical Cancer | cervical cancer | Paclitaxel (NPC208553) | |
| NCT02405910 | Ph2 Nab-paclitaxel With Gemcitabine to Determine Efficacy in Advanced Non-squamous NSCLC. | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00662311 | Vorinostat, Paclitaxel, and Radiation Therapy in Treating Patients Unable to Tolerate Cisplatin With Stage III Non-Small Lung Cancer That Cannot Be Removed By Surgery | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00368992 | S0536: Cetuximab, Paclitaxel, Carboplatin, and Bevacizumab in Treating Patients With Advanced Non-Small Cell Lung Cancer | lung adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT03915444 | Nab-Paclitaxel + Cisplatin + Gemcitabine in Untreated Metastatic Pancreatic Adenocarcinoma | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT01357161 | Study of Ombrabulin in Patients With Platinum-Sensitive Recurrent Ovarian Cancer Treated With Carboplatin/Paclitaxel | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT04223024 | Concurrent Chemoradiotherapy With Nimotuzumab for High Risk Nasopharyngeal Carcinoma | nasopharyngeal neoplasm | Paclitaxel (NPC208553) | |
| NCT03025477 | Safety and Efficacy of Quisinostat, a Histone Deacetylase Inhibitor, in Combination With Chemotherapy | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00539331 | Phase I/II Study of AZD2171 in Combination With Paclitaxel/Carboplatin in Japanese Non-Small Cell Lung Cancer Patients | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT03417921 | A Study of ABTL0812 in Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00002587 | Paclitaxel Plus Topotecan in Treating Patients With Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT00077246 | ABI-007 in Treating Patients With Chemotherapy-Naïve Stage IV Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT01024712 | Paclitaxel, Carboplatin, and Gefitinib in Treating Patients With Advanced Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT01239732 | Study of Intraperitoneal Carboplatin With IV Paclitaxel and Bevacizumab in Untreated Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00025389 | Bevacizumab, Paclitaxel, and Carboplatin Before Surgery in Treating Patients With Stage IB, Stage II, or Stage IIIA Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT02644863 | Autologous Tumor Tissue Antigen-sensitized DC-CIK Cells Combined With Chemotherapy for Esophageal Cancer | esophageal carcinoma | Paclitaxel (NPC208553) | |
| NCT01229930 | Carboplatin and Paclitaxel With or Without Cediranib Maleate in Treating Patients With Metastatic or Recurrent Cervical Cancer That Cannot Be Removed by Surgery | cervical cancer | Paclitaxel (NPC208553) | |
| NCT00088088 | STA-4783 in Combination With Paclitaxel and Carboplatin for the Treatment of Chemotherapy Naive Patients With Stage IIIB/IV Non Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00301730 | Cellular Adoptive Immunotherapy in Treating a Patient Who Has Undergone a Donor Stem Cell Transplant for Breast Cancer That Has Spread to the Lung | breast cancer | Paclitaxel (NPC208553) | |
| NCT01667211 | Clinical Study of Albumin-bound Paclitaxel Plus Nedaplatin in Cervical Cancer | cervical cancer | Paclitaxel (NPC208553) | |
| NCT00433420 | Combination Chemotherapy With or Without Fluorouracil and/or Pegfilgrastim in Treating Women With Node-Positive Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT01224652 | Efficacy Study of Paclitaxel Versus Irinotecan in Patients With Recurrent or Metastatic Gastric Cancer Who Progress Following First-line Therapy | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00883116 | A Study of Ixabepilone as Second-line Therapy for Locally Advanced, Recurrent, or Metastatic Endometrial Cancer | endometrial cancer | Paclitaxel (NPC208553) | |
| NCT02754726 | Combination Therapy for Patients With Untreated Metastatic Pancreatic Ductal Adenocarcinoma | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT04540211 | A Study of Atezolizumab Plus Tiragolumab in Combination With Paclitaxel and Cisplatin Compared With Paclitaxel and Cisplatin as First-Line Treatment in Participants With Unresectable Locally Advanced, Unresectable Recurrent, or Metastatic Esophageal Carcinoma | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00493025 | Paclitaxel, Cisplatin, Gefitinib, and Radiation Therapy Followed by Surgery and Gefitinib in Treating Patients With Locally Advanced Cancer of the Esophagus or Gastroesophageal Junction That Can Be Removed By Surgery | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00006454 | Paclitaxel Plus Carboplatin With or Without Topotecan in Treating Patients With Stage IIB, Stage III, or Stage IV Ovarian Epithelial Cancer | fallopian tube cancer;ovarian cancer | Paclitaxel (NPC208553) | |
| NCT01954355 | Clinical Study in Treatment of Malignant Ascites of Ovarian Cancer With Intraperitoneal Injection Bevacizumab Combined With Intraperitoneal Hyperthermic Perfusion Chemotherapy | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT04707118 | Ntraperitoneal Thermal Perfusion Combined With Chemotherapy Versus Chemotherapy | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT02004093 | EWOC-1 Trial: Carboplatin +/- Paclitaxel in Vulnerable Elderly Patients With Stage III-IV Advanced Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT02220894 | Study of Pembrolizumab (MK-3475) Versus Platinum-Based Chemotherapy for Participants With Programmed Cell Death-Ligand 1 (PD-L1)-Positive Advanced or Metastatic Non-Small Cell Lung Cancer (MK-3475-042/KEYNOTE-042) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT04296175 | Carboplatin Intensified Chemotherapy for TRIple NEgative Breast Cancer(CITRINE) | triple-negative breast cancer | Paclitaxel (NPC208553) | |
| NCT02640755 | Study of AZD6738, DNA Damage Repair/Novel Anti-cancer Agent, in Combination With Paclitaxel, in Refractory Cancer | cancer | Paclitaxel (NPC208553) | |
| NCT00954642 | A Study of MNRP1685A in Combination With Bevacizumab With or Without Paclitaxel in Patients With Locally Advanced or Metastatic Solid Tumors | cancer | Paclitaxel (NPC208553) | |
| NCT05342636 | A Study of Combination Therapies With Pembrolizumab (MK-3475) in Participants With Advanced Esophageal Cancer (MK-3475-06A) | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT03989310 | An Open-label, Phase I/II Study of the Pan-immunotherapy in Patients With Local Advanced/Metastatic Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00070564 | S0221 Adjuvant Doxorubicin, Cyclophosphamide, and Paclitaxel in Treating Patients With Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT01225523 | Perioperative Vs. Preoperative Chemotherapy With Surgery in the Squamous Carcinoma of Esophagus | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00003054 | Paclitaxel in Treating Patients With Recurrent or Persistent Cancer of the Uterus | sarcoma | Paclitaxel (NPC208553) | |
| NCT04341883 | Study of Anti-PD-1 Combined With Albumin-Bound Paclitaxel in Patients With Recurrent Cervical Cancer | cervical cancer | Paclitaxel (NPC208553) | |
| NCT04498689 | Efficacy and Safety of Camrelizumab Combined With Nab-Paclitaxel Plus Gemcitabine for Metastatic Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00003133 | Combination Chemotherapy Following Surgery in Treating Patients With Advanced Bladder Cancer | urinary bladder cancer | Paclitaxel (NPC208553) | |
| NCT01578551 | Study of Metformin Plus Paclitaxel/Carboplatin/Bevacizumab in Patients With Adenocarcinoma. | lung adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT01006252 | A Study of Tasisulam-sodium Versus Paclitaxel as Treatment for Metastatic Melanoma | melanoma | Paclitaxel (NPC208553) | |
| NCT03598270 | Platinum-based Chemotherapy With Atezolizumab and Niraparib in Patients With Recurrent Ovarian Cancer | ovarian carcinoma | Paclitaxel (NPC208553) | |
| NCT00490711 | Treatment of Paclitaxel Plus Carboplatin Followed by Gemcitabine Plus Carboplatin for Patients With Epithelial Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT01248949 | A Study to Evaluate the Safety and Antitumor Activity in Subjects With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT02776917 | Adjuvant Treatment of EC Followed by Taxane +/- Carboplatin in Triple-Negative Breast Cancer | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT00003385 | Combination Chemotherapy in Treating Patients With Untreated Ovarian, Peritoneal, or Fallopian Tube Cancer | fallopian tube cancer;ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT03329248 | QUILT-3.060: NANT Pancreatic Cancer Vaccine: Molecularly Informed Integrated Immunotherapy in Subjects With Pancreatic Cancer Who Have Progressed on or After Standard-of-care Therapy | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT02317419 | Nab-Paclitaxel as Salvage Treatment in Locally Advanced or Metastatic Gastric Cancer | cancer | Paclitaxel (NPC208553) | |
| NCT04672928 | A Phase Ib/III Clinical Study to Evaluate the Efficacy and Safety of IBI318 in Combination With Paclitaxel Versus Placebo in Combination With Paclitaxel in Patients With Small Cell Lung Cancer Who Have Failed First-line or Above Chemotherapies | small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00708812 | Paclitaxel Plus Carboplatin With or Without Endostar in Patients With Advanced Non-small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT04175912 | Testing the Combination of Pevonedistat With Chemotherapy for Bile Duct Cancer of the Liver | hepatocellular carcinoma;intrahepatic cholangiocarcinoma | Paclitaxel (NPC208553) | |
| NCT02876302 | Study Of Ruxolitinib (INCB018424) With Preoperative Chemotherapy For Triple Negative Inflammatory Breast Cancer | inflammatory breast carcinoma | Paclitaxel (NPC208553) | |
| NCT00023673 | Radiation Therapy Combined With Paclitaxel and Carboplatin in Treating Patients With Stage III Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT04825938 | Neoadjuvant Toripalimab in Combination With Carboplatin and Nab-paclitaxel in Untreated Salivary Gland Malignant Neoplasms | salivary gland neoplasm | Paclitaxel (NPC208553) | |
| NCT03169764 | QUILT-3.047: NANT Head and Neck Squamous Cell Carcinoma (HNSCC) Vaccine: Combination Immunotherapy in Subjects With HNSCC Who Have Progressed on or After Chemotherapy and PD-1/PD-L1 Therapy | head and neck squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00194779 | Combination Chemotherapy and Filgrastim Before Surgery in Treating Patients With HER2-Positive Breast Cancer That Can Be Removed By Surgery | breast cancer | Paclitaxel (NPC208553) | |
| NCT00993655 | Two Different Schedules of Carboplatin, Paclitaxel, Gemcitabine, and Surgery in Treating Patients With Newly Diagnosed Stage IIIC or Stage IV Primary Epithelial Ovarian Cancer, Fallopian Tube Cancer, or Primary Peritoneal Cancer | fallopian tube cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT01493843 | Safety and Efficacy of Carboplatin/Paclitaxel and Carboplatin/Paclitaxel/Bevacizumab With and Without Pictilisib in Previously Untreated Advanced or Recurrent Non-small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00974584 | A Study of the Safety and Pharmacology Of PI3-Kinase Inhibitor GDC-0941 In Combination With Either Paclitaxel And Carboplatin (With or Without Bevacizumab) or Pemetrexed, Cisplatin, And Bevacizumab in Patients With Advanced Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT04289792 | Split-course SBRT for Borderline Resectable and Locally Advanced Pancreatic Cancer | pancreatic neoplasm | Paclitaxel (NPC208553) | |
| NCT00003555 | Paclitaxel Plus Chemoprotection With Amifostine in Treating Patients With Recurrent or Refractory Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT00085839 | Erlotinib vs. Standard Chemotherapy in Patients With Advanced Non-small Cell Lung Cancer (NSCLC) and Eastern Cooperative Oncology Group (ECOG)Performance Status (PS) 2 | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00046514 | ABI-007 in Taxol Resistant Patients With Metastatic Breast Cancer | metastasis;breast neoplasm | Paclitaxel (NPC208553) | |
| NCT04247126 | A Study of SY 5609, a Selective CDK7 Inhibitor, in Advanced Solid Tumors | small cell lung carcinoma;pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT02492503 | Effectiveness of Chemotherapy in Metastatic or Recurrent Carcinoma Cervix | cervical carcinoma | Paclitaxel (NPC208553) | |
| NCT00589238 | Paclitaxel, Doxorubicin, and Cyclophosphamide With Or Without Carboplatin in Treating Women With Locally Advanced Breast Cancer That Can Be Removed by Surgery | breast cancer | Paclitaxel (NPC208553) | |
| NCT03064126 | RANGER™ Paclitaxel Coated Balloon vs Standard Balloon Angioplasty | peripheral arterial disease | Paclitaxel (NPC208553) | |
| NCT04581343 | A Phase 1B Study of Canakinumab, Spartalizumab, Nab-paclitaxel, and Gemcitabine in Metastatic PC Patients | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00063401 | Phase II Study in Patients With Epidermal Growth Factor Receptor (EGFR) + Advanced Stage Ovarian, Primary Peritoneal and Fallopian Tube Cancer | Fallopian Tube Carcinoma;ovarian cancer;peritoneal neoplasm | Paclitaxel (NPC208553) | |
| NCT02005315 | A Study of Vantictumab (OMP-18R5) in Combination With Nab-Paclitaxel and Gemcitabine in Previously Untreated Stage IV Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00003877 | Peripheral Stem Cell Transplantation With or Without Stromagen Following Chemotherapy in Treating Women With Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00401674 | MITO-5: Weekly Carboplatin and Paclitaxel as First Line Chemotherapy for Elderly Patients With Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT04077983 | Nab-Paclitaxel Combined With Gemcitabine Adjuvant Chemotherapy After Radical Resection of Intrahepatic Cholangiocarcinoma | cholangiocarcinoma | Paclitaxel (NPC208553) | |
| NCT02303977 | Phase II Study of Abraxane and Gemicitabine in Patients With Advanced Adenocarcinoma Non-Small Cell Lung Cancer Progressing After First-Line Platinum-Based Chemotherapy | lung adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT03981796 | A Study to Evaluate Dostarlimab Plus Carboplatin-paclitaxel Versus Placebo Plus Carboplatin-paclitaxel in Participants With Recurrent or Primary Advanced Endometrial Cancer | neoplasm | Paclitaxel (NPC208553) | |
| NCT00481078 | Vorinostat, Carboplatin, and Paclitaxel in Treating Patients With Advanced or Metastatic Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT03246074 | QUILT-3.051: NANT Ovarian Cancer Vaccine: Combination Immunotherapy in Subjects With Epithelial Ovarian Cancer Who Have Progressed on or After Standard-of-care (SoC) Therapy | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00254891 | Trial of Paclitaxel/Carboplatin + PF-3512676 vs Paclitaxel/Carboplatin Alone in Patients With Advanced Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01158144 | Concurrent Endostar, Paclitaxel/Carboplatin and Radiotherapy for Locally Advanced Non-small Cell Lung (RT0902) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01271725 | Evaluation of an Anti-cancer Immunotherapy Combined With Standard Neoadjuvant Treatment in Patients With WT1-positive Primary Invasive Breast Cancer | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT02520154 | Veliparib in Combination With Carboplatin And Weekly Paclitaxel in Japanese Subjects With Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT03056833 | A Study of Atezolizumab Versus Placebo in Combination With Paclitaxel, Carboplatin, and Bevacizumab in Participants With Newly-Diagnosed Stage III or Stage IV Ovarian, Fallopian Tube, or Primary Peritoneal Cancer | fallopian tube cancer;ovarian cancer | Paclitaxel (NPC208553) | |
| NCT04005170 | Combination of Toripalimab and Chemoradiotherapy in Esophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00278148 | Erlotinib, Paclitaxel, and Carboplatin Combined With Radiation Therapy for Stage III Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT03263741 | Exploratory Study on the Paclitaxel + S-1 + Oxaliplatin (PSOX) for Locally Advanced or Advanced Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT03503604 | A Study of Anti-VEGF Monoclonal Antibody hPV19 in Patients With Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT02135822 | Nab-paclitaxel Plus Gemcitabine in Chinese Patients With Advanced Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00003086 | Repeated Bone Marrow Transplantation in Treating Women With Advanced Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00717340 | A Phase 1b/2a, Open-Label, Multi-Center Study of Tivozanib (AV-951) in Combination With Paclitaxel in Subjects With Advanced or Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00787852 | A Study of Dasatinib With Concurrent Chemoradiation for Stage III Non-Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT03940196 | Chiauranib in Combination With Chemotherapy in Patients With Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT01218516 | A Safety and Efficacy Study of Farletuzumab in Participants With Adenocarcinoma of the Lung | lung adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT02016209 | Neoadjuvant Chemotherapy of Nanoparticle Albumin-bound Paclitaxel in Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00034177 | Safety and Efficacy of S-8184 in Treatment of Locally Advanced, Metastatic, or Recurrent TCC of the Urothelium | urogenital neoplasm | Paclitaxel (NPC208553) | |
| NCT02881125 | Paclitaxel and Nortriptyline Hydrochloride in Treating Patients With Relapsed Small Cell Carcinoma | small cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00422682 | A Study Evaluating BSI-201 in Combination With Chemotherapeutic Regimens in Subjects With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT04938583 | A Study Evaluating the Efficacy and Safety of Biomarker-Driven Therapies in Patients With Persistent or Recurrent Rare Epithelial Ovarian Tumors | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT01249443 | Paclitaxel and Carboplatin in Treating Patients With Metastatic or Recurrent Solid Tumors and HIV Infection | anus cancer;hypopharyngeal carcinoma;verrucous carcinoma;laryngeal squamous cell carcinoma;malignant epithelial tumor of ovary;oropharyngeal carcinoma;esophageal cancer;gastric cancer;HIV infection;non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02121990 | Dose Escalation Study of OMP-54F28 in Combination With Paclitaxel and Carboplatin in Patients With Recurrent Platinum-Sensitive Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT02559674 | QUILT-2.001: ALT-803 in Patients With Advanced Pancreatic Cancer in Conjunction With Gemcitabine and Nab-Paclitaxel | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00971841 | Rollover Study of Weekly Paclitaxel (BMS-181339) in Patients With Advanced or Recurrent Esophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00617409 | To Immunize Patients With Extensive Stage SCLC Combined With Chemo With or Without All Trans Retinoic Acid | small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT04510064 | PD-1 Antibody Combined With Modified FLOT Regimen in the Treatment of Unresectable Locally Advanced or Limited Metastatic Gastric Cancer | gastric adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00191854 | Gemcitabine Combinations in Metastatic Breast Cancer (MBC), 1st Line | breast cancer | Paclitaxel (NPC208553) | |
| NCT05054439 | A Clinical Study of SI-B001 in Combination With Paclitaxel in the Treatment of Recurrent and Metastatic HNSCC | head and neck squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT05007145 | PD-1 Inhibitor Combined With Neoadjuvant Chemotherapy in Subjects With Resectable Locally Advanced Thoracic Esophageal Squamous Cell Carcinoma | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00960297 | Preoperative Chemotherapy and Bevacizumab in Patients With Stage IB (>4 cm), II, or Select Stage III NSCLC | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00277043 | A Phase III Randomized Trial Assessing the Utility of a Test Dose Program With Taxanes | allergic disease | Paclitaxel (NPC208553) | |
| NCT00331630 | Abraxane and Lapatinib in Treating Patients With Stage I, Stage II, or Stage III Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00347412 | Study of NOV-002 in Combination With Chemotherapy to Treat Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT03201861 | Addition of Cisplatin to Adjuvant Chemotherapy for Early Stage Breast Cancer in High-Risk Women | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT00602797 | Vinorelbine Tartrate and Paclitaxel in Treating Older Patients With Advanced Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01091428 | Trial of Best Supportive Care and Either Cisplatin or Paclitaxel to Treat Patients With Primary Ovarian Cancer, Primary Peritoneal Cancer or Fallopian Tube Cancer and Inoperable Malignant Bowel Obstruction | fallopian tube cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT04862585 | Safely Stopping Pre-medications in Patients With Breast Cancer Who Are Receiving Paclitaxel | breast carcinoma in situ | Paclitaxel (NPC208553) | |
| NCT00193193 | Weekly Paclitaxel, Low-Dose Estramustine, and Carboplatin in the Treatment of Hormone Refractory Prostate Carcinoma | prostate cancer | Paclitaxel (NPC208553) | |
| NCT02607332 | Paclitaxel in Patients With Metastatic or Advanced Gastrointestinal Stromal Tumors (GIST) After Failure to Imatinib and Sunitinib | gastrointestinal stromal tumor | Paclitaxel (NPC208553) | |
| NCT00520975 | Bevacizumab in Treating Patients With Metastatic Breast Cancer That Overexpresses HER-2/NEU | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT00140140 | A Phase I/II Study of ABI-007 (Abraxane®, Nab®-Paclitaxel)and Vinorelbine in Patients With Stage IV (Metastatic) Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00003387 | Carboplatin, Paclitaxel, and Radiation Therapy in Treating Patients With Stage III Non-small Cell Lung Cancer That Cannot Be Removed During Surgery | lung cancer | Paclitaxel (NPC208553) | |
| NCT00716534 | Study of Carboplatin/Paclitaxel in Combination With ABT-869 in Subjects With Advanced or Metastatic Non-Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00591851 | Phase II Study of Dose-Dense Doxurubicin and Cyclophosphamide (AC) Followed By Paclitaxel With Trastuzumab in HER2/ NEU-Amplified Breast Cancer: Feasibility | breast cancer | Paclitaxel (NPC208553) | |
| NCT00553462 | Carboplatin and Paclitaxel Albumin-Stabilized Nanoparticle Formulation Followed by Radiation Therapy and Erlotinib in Treating Patients With Stage III Non-Small Cell Lung Cancer That Cannot Be Removed By Surgery | lung cancer | Paclitaxel (NPC208553) | |
| NCT00948675 | Study of Participants With Advanced Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00322452 | First Line IRESSA™ Versus Carboplatin/Paclitaxel in Asia | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02948075 | Pembrolizumab, Paclitaxel, and Carboplatin in Patients With Advanced Stage Epithelial Ovarian Cancer (EOC). | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00002984 | Combination Chemotherapy Plus Radiation Therapy in Treating Patients With Advanced or Metastatic Cancer of the Esophagus | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00520000 | Carboplatin and Paclitaxel Albumin-Stabilized Nanoparticle Formulation in Treating Patients With Advanced or Metastatic Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT01822613 | Study of Safety & Efficacy of the Combination of LJM716 & BYL719 in Patients With Previously Treated Esophageal Squamous Cell Carcinoma (ESCC) | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT01548924 | Determination of Dose of Antiangiogenic Multitargeted DOVITINIB (TKI258) Plus Paclitaxel in Patients With Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT02654119 | Cyclophosphamide, Paclitaxel, and Trastuzumab in Treating Patients With Stage I-II HER2/Neu Positive Breast Cancer After Surgery | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT04216472 | Nab-paclitaxel and Alpelisib for the Treatment of Anthracycline Refractory Triple Negative Breast Cancer With PIK3CA or PTEN Alterations | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT00479817 | Phase 2 AMG 386 in Comb. Paclitaxel for Subjects With Advanced Recurrent Epithelial Ovarian or Primary Peritoneal Cancer | fallopian tube cancer;ovarian cancer | Paclitaxel (NPC208553) | |
| NCT01688700 | Study of Nimotuzumab in Combination With Neoadjuvant Chemotherapy for Resectable Esophageal Squamous Cell Carcinoma | squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT04902261 | Tislelizumab Combined With Nab-paclitaxel and Gemcitabine for Recurrent Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00004092 | Combination Chemotherapy in Treating Patients With High-Risk Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT01834235 | QUILT-3.010: A Study of Gemcitabine and Nab-paclitaxel With or Without NPC-1C to Treat Patients With Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT02050009 | Nab-Paclitaxel and Bevacizumab in Treating Patients With Unresectable Stage IV Melanoma or Gynecological Cancers | peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT00003440 | Paclitaxel With or Without Trastuzumab in Treating Patients With or Without HER-2/Neu Breast Cancer That is Inoperable, Recurrent, or Metastatic | breast cancer | Paclitaxel (NPC208553) | |
| NCT00394082 | ABI-007 In Combination With Bevacizumab in Women With Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT01075464 | A Study of the Safety and Pharmacology of MEGF0444A in Combination With Bevacizumab With or Without Paclitaxel in Patients With Locally Advanced or Metastatic Solid Tumors | cancer | Paclitaxel (NPC208553) | |
| NCT03693677 | First Line Metastatic Pancreatic Cancer : 5FU/LV+Nal-IRI, Gemcitabine+Nab-paclitaxel or a Sequential Regimen of 2 Months 5FU/LV+Nal-IRI | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00189371 | Reinduction Chemotherapy Containing Carboplatin and Paclitaxel With or Without Epoetin Alpha in Recurrent Platinum Sensitive Ovarian Cancer, Cancer of the Fallopian Tube or Peritoneum | anemia (phenotype);ovarian cancer;peritoneal neoplasm | Paclitaxel (NPC208553) | |
| NCT00052468 | Carboplatin/Paclitaxel +/-Gemcitabine in Treating Patients With Ovarian Epithelial or Fallopian Tube Cancer | fallopian tube cancer;ovarian cancer | Paclitaxel (NPC208553) | |
| NCT04895358 | Capivasertib+Paclitaxel as First Line Treatment for Patients With Locally Advanced or Metastatic TNBC | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT05233696 | Radiotherapy in Combo With Chemo and Immunotherapy in Patients With PD-L1 Positive Metastatic TNBC | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT03143491 | Study of SOR007 Ointment for Cervical Intraepithelial Neoplasia (CIN) | cervical intraepithelial neoplasia | Paclitaxel (NPC208553) | |
| NCT00072215 | Cisplatin and Ifosfamide Combined With Either Paclitaxel or Vinblastine in Treating Men With Progressive or Recurrent Metastatic Germ Cell Tumors | testicular neoplasm | Paclitaxel (NPC208553) | |
| NCT00876395 | Everolimus in Combination With Trastuzumab and Paclitaxel in the Treatment of HER2 Positive Locally Advanced or Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00148681 | Preoperative Herceptin and Navelbine for Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00915369 | A Clinical Trial to Study the Effects of Nanoparticle Based Paclitaxel Drug, Which Does Not Contain the Solvent Cremophor, in Advanced Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT02879513 | Trial of Adjuvant Chemotherapy in Breast Cancer Patients With Pathological Partial Response and Complete Response to Neoadjuvant Chemotherapy | inflammatory breast carcinoma | Paclitaxel (NPC208553) | |
| NCT04761601 | First in Human Study of UCT-01-097 in Participants With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT00022620 | Paclitaxel in Treating Patients With Refractory or Recurrent Endometrial Cancer | endometrial cancer | Paclitaxel (NPC208553) | |
| NCT00043108 | Combination Chemotherapy, Surgery, and Radiation Therapy in Treating Patients With Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT03193918 | Study of Crenolanib With Ramucirumab and Paclitaxel for Advanced Esophagogastric Adenocarcinoma | gastric adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT01555853 | Phase I/II Study of Abraxane in Recurrent and Refractory Lymphoma | Hodgkins lymphoma;non-Hodgkins lymphoma | Paclitaxel (NPC208553) | |
| NCT00003117 | Paclitaxel With or Without Carboplatin in Treating Patients With Advanced Non-small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00107341 | Bortezomib, Paclitaxel, and Carboplatin in Treating Patients With Unresectable, Metastatic Cancer of the Esophagus or Gastroesophageal Junction | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00002837 | High-Dose Combination Chemotherapy and Peripheral Stem Cell Transplantation in Treating Patients With Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT03997123 | A Study Comparing Two Standard of Care Adjuvant Chemotherapy Regimens for Lower Risk HER-2 Positive Breast Cancer | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT00687817 | Study of Bavituximab Plus Paclitaxel and Carboplatin in Patients With Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02050009 | Metformin Hydrochloride, Carboplatin, and Paclitaxel in Treating Patients With Recurrent Ovarian, Fallopian Tube, or Primary Peritoneal Cancer | ovarian serous cystadenocarcinoma | Paclitaxel (NPC208553) | |
| NCT02125513 | Neoadjuvant Chemotherapy in Epithelial Ovarian Cancer | Fallopian Tube Carcinoma;peritoneal neoplasm | Paclitaxel (NPC208553) | |
| NCT00855764 | Late Phase II Study of Weekly Paclitaxel (BMS-181339) in Patients With Advanced or Recurrent Head and Neck Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT00397761 | Capecitabine and Paclitaxel (Albumin-Stabilized Nanoparticle Formulation) in Treating Women Undergoing Surgery for Stage II or Stage III Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT01868984 | Paclitaxel for the Treatment of Upper-Extremity Arteriovenous Access Fistula Stenosis | stricture | Paclitaxel (NPC208553) | |
| NCT00154726 | Paclitaxel-HDFL for Locally Advanced and Recurrent/Metastatic Gastric Cancers | gastric cancer | Paclitaxel (NPC208553) | |
| NCT02279732 | Phase 3 Trial in Squamous Non Small Cell Lung Cancer Subjects Comparing Ipilimumab Plus Paclitaxel and Carboplatin Versus Placebo Plus Paclitaxel and Carboplatin | lung cancer | Paclitaxel (NPC208553) | |
| NCT04888403 | Exploring the Safety and Effectiveness of Toripalimab Combined With Neoadjuvant Radiotherapy and Chemotherapy in the Locally Advanced Esophageal Squamous Cell Carcinoma | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT02241551 | Phase II Neoadjuvant Chemotheraphy (Gemcitabine and Nab-Paclitaxel vs. mFOLFIRINOX) and Sterotatic Body Radiation Therapy for Borderline Resectable Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT04634539 | Trial of First-line L-glutamine With Gemcitabine and Nab-paclitaxel in Advanced Pancreatic Cancer | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00574587 | Trial for Locally Advanced Breast Cancer Using Vorinostat Plus Chemotherapy | breast cancer | Paclitaxel (NPC208553) | |
| NCT04815408 | Batiraxcept (AVB-S6-500)/Placebo in Combination With Paclitaxel in Patients With Platinum-Resistant Recurrent Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00024375 | DHA-Paclitaxel in Treating Patients With Metastatic Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT04865289 | Pembrolizumab (MK-3475) Plus Lenvatinib (E7080/MK-7902) Versus Chemotherapy for Endometrial Carcinoma (ENGOT-en9 / MK-7902-001) - China Extension Study | endometrial neoplasm | Paclitaxel (NPC208553) | |
| NCT01998347 | Paclitaxel Liposome and Cisplatin as First-line Chemotherapy in Patients With Metastatic Esophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT02430311 | The Pharmacokinetics and Safety of Olaparib Alone and With Paclitaxel in Chinese Patients With Advanced Solid Tumour. | neoplasm | Paclitaxel (NPC208553) | |
| NCT02307227 | A Study to Evaluate the Potential Benefit of the Addition of BYL719 to Paclitaxel in the Treatment of Breast Cancer and Head-and-neck Cancer | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT00031577 | Paclitaxel Plus Radiation Therapy in Treating Children With Newly Diagnosed Brain Stem Glioma | Central Nervous System Neoplasm | Paclitaxel (NPC208553) | |
| NCT00832494 | Phase II Study of DMXAA (ASA404) in Combination With Chemotherapy in Patients With Advanced Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02341456 | Phase Ib Study AZD1775 in Combination With Carboplatin and Paclitaxel in Adult Asian Patients With Solid Tumours | neoplasm | Paclitaxel (NPC208553) | |
| NCT02199418 | Addition of Cisplatin to Neoadjuvant Therapy for T Locally Advanced Breast Cancer | inflammatory breast carcinoma | Paclitaxel (NPC208553) | |
| NCT02051751 | A Study to Evaluate the Potential Benefit of the Addition of BYL719 to Paclitaxel in the Treatment of Breast Cancer and Head-and-neck Cancer | upper aerodigestive tract neoplasm | Paclitaxel (NPC208553) | |
| NCT02762981 | Study to Evaluate Relacorilant (CORT125134) in Combination With Nab-paclitaxel in Participants With Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT00737243 | Treatment Based on Molecular Profiling Diagnosis Carcinoma of Unknown Primary Site | carcinoma | Paclitaxel (NPC208553) | |
| NCT02883062 | Carboplatin and Paclitaxel With or Without Atezolizumab Before Surgery in Treating Patients With Newly Diagnosed, Stage II-III Triple-Negative Breast Cancer | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT02358161 | Phase I/II Study of LDE225 With Gemcitabine and Nab-paclitaxel in Patients With Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT02159820 | Intraperitoneal tgDCC-E1 and Intravenous Paclitaxel in Women With Platinum-Resistant Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT04266249 | CompassHER2-pCR: Decreasing Chemotherapy for Breast Cancer Patients After Pre-surgery Chemo and Targeted Therapy | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT00003696 | Combination Chemotherapy in Treating Patients Who Have Extensive-Stage Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00049790 | Safety and Efficacy Study of rhAngiostatin Administered in Combination With Paclitaxel and Carboplatin to Patients With Non-Small-Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01121406 | BI 6727 (Volasertib) Randomised Trial in Ovarian Cancer | ovarian neoplasm | Paclitaxel (NPC208553) | |
| NCT04843098 | Study of TL117 in Patients With Recurrent or Metastatic Head and Neck Squamous Cell Carcinoma | head and neck squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT04778839 | Study of Paclitaxel Micelles for Injection in Chinese Patients With Advanced Solid Tumors. | neoplasm | Paclitaxel (NPC208553) | |
| NCT00691054 | Abraxane Therapy in Patients With Pancreatic Cancer Who Failed First-Line Gemcitabine Therapy | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00511849 | Study Of SU011248 In Combination With Paclitaxel/Carboplatin In Patients With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT02924909 | Phase II Trial of Neoadjuvant Treatment and Minimal Invasive Surgery for Esophageal and GEJ Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT02767557 | Study of Nab-Paclitaxel and Gemcitabine With or Without Tocilizumab in Pancreatic Cancer Patients | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT01253681 | Study of AMG 386 in Combination With Paclitaxel and Carboplatin in Subjects With Ovarian Cancer | carcinoma | Paclitaxel (NPC208553) | |
| NCT03126812 | Ribociclib (Ribociclib (LEE-011)) With Platinum-based Chemotherapy in Recurrent Platinum Sensitive Ovarian Cancer | fallopian tube cancer | Paclitaxel (NPC208553) | |
| NCT00007904 | Adjuvant Stage 2-3A Breast Cancer With Positive Lymph Nodes | breast cancer | Paclitaxel (NPC208553) | |
| NCT02122770 | Effects of Fluconazole and Itraconazole CYP3A-Mediated Inhibition on the Pharmacokinetics, Safety, and Tolerability of MLN4924 in Participants With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT02459457 | A Comparison of Paclitaxel-based Three Regimens Concurrent With Radiotherapy for Patients With Local Advanced Esophageal Cancer | squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00281788 | Combination Chemotherapy and Radiation Therapy in Treating Patients With Esophageal Cancer or Gastroesophageal Junction Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00445458 | A Phase 1/2 Study of HKI-272 (Neratinib) in Combination With Paclitaxel (Taxol) in Subjects With Solid Tumors and Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00080418 | Liposome Entrapped Paclitaxel Easy to Use (LEP-ETU) in Patients With Advanced Cancer | neoplasm | Paclitaxel (NPC208553) | |
| NCT01836679 | Chidamide in Combination With Carboplatin and Paclitaxel in Advanced Non-small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00491855 | Oxaliplatin and Paclitaxel Plus Bevacizumab in Advanced Peritoneal Carcinomatosis | peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT04390399 | Combination Immunotherapy Plus Standard-of-Care Chemotherapy Versus Standard-of-Care Chemotherapy for the Treatment of Locally Advanced or Metastatic Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT03942068 | Effect of Tumor Treating Fields (TTFields, 200 kHz) Concomitant With Weekly Paclitaxel for the Treatment of Recurrent Ovarian Cancer (ENGOT-ov50 / GOG-3029 / INNOVATE-3) | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00407888 | Doxorubicin Hydrochloride, Cyclophosphamide, and Filgrastim Followed By Paclitaxel Albumin-Stabilized Nanoparticle Formulation With or Without Trastuzumab in Treating Patients With Breast Cancer Previously Treated With Surgery | breast cancer | Paclitaxel (NPC208553) | |
| NCT04155671 | POF Versus FOLFOX Plus IP Paclitaxel in AGC | gastric cancer | Paclitaxel (NPC208553) | |
| NCT04528680 | Ultrasound-based Blood-brain Barrier Opening and Albumin-bound Paclitaxel and Carboplatin for Recurrent Glioblastoma | gliosarcoma | Paclitaxel (NPC208553) | |
| NCT00079430 | Paclitaxel, Bevacizumab And Adjuvant Intraperitoneal Carboplatin in Treating Patients Who Had Initial Debulking Surgery for Stage II, Stage III, or Stage IV Ovarian Epithelial, Primary Peritoneal, or Fallopian Tube Cancer | ovarian serous cystadenocarcinoma;endometrioid carcinoma;fallopian tube cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT00881816 | Dose Escalation Study of Liposomal Paclitaxel Plus Capecitabine in Chinese Patients With Advanced Gastric Carcinoma | gastric carcinoma | Paclitaxel (NPC208553) | |
| NCT04723875 | Postoperative Adjuvant Chemotherapy in Early-stage Cervical Cancer That Not Meet Criteria of Adjuvant Therapeutic According to NCCN Guideline | cervical adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT01783197 | Study of Selumetinib in Patients With Previously Treated or Untreated Advanced/Metastatic NSCLC | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01862081 | A Dose-escalation Study to Assess the Safety, Tolerability, and Pharmacokinetics of GDC-0032 in Combination With Docetaxel or With Paclitaxel in Patients With HER2-negative Locally Recurrent or Metastatic Breast Cancer or Non-small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT02622074 | Phase II Study With Trastuzumab + Paclitaxel in Locally Advanced HER2+ Tumors or Epirubicin + Taxotere in HER2- Tumors | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT03636308 | Nab-paclitaxel Plus S-1(AS) Versus Nab-paclitaxel Plus Gemcitabine(AG) in Patients With Advanced Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00915018 | Study Evaluating Neratinib Plus Paclitaxel VS Trastuzumab Plus Paclitaxel In ErbB-2 Positive Advanced Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT05394415 | Chemoradiation Plus Tislelizumab for Conversion Therapy of Locally Nonresectable ESCC | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT02023710 | Bevacizumab Combined With Carboplatin Plus Paclitaxel Chemotherapy to Treat Metastatic Mucosal Melanoma | melanoma | Paclitaxel (NPC208553) | |
| NCT01746771 | A Phase I-II Study of HM781-36B Combined With Paclitaxel and Trastuzumab in HER-2 Positive Advanced Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT04374630 | Evaluation of the Sensitivity to Different Chemotherapy Regimens in Platinum-Partial Sensitive Recurrent Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00291850 | Phase II Trial of Dose-dense Paclitaxel and Cisplatin as Neo-adjuvant Chemotherapy for Operable Stage II and IIA NSCLC | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT03085914 | A Study of Epacadostat in Combination With Pembrolizumab and Chemotherapy in Participants With Advanced or Metastatic Solid Tumors (ECHO-207/KEYNOTE-723) | neoplasm | Paclitaxel (NPC208553) | |
| NCT00031954 | Combination Chemotherapy in Treating Patients With Ovarian Epithelial, Fallopian Tube, or Peritoneal Cancer | fallopian tube cancer;ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT00198367 | Three Modalities of Treatment in Operable and Resectable Stage IIIA (T1-3, N2) Non-Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00113438 | Safety and Effectiveness of Combretastatin A-4 Phosphate Combined With Chemotherapy in Advanced Solid Tumors | cancer | Paclitaxel (NPC208553) | |
| NCT03740165 | Durvalumab Treatment in Combination With Chemotherapy and Bevacizumab, Followed by Maintenance Durvalumab, Bevacizumab and Olaparib Treatment in Advanced Ovarian Cancer Patients | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT03556839 | Platinum Chemotherapy Plus Paclitaxel With Bevacizumab and Atezolizumab in Metastatic Carcinoma of the Cervix | cervical carcinoma | Paclitaxel (NPC208553) | |
| NCT03957590 | Study of Tislelizumab (BGB-A317) Versus Placebo in Combination With Chemoradiotherapy in Participant With ESCC | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00276588 | Gemcitabine and Carboplatin Followed by Paclitaxel in Treating Patients With Stage III or Stage IV Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00506779 | Gleevec/Taxol for Patients With Uterine Papillary Serous Carcinoma | uterine cancer | Paclitaxel (NPC208553) | |
| NCT02916511 | Study of Extensive Clinical Target Volumes in Postoperative Radiotherapy Concurrent With Chemotherapy for Esophageal Squamous Cell Carcinoma | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT04572542 | Camrelizumab Combined With Apatinib Mesylate Tablets and Nab-paclitaxel in the Second-line Treatment of Advanced Gastric Cancer | gastric adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00252798 | ZD1839 (Iressa™) and Concurrent Chemo-Radiation in Patients With Locally Advanced Non Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01993810 | Comparing Photon Therapy To Proton Therapy To Treat Patients With Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00201435 | Wkly Taxol x 12 | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT01493310 | Nab-paclitaxel (Abraxane) With or Without Mifepristone in Patients With Advanced Breast Cancer | male breast carcinoma | Paclitaxel (NPC208553) | |
| NCT00002928 | Paclitaxel in Treating Patients With Recurrent or Progressive Advanced Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00113399 | Combination Chemotherapy With or Without Radiation Therapy in Treating Patients With Recurrent Head and Neck Cancer That Cannot Be Removed By Surgery | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT01352689 | CKD-828(80/2.5mg) Pharmacokinetic Study_2nd | hypertension | Paclitaxel (NPC208553) | |
| NCT01963325 | S-1 in Combination With Abraxane in Treating Cholangiocarcinoma | cholangiocarcinoma | Paclitaxel (NPC208553) | |
| NCT01702844 | Single Arm on the Tolerability of Weekly Nab-paclitaxel | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT05108870 | TheraT® Vectors (Vaccines) Combined With Chemotherapy to Treat HPV16 Head and Neck Cancers | oropharynx squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT04001569 | AZD8186 and Paclitaxel in Advanced Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT04329949 | Study of Relacorilant in Combination With Nab-Paclitaxel in Patients With Metastatic Pancreatic Ductal Adenocarcinoma | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT02608229 | BVD-523 Plus Nab-paclitaxel and Gemcitabine in Patients With Metastatic Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT01235897 | MK-2206, Paclitaxel and Trastuzumab in Treating Patients With HER2-overexpressing Solid Tumor Malignancies | cancer | Paclitaxel (NPC208553) | |
| NCT03435250 | Study of AG-270 in Participants With Advanced Solid Tumors or Lymphoma With MTAP Loss | lymphoma | Paclitaxel (NPC208553) | |
| NCT02855788 | Metronomic Chemotherapy in Advanced Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT05170438 | Lenvatinib Plus Paclitaxel for Patients With Advanced Biliary Tract Cancer Who Failed to Gemcitabine-based Treatment | biliary tract cancer | Paclitaxel (NPC208553) | |
| NCT05145569 | A Clinical Study of TQB2450 Injection Combined With Anlotinib Hydrochloride Capsules Versus Paclitaxel as Weekly Treatment of Relapsed Platinum-resistant Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT05024773 | Study of ONCOFID-P-B (PACLITAXEL-HYALURONIC ACID) | urinary bladder carcinoma | Paclitaxel (NPC208553) | |
| NCT01676259 | A Phase 2 Study of siG12D LODER in Combination With Chemotherapy in Patients With Locally Advanced Pancreatic Cancer | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT04195828 | Camrelizumab Combined With Apatinib Mesylate Tablets, Nab-paclitaxel and S-1 in the Treatment of Locally Advanced Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00861705 | Paclitaxel With or Without Carboplatin and/or Bevacizumab Followed by Doxorubicin and Cyclophosphamide in Treating Patients With Breast Cancer That Can Be Removed by Surgery | male breast carcinoma | Paclitaxel (NPC208553) | |
| NCT04338399 | The BURAN Study of Buparlisib in Patients With Recurrent or Metastatic HNSCC | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT02569242 | Study of Nivolumab in Unresectable Advanced or Recurrent Esophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT05316376 | MITO 35B: Olaparib Beyond Progression Compared to Platinum Chemotherapy After Secondary Cytoreductive Surgery in Recurrent Ovarian Cancer Patients. | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00093145 | Study of Albumin-bound Paclitaxel (Abraxane) in Combination With Carboplatin and Herceptin in Patients With Advanced Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT03410784 | A Study of VB-111 With Paclitaxel vs Paclitaxel for Treatment of Recurrent Platinum-Resistant Ovarian Cancer (OVAL) | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00075712 | Timing of Surgery and Chemotherapy in Treating Patients With Newly Diagnosed Advanced Ovarian Epithelial, Fallopian Tube, or Primary Peritoneal Cavity Cancer | fallopian tube cancer;ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT00003159 | Surgery With or Without Preoperative Chemotherapy in Treating Patients With Resectable Non-small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00866528 | Study of Pazopanib and Paclitaxel in Advanced Non-small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01010945 | Erlotinib, Gemcitabine and Nab-Paclitaxel in Advanced Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00003326 | Paclitaxel in Treating Patients With Metastatic, Recurrent, or Unresectable Cancer of the Esophagus | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00585689 | Neoadjuvant ABI-007, Carboplatin and Gemcitabine in Locally Advanced Bladder Cancer | urinary bladder cancer | Paclitaxel (NPC208553) | |
| NCT02414685 | First Line TIP in Poor Prognosis TGCTs. | germ cell tumor | Paclitaxel (NPC208553) | |
| NCT05272696 | Pembrolizumab and Induction Chemotherapy in Locally Advanced HNSCC | head and neck squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT01839487 | PEGPH20 Plus Nab-Paclitaxel Plus Gemcitabine Compared With Nab-Paclitaxel Plus Gemcitabine in Participants With Stage IV Untreated Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT03871036 | Improve Checkpoint-blockade Response in Advanced Urothelial Cancer | urothelial carcinoma | Paclitaxel (NPC208553) | |
| NCT04506138 | Camrelizumab in Combination With Neoadjuvant Chemotherapy for Resectable Thoracic Esophageal Squamous Cell Carcinoma | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00193297 | Topotecan Plus Paclitaxel and Carboplatin in the Initial Treatment of Advanced Ovarian and Primary Peritoneal Carcinoma | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00003397 | Peripheral Stem Cell Transplantation Plus Combination Chemotherapy and Monoclonal Antibody Therapy in Treating Patients With Non-Hodgkin's Lymphoma | lymphoma | Paclitaxel (NPC208553) | |
| NCT02181634 | Phase II Trial of Nab-Paclitaxel and Gemcitabine for First-Line Treatment of Patients With Cholangiocarcinoma | cholangiocarcinoma | Paclitaxel (NPC208553) | |
| NCT02487277 | PEGPH20, Gemicitabine and Nab-Paclitaxel for Pancreatic Ductal Adenocarcinoma | adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00820170 | Dasatinib In Combination With Weekly Paclitaxel For Patients With Metastatic Breast Carcinoma CA 180 194 | breast cancer | Paclitaxel (NPC208553) | |
| NCT04807673 | Pembrolizumab Plus Neoadjuvant Chemotherapy vs. Neoadjuvant Chemoradiotherapy for Locally Advanced ESCC (KEYSTONE-002) | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT02835586 | STREAMER : STent Restenosis And MEdicaments Release | ischemia | Paclitaxel (NPC208553) | |
| NCT04943653 | Intraperitoneal Paclitaxel With XELOX in Gastric Cancer With Peritoneal Metastasis | stomach neoplasm | Paclitaxel (NPC208553) | |
| NCT03719924 | Nal-iri/lv5-fu Versus Paclitaxel as Second Line Therapy in Patients With Metastatic Oesophageal Squamous Cell Carcinoma | squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT02327169 | A Study of RO6927005 Either As Monotherapy (Part A) or in Combination With Gemcitabine and Nab-Paclitaxel (Part B) to Evaluate the Safety, Tolerability, Pharmacokinetics, and Preliminary Clinical Activity in Patients With Mesothelin-positive Metastatic and/or Locally Advanced Malignant Solid Tumors | cancer | Paclitaxel (NPC208553) | |
| NCT00110084 | ABI-007 (Nab-Paclitaxel) and Gemcitabine in Treating Women With Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT04054362 | Paricalcitol Addition to Chemotherapy in Patients With Previously Untreated Metastatic Pancreatic Ductal Adenocarcinoma | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT04046575 | Radiation Dose Intensification With Accelerated Hypofractionated Intensity Modulated Radiation Therapy and Concurrent Carboplatin and Paclitaxel for Inoperable Esophageal Cancer | esophageal carcinoma | Paclitaxel (NPC208553) | |
| NCT01646762 | Paclitaxel Albumin-Stabilized Nanoparticle Formulation in Treating Patients With Relapsed or Refractory Multiple Myeloma | multiple myeloma | Paclitaxel (NPC208553) | |
| NCT00034541 | Study of Cetuximab in Combination With Carboplatin-Paclitaxel in Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00269828 | A Survival Study for Women With Advanced Lung Cancer Who Have Not Previously Received Chemotherapy. | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT03272477 | Study of Cirmtuzumab and Paclitaxel for Metastatic or Locally Advanced, Unresectable Breast Cancer | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT02975141 | Afatinib and Gemcitabine/Nab-paclitaxel in Metastatic Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT04267549 | Conversion Therapy of Sintilimab in Combination With Apatinib and Chemotherapy in Unresectable Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT01455532 | A Dose Escalation Study of Iniparib as a Single Agent and in Combination in Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT00483301 | A Phase II Trial OF Carboplatin, ABI-007 (Abraxane) And Sorafenib (BAY 43-9006) in Metastatic Melanoma | melanoma | Paclitaxel (NPC208553) | |
| NCT00003065 | Topotecan and Paclitaxel in Treating Patients With Recurrent or Metastatic Cancer of the Cervix | cervical cancer | Paclitaxel (NPC208553) | |
| NCT01031212 | ASA404 in Combination With Carboplatin/Paclitaxel/Cetuximab in Treating Patients With Refractory Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT00687687 | Evaluation of Paclitaxel (Taxol, NSC | Uterine Carcinosarcoma | Paclitaxel (NPC208553) | |
| NCT03534713 | Induction Chemotherapy Followed by Standard Therapy in Cervical Cancer With Aortic Lymph Node Spread | cervical cancer | Paclitaxel (NPC208553) | |
| NCT02215447 | A Feasibility Study Of NAB-Paclitaxel In Combination With Carboplatin As First Line Treatment Of Gastrointestinal Neuroendocrine Carcinomas | neuroendocrine carcinoma | Paclitaxel (NPC208553) | |
| NCT01336062 | Trial of ABI-007 Plus S-1 as Second-line Chemotherapy in Advanced Gastric Cancer Patients | gastric adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00580333 | Preoperative Cisplatin and Bevacizumab in ER-, PR-, HER2 Negative Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00356681 | A Study of AMG 706 or Bevacizumab, in Combination With Paclitaxel Chemotherapy, as Treatment for Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00003214 | Chemosensitivity Testing to Assign Treatment for Patients With Stage III or Stage IV Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT02697838 | Clinical Trial of Apatinib Reverses Chemotherapy-Resistance of Patients With Advanced Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT02383212 | Phase I Study of Oral BAY 1217389 in Combination With Intravenous Paclitaxel | cancer | Paclitaxel (NPC208553) | |
| NCT00002826 | Drug Resistance Inhibition in Treating Women With Recurrent or Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT02024009 | Systemic Therapy and Chemoradiation in Advanced Localised Pancreatic Cancer - 2 | pancreatic neoplasm | Paclitaxel (NPC208553) | |
| NCT00062179 | Paclitaxel and Carboplatin With or Without Celecoxib Before Surgery in Treating Patients With Stage IIIA Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00088556 | Carboplatin, Paclitaxel and TLK286 (Telcyta) as First-Line Therapy in Advanced Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT03872791 | A Study of KN046 in Subjects With Locally Advanced or Metastatic Triple-negative Breast Cancer | triple-negative breast cancer | Paclitaxel (NPC208553) | |
| NCT03742102 | 9-ING-41 in Patients With Advanced Cancers | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT03512834 | Paclitaxel-Avelumab for Angiosarcoma | angiosarcoma | Paclitaxel (NPC208553) | |
| NCT02574663 | TGR-1202 Alone and in Combination With Either Nab-paclitaxel + Gemcitabine or With FOLFOX in Patients With Select Relapsed or Refractory Solid Tumors | rectum cancer;colorectal carcinoma;gastrointestinal stromal tumor;esophageal cancer;gastric cancer;pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT02273713 | The Addition of Nab-paclitaxel (Abraxane) to First Line Treatment of Metastasized Oesophagogastric Carcinoma (ACTION) | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00046423 | A Trial of ABI-007 in Patients With Advanced Non-Hematologic Malignancies | metastasis;neoplasm | Paclitaxel (NPC208553) | |
| NCT04469556 | Pancreatic Adenocarcinoma Signature Stratification for Treatment | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT04475016 | TIP (Paclitaxel + Ifosfamide + Cisplatin) Combined With Nimotuzumab & Triprilimab as Neoadjuvant Treatment in Locally Advanced Penile Cancer | penile cancer | Paclitaxel (NPC208553) | |
| NCT03486314 | A Study to Evaluate the Effects of Rifampin on Pharmacokinetics (PK) of Pevonedistat in Participants With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT04643405 | APG-1387 Plus Chemotherapy in Advanced Pancreatic Adenocarcinoma | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00152477 | A Study of Paclitaxel/Carboplatin With or Without CDP791 in Patients With Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT03668730 | Reduced-dose Radiotherapy for Low-risk Stage III Patients With Nasopharyngeal Carcinoma | nasopharyngeal neoplasm | Paclitaxel (NPC208553) | |
| NCT00001450 | Phase II Trial of a 96-Hour Continuous Infusion of Paclitaxel Followed by Cisplatin for Patients With Stage III/IV and Relapsed NSCLC | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT03694002 | Carboplatin and Paclitaxel With or Without Ramucirumab in Treating Patients With Locally Advanced, Recurrent, or Metastatic Thymic Cancer That Cannot Be Removed by Surgery | Thymic Carcinoma | Paclitaxel (NPC208553) | |
| NCT01579578 | Assess the Efficacy of AZD8931 in Combination With Paclitaxel Versus Paclitaxel Alone in Patients With Gastric Cancer | cancer | Paclitaxel (NPC208553) | |
| NCT02429622 | Simultaneous Integrated Boost Radiotherapy and Concurrent Chemotherapy for Locally Advanced Esophageal Carcinoma | esophageal carcinoma | Paclitaxel (NPC208553) | |
| NCT01934634 | Phase I Trial of LCL161 and Gemcitabine Plus Nab-Paclitaxel in Metastatic Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT04979585 | Camrelizumab Plus Amlotinib and Chemotherapy in the First-line Treatment of Melanoma | melanoma | Paclitaxel (NPC208553) | |
| NCT00268450 | Cisplatin, Bevacizumab, and Gemcitabine Followed by Surgery, Bevacizumab, and Paclitaxel in Treating Patients With Locally Advanced Nonmetastatic Bladder Cancer That Can Be Removed By Surgery | urinary bladder cancer | Paclitaxel (NPC208553) | |
| NCT03101748 | Neratinib and Paclitaxel With or Without Pertuzumab and Trastuzumab Before Combination Chemotherapy in Treating Patients With Metastatic or Locally Advanced Breast Cancer | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT00568451 | Paclitaxel and Carboplatin or Temozolomide in Treating Patients With Stage IV Melanoma | cutaneous melanoma | Paclitaxel (NPC208553) | |
| NCT00966914 | Phase 3 Study of Tavocept Versus Placebo in Patients With Newly Diagnosed or Relapsed Advanced Primary Adenocarcinoma of the Lung Treated With Docetaxel or Paclitaxel Plus Cisplatin | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01701349 | Fosbretabulin or Placebo in Combination With Carboplatin/Paclitaxel in Anaplastic Thyroid Cancer | thyroid cancer | Paclitaxel (NPC208553) | |
| NCT01246193 | CKD-828(80/2.5mg) Pharmacokinetic Study | hypertension | Paclitaxel (NPC208553) | |
| NCT05235542 | A Study to Investigate Safety and Tolerability of TransCon IL-2 β/γ Alone or in Combination With Pembrolizumab and/or Chemotherapy or TransCon TLR7/8 Agonist in Adult Participants With Locally Advanced or Metastatic Solid Tumor Malignancies | cancer | Paclitaxel (NPC208553) | |
| NCT02075021 | Phase I/II Trial of the Combination of Lenalidomide (Revlimid) and Nab-paclitaxel (Abraxane) in the Treatment of Relapsed/Refractory Multiple Myeloma | multiple myeloma | Paclitaxel (NPC208553) | |
| NCT00777673 | Preoperative Chemotherapy in Triple Negative Invasive Breast Cancer That Can be Removed by Surgery. | breast cancer | Paclitaxel (NPC208553) | |
| NCT00006929 | Suramin, Paclitaxel, and Carboplatin in Treating Patients With Stage IIIB or Stage IV Non-small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02630199 | A Study of BBI503 in Combination With Selected Anti-Cancer Therapeutics in Adult Patients With Advanced Cancer | cancer | Paclitaxel (NPC208553) | |
| NCT01519804 | A Study of Onartuzumab (MetMAb) Versus Placebo in Combination With Paclitaxel Plus Platinum in Patients With Squamous Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00003013 | Chemotherapy Plus Surgery in Treating Women With Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00331422 | Carboplatin, Paclitaxel, and Surgery in Treating Patients With Advanced Ovarian Epithelial, Fallopian Tube, or Primary Peritoneal Cavity Cancer | fallopian tube cancer;ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT03975270 | Sintilimab in Combination With Nab-paclitaxel in Patients With HNSCC (Head and Neck Squamous Cell Carcinoma ) | squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT04261465 | Adoptive T Cell Therapy in Patients With Recurrent Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00301028 | Cetuximab, Carboplatin, and Paclitaxel Followed by Radiation Therapy, With or Without Cisplatin, in Treating Patients With Metastatic Head and Neck Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT00005581 | Combination Chemotherapy in Treating Women With Stage II or Stage III Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT04209686 | Paclitaxel, Pembrolizumab and Olaparib in Previously Treated Advanced Gastric Adenocarcinoma | gastric adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT01008150 | Phase II Randomized Trial Evaluating Neoadjuvant Therapy With Neratinib and/or Trastuzumab Followed by Postoperative Trastuzumab in Women With Locally Advanced HER2-positive Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT04210037 | Study of APG-1252 Plus Paclitaxel in Patients With Relapsed/Refractory Small Cell Lung Cancer | small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00085358 | Carboplatin and Paclitaxel With or Without Bevacizumab Compared to Docetaxel, Carboplatin, and Paclitaxel in Treating Patients With Stage II, Stage III, or Stage IV Ovarian Epithelial, Fallopian Tube, or Primary Peritoneal Cavity Carcinoma (Cancer) | ovarian serous cystadenocarcinoma;endometrioid carcinoma;fallopian tube cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT02107378 | Efficacy of DCVAC/OvCa Plus Standard of Care in Relapsed Platinum Resistant Epithelial Ovarian Carcinoma | ovarian carcinoma | Paclitaxel (NPC208553) | |
| NCT02134067 | Dose-escalating, Safety, Tolerability and PK Study of TAS-119 in Combination With Paclitaxel in Patients With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT02730481 | A Study to Evaluate the Safety, Tolerability, MTD, PK, and Activity of Oraxol in Subjects w Adv. Malignancies | neoplasm | Paclitaxel (NPC208553) | |
| NCT03776812 | A Phase 1b Study of Sevacizumab in Combination With Chemotherapy in Chinese Patients With Platinum-Resistant Recurrent Ovarian Cancer. | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT01003158 | Study Assessing Safety and Tolerability of AZD8931 Alone or in Combination With Paclitaxel in Japanese Patients. | breast cancer | Paclitaxel (NPC208553) | |
| NCT04931342 | Chiauranib Plus Weekly Paclitaxel in Patients With Platinum-refractory or Platinum-resistant Recurrent Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT01953536 | Effect of Trastuzumab on Disease Free Survival in Early Stage HER2-Negative Breast Cancer Patients With ERBB2 Expressing Disseminated Tumor Cells | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT00006245 | Flavopiridol and Paclitaxel in Treating Patients With Locally Advanced or Metastatic Esophageal Cancer That Has Not Responded to Previous Paclitaxel | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00002922 | Paclitaxel in Treating Patients With Advanced Head and Neck Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT00003299 | Cisplatin Plus Etoposide With or Without Paclitaxel in Treating Patients With Extensive-Stage Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT03462212 | Study of GEN-1 With NACT for Treatment of Ovarian Cancer (OVATION 2) | fallopian tube cancer | Paclitaxel (NPC208553) | |
| NCT01126216 | Reduced Radiotherapy With Pac/Cis vs Standard Radiotherapy With 5-FU/Cis in Locally Advanced Head and Neck Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT03038100 | A Study of Atezolizumab Versus Placebo in Combination With Paclitaxel, Carboplatin, and Bevacizumab in Participants With Newly-Diagnosed Stage III or Stage IV Ovarian, Fallopian Tube, or Primary Peritoneal Cancer | peritoneal neoplasm | Paclitaxel (NPC208553) | |
| NCT00404235 | Carboplatin and ABI-007 in Treating Patients With Stage IV Melanoma That Cannot Be Removed By Surgery | cutaneous melanoma | Paclitaxel (NPC208553) | |
| NCT00004125 | Combination Chemotherapy in Treating Women With Stage II or Stage IIIA Breast Cancer That Has Spread to the Lymph Nodes | breast cancer | Paclitaxel (NPC208553) | |
| NCT04492033 | A Study of CTX-009 (ABL001) in Combination With Irinotecan or Paclitaxel in Advanced or Metastatic Solid Tumor Patients | biliary tract cancer | Paclitaxel (NPC208553) | |
| NCT03850769 | Neoadjuvant Nab-Paclitaxel and S-1 in Borderline Resectable Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT02926183 | Study of NAC of GA Therapy for Patients With BRPC | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT02715804 | A Study of PEGylated Recombinant Human Hyaluronidase in Combination With Nab-Paclitaxel Plus Gemcitabine Compared With Placebo Plus Nab-Paclitaxel and Gemcitabine in Participants With Hyaluronan-High Stage IV Previously Untreated Pancreatic Ductal Adenocarcinoma | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00146562 | Pegfilgrastim and Darbepoetin Alfa in Support of Adjuvant Chemotherapy for Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT01570582 | TRINOVA-3: A Study of AMG 386 or AMG 386 Placebo in Combination With Paclitaxel and Carboplatin to Treat Ovarian Cancer | fallopian tube cancer;ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00100789 | S0329, Gemcitabine and Paclitaxel in Treating Patients With Persistent, Recurrent, or Metastatic Head and Neck Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT03691090 | Study of SHR-1210 in Combination With Chemotherapy in Advanced Esophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT01730222 | A Phase I-II Study of PAXG in Stage III-IV Pancreatic Adenocarcinoma | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT01486602 | Specialized Radiation Therapy and Chemotherapy in Treating Patients With Stage III Non-Small Cell Lung Cancer That Cannot Be Removed by Surgery | lung cancer | Paclitaxel (NPC208553) | |
| NCT00096226 | Paclitaxel, Carboplatin, and Radiation Therapy in Treating Patients Who Are Undergoing Surgery for Stage III Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT04527991 | Study of Sacituzumab Govitecan-hziy (IMMU-132) Versus Treatment of Physician's Choice in Participants With Metastatic or Locally Advanced Unresectable Urothelial Cancer | urogenital neoplasm | Paclitaxel (NPC208553) | |
| NCT01011075 | Study of Gleevec and Weekly Paclitaxel in Patients Aged 70 or Older With Advanced Non-small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT04807140 | Neoadjuvant Toripalimab or Toripalimab in Combination With Carboplatin and Nab-paclitaxel in Untreated HNSCC (HNSCC-002) | head and neck squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT02395705 | Neoadjuvant Chemotherapy Versus Surgery Alone for Esophageal Squamous Cell Carcinoma | neoplasm of esophagus | Paclitaxel (NPC208553) | |
| NCT02182232 | A Dose Escalation Study of BIBF 1120 Together With Paclitaxel and Carboplatin in Patients With Advanced Stage Non-small-cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00479765 | A Phase 1 / 2 Dose Escalation Study of Locally-Administered OncoGel™ in Subjects With Recurrent Glioma | brain neoplasm;glioblastoma multiforme | Paclitaxel (NPC208553) | |
| NCT00255762 | Carboplatin, Paclitaxel, and Bevacizumab in Treating Patients With Stage IV Melanoma That Cannot Be Removed By Surgery | melanoma | Paclitaxel (NPC208553) | |
| NCT00507429 | Study of Combretastatin and Paclitaxel/Carboplatin in the Treatment of Anaplastic Thyroid Cancer | thyroid cancer | Paclitaxel (NPC208553) | |
| NCT00042835 | Erlotinib and Radiation Therapy Plus Combination Chemotherapy in Treating Patients With Inoperable Stage III Non-Small Cell Lung Cancer | lung adenocarcinoma;squamous cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT04859465 | Albumin-bound Paclitaxel Combined With Liposomal Doxorubicin in the Treatment of Advanced or Unresectable Angiosarcoma | angiosarcoma | Paclitaxel (NPC208553) | |
| NCT04718415 | Neoadjuvant Sintilimab in Combination With Carboplatin and Nab-paclitaxel in Resectable Oral Cavity or Oropharyngeal Squamous Cell Carcinoma | oral squamous cell carcinoma;oropharynx squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00455533 | Study to Assess Effectiveness of Giving Combination of Standard Chemotherapy Drugs Versus Combination of Standard Chemotherapy and New Drug Ixabepilone When Given Before Surgical Removal of Early Stage Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT02072317 | Paclitaxel Plus Raltitrexed Plug Compare With Taxol Second-line Treatment for Advanced Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00129389 | FAC Versus FAC Plus Weekly Paclitaxel as Adjuvant Treatment of Node Negative High Risk Breast Cancer Patients | breast cancer | Paclitaxel (NPC208553) | |
| NCT02391662 | Nab-paclitaxel and Gemcitabine, in Elderly Patients Untreated Metastatic Pancreatic Adenocarcinoma | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00021320 | Combination Chemotherapy Plus Radiation Therapy in Treating Patients With Esophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT05247619 | A Clinical Study to Explore the Efficacy and Safety of Tislelizumab in Combination With Bevacizumab and Chemotherapy in Patients With Persistent, Recurrent, or Metastatic Cervical Cancer | cervical cancer | Paclitaxel (NPC208553) | |
| NCT00193141 | Chemotherapy With or Without Surgical Resection in Locally Advanced Esophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT02163291 | Study on Neoadjuvant Chemotherapy for Advanced Gastric Cancer | gastric carcinoma | Paclitaxel (NPC208553) | |
| NCT00532285 | Primary Paclitaxel Plus Gemcitabine in Patients With Stage II and III Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00629499 | Nanoparticle Albumin-Bound (Nab) Paclitaxel/Cyclophosphamide in Early-Stage Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT01980810 | First Line Chemotherapy for Advanced Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00002628 | Addition of Paclitaxel to High-Dose Combination Chemotherapy in Treating Women With Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT05204173 | Efficacy and Safety of Sintilimab Combined Intraperitoneal and Intravenous Paclitaxel Plus Oral S-1 in Gastric Cancer Patients With Peritoneal Metastasis | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00661167 | Phase II Study of ABI-007 for Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00429299 | Pazopanib (VOTRIENT) Plus Paclitaxel (TAXOL), Pazopanib Plus Paclitaxel (TAXOL) Plus Carboplatin (PARAPLATIN), and Pazopanib Plus Paclitaxel (TAXOL) Plus Lapatinib (TYKERB) | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT02446574 | Postoperative Chemoradiation in Patients With Node-positive Esophageal Squamous Cell Carcinoma | esophageal carcinoma | Paclitaxel (NPC208553) | |
| NCT02043730 | Abraxane and Gemcitabine Versus Gemcitabine Alone in Locally Advanced Unresectable Pancreatic Cancer. | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT04138212 | Neoadjuvant Chemotherapy Versus Neoadjuvant Chemoradiotherapy for Resectable Locally Advanced Esophageal Cancer (HCHTOG1903) | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT05319639 | Phase I Study of the Combination of Irinotecan and POF (POFI) and Tislelizumab | gastric cancer | Paclitaxel (NPC208553) | |
| NCT03396445 | Study to Compare Neoadjuvant Combination of Trastuzumab and Pertuzumab With Concurrent Taxane Chemotherapy or Endocrine Therapy and Quality of Life Assessment Under Adjuvant Therapy in Operable HER2+/HR+ Breast Cancer Patients | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT00337532 | A Phase II Trial Neoadjuvant Paclitaxel and Cisplatin in Patients With Locally Advanced Head and Neck Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT05173987 | Study of Pembrolizumab (MK-3475) Versus Chemotherapy in Mismatch Repair Deficient (dMMR) Advanced or Recurrent Endometrial Carcinoma (MK-3475-C93/KEYNOTE-C93/GOG-3064/ENGOT-en15) | endometrial neoplasm | Paclitaxel (NPC208553) | |
| NCT00515073 | Papillary Serous Carcinoma of the Endometrium | endometrial cancer | Paclitaxel (NPC208553) | |
| NCT00459121 | Vandetanib, Carboplatin, and Paclitaxel in Treating Patients With Stage I, Stage II, or Stage III Non-Small Cell Lung Cancer That Can Be Removed by Surgery | lung cancer | Paclitaxel (NPC208553) | |
| NCT04808687 | Neoadjuvant Nab-Paclitaxel and S-1 in Resectable Pancreatic Cancer | pancreatic neoplasm | Paclitaxel (NPC208553) | |
| NCT03907475 | Durvalumab in Combination With Chemotherapy in Treating Patients With Advanced Solid Tumors, (DURVA+ Study) | neoplasm | Paclitaxel (NPC208553) | |
| NCT00088530 | BBR 2778 for Relapsed, Aggressive Non-Hodgkin's Lymphoma (NHL) | non-Hodgkins lymphoma | Paclitaxel (NPC208553) | |
| NCT00954512 | Study of Robatumumab (SCH 717454, MK-7454) in Combination With Different Treatment Regimens in Participants With Advanced Solid Tumors (P04722, MK-7454-004) | neoplasm | Paclitaxel (NPC208553) | |
| NCT00360360 | Paclitaxel/Carboplatin Plus Bevacizumab/Erlotinib in the First Line Treatment of Carcinoma of Unknown Primary Site | neoplasm | Paclitaxel (NPC208553) | |
| NCT00344552 | Phase II Study of Weekly Paclitaxel (BMS-181339)in Patients With Advanced or Recurrent Esophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00004887 | Paclitaxel and Carboplatin Chemotherapy Compared With Standard Chemotherapy in Treating Patients With Stage III or Stage IV Non-small Cell Lung Cancer That Cannot Be Removed During Surgery | lung cancer | Paclitaxel (NPC208553) | |
| NCT00503906 | Abraxane, Avastin, and Gemcitabine as First-Line Therapy for Patients With Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00003242 | Bryostatin 1 Plus Paclitaxel and Cisplatin in Treating Patients With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT04151277 | PRIMUS 001: A Study Looking at Two Different Chemotherapy Regimens in Patients With Metastatic Pancreatic Cancer | neoplasm | Paclitaxel (NPC208553) | |
| NCT00915005 | Trial of Image-Guided Adaptive Conformal Photon vs Proton Therapy, With Concurrent Chemotherapy, for Locally Advanced Non-Small Cell Lung Carcinoma: Treatment Related Pneumonitis and Locoregional Recurrence | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT04354961 | Almonertinib Versus Paclitaxel Plus Carboplatin as First-line Treatment in Patients With EGFR Mutation Positive Locally Advanced or Metastatic Pulmonary Adenosquamous Carcinoma (ARISE) | squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00006256 | Paclitaxel Plus Radiation Therapy in Treating Women With Stage II or Stage III Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00003230 | Paclitaxel in Treating Patients With Refractory or Recurrent Acute Leukemia or Chronic Myelogenous Leukemia | leukemia | Paclitaxel (NPC208553) | |
| NCT02449655 | Trial of AZD5363 Plus Paclitaxel /AZD2014 Plus Paclitaxel in Biomarker Negative (PIK3CA/MEK/RAS/TP53/MET) Gastric Adenocarcinoma Patients as Second-line Chemotherapy | gastric adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT01253525 | Study of Weekly Paclitaxel With Ramucirumab in Participants With Advanced Gastric Adenocarcinomas | adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00004160 | Chemotherapy Plus Radiation Therapy in Treating Patients With Stage III Non-small Cell Lung Cancer That Cannot Be Surgically Removed | lung cancer | Paclitaxel (NPC208553) | |
| NCT00006276 | Micellar Paclitaxel to Treat Severe Psoriasis | psoriasis | Paclitaxel (NPC208553) | |
| NCT01704287 | Study of Pembrolizumab (MK-3475) Versus Chemotherapy in Participants With Advanced Melanoma (MK-3475-002/P08719/KEYNOTE-002) | melanoma | Paclitaxel (NPC208553) | |
| NCT00003087 | Combination Chemotherapy, Radiation Therapy and Surgery in Treating Patients With Cancer of the Esophagus | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT05185869 | SHR6390 Plus Nab-paclitaxel and Gemcitabine in Advanced/Metastatic Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT03423979 | Optilume™ BPH Prostatic Drug Coated Balloon Dilation Catheter | benign prostatic hyperplasia | Paclitaxel (NPC208553) | |
| NCT01009983 | Paclitaxel, Carboplatin, and Panitumumab in Treating Patients With Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00004865 | Cetuximab Plus Cisplatin in Treating Patients With Metastatic or Recurrent Cancer of the Head and Neck That Has Not Responded to Cisplatin Chemotherapy | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT04672005 | Modified FOLFIRINOX Alternated With Biweekly Gemcitabine Plus Nab-Paclitaxel Untreated Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00226590 | Induction Gemcitabine & Carboplatin Followed by Paclitaxel & Carboplatin +XRT in NSCLC | lung cancer | Paclitaxel (NPC208553) | |
| NCT03136406 | QUILT-3.039: NANT Pancreatic Cancer Vaccine: Combination Immunotherapy in Subjects With Pancreatic Cancer Who Have Progressed on or After Standard-of-care Therapy | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00003624 | Amifostine in Treating Women With Ovarian, Peritoneal, Cervical, Fallopian Tube, Uterine, or Endometrial Cancer | cervical cancer;endometrial cancer;fallopian tube cancer;ovarian cancer;peritoneum cancer;sarcoma | Paclitaxel (NPC208553) | |
| NCT00322400 | Phase1b to Evaluate Safety of AMG706 in Combination With Paclitaxel or Docetaxel for Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT03306121 | TPF+CCRT vs.CCRT+PF in High Risk Nasopharyngeal Carcinoma | nasopharyngeal neoplasm | Paclitaxel (NPC208553) | |
| NCT02477826 | An Investigational Immuno-therapy Trial of Nivolumab, or Nivolumab Plus Ipilimumab, or Nivolumab Plus Platinum-doublet Chemotherapy, Compared to Platinum Doublet Chemotherapy in Patients With Stage IV Non-Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT04143711 | Study of DF1001 in Patients With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT00738699 | First Line Tx Stage III Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT02300935 | Study of Trametinib and Nab-paclitaxel in Patients With Melanoma | melanoma | Paclitaxel (NPC208553) | |
| NCT02279134 | Phase II/III Study Compare Adjuvant Chemoradiotherapy, Radiotherapy and Surgery Alone for Esophageal Carcinoma | esophageal carcinoma | Paclitaxel (NPC208553) | |
| NCT02419495 | Selinexor With Multiple Standard Chemotherapy or Immunotherapy Regimens in Treating Patients With Advanced Malignancies | renal cell carcinoma;cutaneous melanoma;Fallopian Tube Carcinoma;metastatic melanoma;ovarian carcinoma;breast carcinoma;non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT04056949 | Efficacy and Safety of IBI308 and Paclitaxel/Albumin Paclitaxel for SCLC Patients Who Failed Etoposide Chemotherapy | small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02732938 | Ph1b/2 Study of PF-04136309 in Combination With Gem/Nab-P in First-line Metastatic Pancreatic Patients | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00003558 | Chemotherapy in Treating Patients With Cancer of Unknown Primary Origin | carcinoma | Paclitaxel (NPC208553) | |
| NCT03387111 | QUILT-3.090: NANT Squamous Cell Carcinoma (SCC) Vaccine: Subjects With SCC Who Have Progressed | squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT02705196 | LOAd703 Oncolytic Virus Therapy for Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT01654146 | Dose-finding Study in Platinum-Resistant Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT03776812 | Study of Relacorilant in Combination With Nab-Paclitaxel for Patients With Recurrent Platinum-Resistant Ovarian, Fallopian Tube, or Primary Peritoneal Cancer | Fallopian Tube Carcinoma | Paclitaxel (NPC208553) | |
| NCT00088114 | STA-4783 and Paclitaxel for Treatment of Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT02064491 | Erlotinib Treatment Beyond Progression in EGFR Mutant NSCLC | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01457846 | Efficacy and Safety of AZD4547 Versus Paclitaxel in Patients With Advanced Gastric or Gastro-oesophageal Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT03273595 | Study of Pembrolizumab (MK-3475) Plus Chemotherapy vs Placebo Plus Chemotherapy as Neoadjuvant Therapy and Pembrolizumab vs Placebo as Adjuvant Therapy in Participants With Triple Negative Breast Cancer (TNBC) (MK-3475-522/KEYNOTE-522) | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT00930930 | Cisplatin and Paclitaxel With or Without Everolimus in Treating Patients With Stage II or Stage III Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00801736 | ERCC1 Targeted Trial | lung cancer | Paclitaxel (NPC208553) | |
| NCT00748163 | Paclitaxel Albumin-Stabilized Nanoparticle Formulation and Sunitinib as First-Line Therapy in Treating Patients With Stage IV Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT03057366 | A Study of [14 C]-Pevonedistat in Participants With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT00196872 | A Study to Compare ETC vs. EC-TX and Ibandronate vs. Observation in Patients With Node-positive Primary Breast Cancer (GAIN) | breast cancer | Paclitaxel (NPC208553) | |
| NCT00838656 | First Line Tx Stage III Ovarian Cancer | fallopian tube cancer | Paclitaxel (NPC208553) | |
| NCT01238133 | Gamma-Secretase/Notch Signalling Pathway Inhibitor RO4929097, Paclitaxel, and Carboplatin Before Surgery in Treating Patients With Stage II or Stage III Triple-Negative Breast Cancer | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT01621243 | M402 in Combination With Nab-Paclitaxel and Gemcitabine in Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00003998 | Carboplatin Plus Paclitaxel or Docetaxel in Treating Patients With Ovarian Epithelial Cancer | ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT00919984 | Efficacy Study of Additional Intraperitoneal Chemotherapy to Treat Ovarian Cancer | ovarian neoplasm | Paclitaxel (NPC208553) | |
| NCT00028873 | R101933 Combined With Chemotherapy in Treating Patients With Metastatic Breast Cancer That Has Not Responded to Previous Chemotherapy | breast cancer | Paclitaxel (NPC208553) | |
| NCT00107094 | Study of Adriamycin Plus Cyclophosphamide Followed by Abraxane as Adjuvant Therapy for Patients With Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT02891083 | Adjuvant Therapies or Surgery Alone for High Risk pN0 Esophageal Cancer | esophageal carcinoma | Paclitaxel (NPC208553) | |
| NCT02630264 | E10A for the Treatment of Squamous Cell Carcinoma of the Head and Neck | upper aerodigestive tract neoplasm | Paclitaxel (NPC208553) | |
| NCT02050178 | Dose Escalation Study of OMP-54F28 in Combination With Nab-Paclitaxel and Gemcitabine in Patients With Previously Untreated Stage IV Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT01839773 | A Phase 3 Study to Compare Efficacy and Safety of DHP107 Versus Taxol® in Patients With Metastatic or Recurrent Gastric Cancer After Failure of First-line Chemotherapy | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00991796 | CS-1008 With Carboplatin/Paclitaxel in Chemotherapy naïve Subjects With Metastatic or Unresectable Non-small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00055133 | A Study Using Intravenous Paxceed™ to Treat Patients With Rheumatoid Arthritis | rheumatoid arthritis | Paclitaxel (NPC208553) | |
| NCT03165994 | APX005M With Concurrent Chemoradiation for Resectable Esophageal and Gastroesophageal Junction Cancers | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00005644 | Combination Chemotherapy in Treating Patients With Advanced Cancer of the Urothelium and Decreased Kidney Function | urethra cancer;urinary bladder cancer | Paclitaxel (NPC208553) | |
| NCT03678883 | 9-ING-41 in Patients With Advanced Cancers | Malignant Bone Neoplasm;malignant glioma;kidney cancer;lung neoplasm;metastasis;sarcoma | Paclitaxel (NPC208553) | |
| NCT00245154 | Paclitaxel + Carboplatin With/Out Cediranib Maleate in Stage III or Stage IV Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT05319730 | A Study to Evaluate Pembrolizumab (MK-3475) in Participants With Advanced Esophageal Cancer Previously Exposed to Programmed Cell Death 1 Protein (PD-1)/ Programmed Cell Death Ligand 1 (PD-L1) Treatment | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT01750073 | Paclitaxel and Cyclophosphamide With or Without Trastuzumab Before Surgery in Treating Patients With Previously Untreated Breast Cancer | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT00317200 | A Study of Paclitaxel Plus Bevacizumab in Patients With Chemosensitive Relapsed Small Cell Lung Cancer | small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT03498521 | Study of Paclitaxel in Combination With BOS172722 in Patients With Advanced Nonhaematologic Malignancies | cancer | Paclitaxel (NPC208553) | |
| NCT00542191 | Phase II Trial of Neoadjuvant Metronomic Chemotherapy in Triple-Negative Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT03020329 | Induction Chemotherapy in Young Patients With Locoregionally Advanced Nasopharyngeal Carcinoma | nasopharyngeal neoplasm | Paclitaxel (NPC208553) | |
| NCT00434226 | A Study of Sunitinib in Combination With Bevacizumab, Carboplatin, and Paclitaxel in Patients With Advanced Non-Small Cell Lung Cancer (SABRE-L) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02733250 | Pembrolizumab With Nab-Paclitaxel in Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT05203913 | Cisplatin, Nab-paclitaxel, Nivolumab With Radiotherapy After Resection of Non-Metastatic Muscle Invasive Bladder Cancer | urinary bladder carcinoma | Paclitaxel (NPC208553) | |
| NCT02020707 | Nab-Paclitaxel and Bevacizumab in Treating Patients With Unresectable Stage IV Melanoma or Gynecological Cancers | cervical adenocarcinoma;cutaneous melanoma | Paclitaxel (NPC208553) | |
| NCT04675528 | Food Effect on Pharmacokinetics and SafEty of DHP107 (Liporaxel®) FEEL Study | neoplasm | Paclitaxel (NPC208553) | |
| NCT00976911 | Paclitaxel and Cisplatin as First-Line Treatment for Patients With Stage I, Stage II, Stage III, or Stage IV Ovarian Epithelial Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00522834 | Elesclomol (STA-4783) With Paclitaxel Versus Paclitaxel Alone in Melanoma | melanoma | Paclitaxel (NPC208553) | |
| NCT00002937 | Paclitaxel With or Without PSC 833 in Treating Patients With Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00290537 | Phase II Study of ZD6474 in Advanced NSCLC | lung cancer | Paclitaxel (NPC208553) | |
| NCT02437812 | Study of Paclitaxel, Carboplatin and Oral Metformin in the Treatment of Advanced Stage Ovarian Carcinoma | ovarian carcinoma | Paclitaxel (NPC208553) | |
| NCT04447092 | Pembrolizumab Plus Chemotherapy in 1st Line Treatment of Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00003614 | Paclitaxel Plus Estramustine in Treating Patients With Metastatic Prostate Cancer | prostate cancer | Paclitaxel (NPC208553) | |
| NCT00081302 | Cetuximab, Paclitaxel, Carboplatin, and Radiation Therapy in Treating Patients With Unresectable Stage IIIA or Stage IIIB Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT02244502 | Safety, Feasibility and Effect of TTFields (200 kHz) Concomitant With Weekly Paclitaxel in Recurrent Ovarian Carcinoma (INNOVATE) | ovarian carcinoma | Paclitaxel (NPC208553) | |
| NCT00004089 | Chemotherapy Plus Radiation Therapy in Treating Patients With Previously Untreated Thyroid Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT00040794 | Combination Chemotherapy, Radiation Therapy, and Gefitinib in Treating Patients With Stage III Non-Small Cell Lung Cancer | lung adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00793897 | Multiple Dose Study In Cancer Patients: Safety and Tolerability of BMS-754807 in Combination With Paclitaxel and Carboplatin in Patients With Advanced or Metastatic Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT04729608 | Alpelisib Plus Olaparib in Platinum-resistant/Refractory, High-grade Serous Ovarian Cancer, With no Germline BRCA Mutation Detected | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT04196283 | Nab-paclitaxel in Combination With Gemcitabine for Pediatric Relapsed and Refractory Solid Tumors | cancer | Paclitaxel (NPC208553) | |
| NCT00934895 | Phase I/II Study of Weekly Abraxane and RAD001 in Women With Locally Adv. or Metastatic Breast Ca | breast cancer | Paclitaxel (NPC208553) | |
| NCT00108745 | Paclitaxel, Polyglutamate Paclitaxel, or Observation in Treating Patients With Stage III or Stage IV Ovarian Epithelial, Peritoneal Cancer, or Fallopian Tube Cancer | endometrioid carcinoma;undifferentiated carcinoma | Paclitaxel (NPC208553) | |
| NCT03126708 | Cetuximab in Combination With Paclitaxel Plus Cisplatin Versus Paclitaxel Plus Cisplatin Alone for the First-line Treatment of Metastatic Esophageal Squamous Cell Carcinoma | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT02607982 | CCRT for Esophageal Cancer. | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00055887 | Chemotherapy and Radiation Therapy With or Without Efaproxiral in Treating Patients With Stage III Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT03635489 | Study on MK-3475 Plus Chemotherapy Versus Chemotherapy Alone in Recurrent, Platinum-resistant Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT03918278 | A Study of MK-0482 as Monotherapy and in Combination With Pembrolizumab (MK-3475) in Participants With Advanced Solid Tumors (MK-0482-001) | neoplasm | Paclitaxel (NPC208553) | |
| NCT02382406 | Carboplatin/Nab-Paclitaxel and Pembrolizumab in NSCLC | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT04190745 | Toripalimab Combined With Apatinib Mesylate for the Treatment of Gastric Adenocarcinoma in a Prospective Randomized Multicenter Phase II Clinical Study | gastric adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00005647 | SU5416 and Paclitaxel in Treating Patients With Recurrent, Locally Advanced or Metastatic Cancer of the Head and Neck | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT04390958 | Neoadjuvant Chemotherapy With Nab-paclitaxel Plus Cisplatin and Capecitabine for Locally Advanced Thoracic Esophageal Squamous Cell Carcinoma | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00679029 | Combination Chemotherapy and Bevacizumab in Treating Women With HER2/Neu-Negative Stage II or Stage III Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT01641939 | A Study of Trastuzumab Emtansine Versus Taxane in Participants With Human Epidermal Growth Factor Receptor 2 (HER2)-Positive Advanced Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT04770272 | Study to Compare a Mono Atezolizumab Window Followed by a Atezolizumab - CTX Therapy With Atezolizumab - CTX Therapy | triple-negative breast cancer | Paclitaxel (NPC208553) | |
| NCT00017303 | Combination Chemotherapy Plus IM-862 in Treating Patients With Resected Stage III Ovarian Cancer or Primary Peritoneal Cancer | ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT00933387 | A Study of Neoadjuvant Bio-C/T Followed by Concurrent Bio-R/T in High-risk Locally Advanced Oral Squamous Cell Carcinoma | mouth neoplasm | Paclitaxel (NPC208553) | |
| NCT02985333 | Paclitaxel,Cyclophosphamide and Dexamethasone for Relapsed or Refractory Multiple Myeloma | multiple myeloma | Paclitaxel (NPC208553) | |
| NCT00005048 | Estramustine and Paclitaxel in Treating Patients With Prostate Cancer That Has Not Responded to Hormone Therapy | prostate cancer | Paclitaxel (NPC208553) | |
| NCT00353717 | Study In Women And Men With Metastatic Breast Cancer That Have Overexpression Of ErbB2 | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT00094835 | Study to Evaluate Motesanib With or Without Carboplatin/Paclitaxel or Panitumumab in the Treatment of Patients With Advanced Non-Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00496028 | Phase I Study in Patients With Solid Tumours | neoplasm | Paclitaxel (NPC208553) | |
| NCT04799639 | Efficacy and Toxicity of Paclitaxel, Cisplatin Combined With Sindilimab in NACT for Locally Advanced Cervical Cancer | cervical cancer | Paclitaxel (NPC208553) | |
| NCT00231075 | Evaluation of the Safety and Efficacy of Paraplatin in Combination With Taxol in Patients of 70 Years and Older With Ovary Cancer F.I.G.O. Stages III-IV | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT03943043 | Gemcitabine + Oxaliplatin +Nab-paclitaxel in Subjects With Advanced Biliary Tract Cancer | cholangiocarcinoma | Paclitaxel (NPC208553) | |
| NCT04821765 | Study of PD-1 Antibody Combined With Chemoradiotherapy in Oligometastatic Esophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00022230 | Combination Chemotherapy Plus Biological Therapy in Treating Patients With Stage II or Stage III Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT04337632 | NUVOLA TRIAL Open-label Multicentre Study | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00003158 | S9712: Radiation Therapy and Combination Chemotherapy in Treating Patients With Stage III Non-small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT03751761 | GC 2nd Line Durvalumab(MEDI4736)/Tremelimumab Plus Paclitaxel Study | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00971945 | Rollover Study of Weekly Paclitaxel (BMS-181339) in Patients With Advanced Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT02432365 | Study of Weekly Paclitaxel and Cisplatin in FIGO IB2 and IIA2 Cervical Cancer Followed by Radical Hysterectomy | cervical cancer | Paclitaxel (NPC208553) | |
| NCT01268059 | A Study of Carboplatin and Paclitaxel With or Without MEDI-575 in Untreated, Advanced Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01471132 | Sequential HIPEC of Oxaliplatin and Paclitaxel for Gastric Cancer Patients With Peritoneum Metastasis | gastric cancer | Paclitaxel (NPC208553) | |
| NCT01494688 | A Study of RO5509554 as Monotherapy and in Combination With Paclitaxel in Participants With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT04827953 | Study to Evaluate the Safety and Efficacy of Treatment With NLM-001 and Standard Chemotherapy Plus Zalifrelimab in Patients With Advanced Pancreatic Cancer | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT01411410 | Phase I Study of PI3(Phosphoinositol 3)-Kinase Inhibitor BAY80-6946 With Paclitaxel in Patients With Advanced Cancer | neoplasm | Paclitaxel (NPC208553) | |
| NCT01560104 | A Clinical Study Conducted in Multiple Centers Comparing Veliparib in Combination With Carboplatin and Paclitaxel Versus a Placebo in Combination With Carboplatin and Paclitaxel in Patients With Advanced Non-small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02723955 | Dose Escalation and Expansion Study of GSK3359609 in Participants With Selected Advanced Solid Tumors (INDUCE-1) | neoplasm | Paclitaxel (NPC208553) | |
| NCT00113516 | A Study Of SU011248 As Therapy In Patients With Locally Advanced Or Metastatic Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00358163 | Trial of PTK787/ZK 222584 Plus Paclitaxel | cancer | Paclitaxel (NPC208553) | |
| NCT03932409 | Frontline Immunotherapy Combined With Radiation and Chemotherapy in High Risk Endometrial Cancer | endometrial cancer | Paclitaxel (NPC208553) | |
| NCT04282109 | Study to Assess the Efficacy and Safety of Nivolumab in Combination With Paclitaxel in Subjects With Head and Neck Cancer Unable for Cisplatin-based Chemotherapy (NIVOTAX) | head and neck squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00434135 | Alimta and Gemcitabine in Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT03579771 | Gemcitabine, Cisplatin, and Nab-Paclitaxel Before Surgery in Patients With High-Risk Liver Bile Duct Cancer | intrahepatic cholangiocarcinoma | Paclitaxel (NPC208553) | |
| NCT00918203 | A Study of Paclitaxel/Carboplatin With or Without Olaratumab (IMC-3G3) in Previously Untreated Locally Advanced or Metastatic Non-Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00403130 | Phase 2 Study of Gemzar, Taxol & Avastin Combination as 1st Line Treatment for Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT04590625 | Study With Afuresertib and Paclitaxel in Platinum Resistant Ovarian | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00006459 | Paclitaxel With or Without Gemcitabine in Treating Women With Advanced Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT01946074 | A Study of ABT-165 in Subjects With Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT05070598 | Camrelizumab Combined With Chemotherapy in First-line Treatment of HER2-positive Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT01506973 | A Phase I/II/Pharmacodynamic Study of Hydroxychloroquine in Combination With Gemcitabine/Abraxane to Inhibit Autophagy in Pancreatic Cancer | adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT05227664 | A Study of AK117/AK112 in Metastatic Triple-Negative Breast Cancer | triple-negative breast cancer | Paclitaxel (NPC208553) | |
| NCT00009776 | Monoclonal Antibody Therapy, Paclitaxel, and Cyclosporine in Treating Patients With Recurrent or Refractory Non-Hodgkin's Lymphoma | lymphoma | Paclitaxel (NPC208553) | |
| NCT02109341 | Phase I/II Study to Evaluate Nab-paclitaxel in Substitution of CPT11 or Oxaliplatin in FOLFIRINOX Schedule as First Line Treatment in Metastatic Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00003376 | Two-Drug Combination Chemotherapy Compared With Four-Drug Combination Chemotherapy in Treating Patients With Advanced Cancer of the Urothelium | urethra cancer;urinary bladder cancer | Paclitaxel (NPC208553) | |
| NCT00003313 | Amifostine in Treating Patients With Stage II or Stage III Non-small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00660842 | Study Comparing Weekly Versus Every 3 Week Chemotherapy in Patients With Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT03892018 | The Effect of Food on the Pharmacokinetics of Paclitaxel Administered Orally as Oraxol | neoplasm | Paclitaxel (NPC208553) | |
| NCT00004202 | Combination Chemotherapy, Radiation Therapy, and RSR13 in Treating Patients With Stage III Non-small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT04104672 | A Study to Evaluate the Safety and Tolerability of AB680 in Participants With Gastrointestinal Malignancies | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00004979 | Topotecan and Paclitaxel in Treating Patients With Advanced Non-small Cell Lung Cancer or Other Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT00786552 | Pemetrexed + Paclitaxel in Patients With Recurrent/Advanced Thyroid Cancer | thyroid cancer | Paclitaxel (NPC208553) | |
| NCT00002949 | Vinorelbine and Paclitaxel Plus Radiation Therapy in Treating Patients With Advanced Cancer Arising in the Pelvis | vaginal cancer;cervical cancer;endometrial cancer;urinary bladder cancer | Paclitaxel (NPC208553) | |
| NCT00645177 | Phase 2 Study of ABT-869 in Combination With Paclitaxel Versus Paclitaxel Alone to Treat Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00009997 | Trastuzumab and Chemotherapy Followed by Surgery and Combination Chemotherapy in Treating Women With Locally Advanced Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00378313 | A Study of Gemcitabine, Epirubicin, and Paclitaxel Combination Chemotherapy Given Before Surgery to Patients With Locally Advanced Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT01009515 | Carboplatin, Paclitaxel, and Temozolomide for Patients With Metastatic Melanoma | melanoma | Paclitaxel (NPC208553) | |
| NCT00976573 | Carboplatin, Paclitaxel, and Bevacizumab With or Without Everolimus in Treating Patients With Metastatic Malignant Melanoma | cutaneous melanoma | Paclitaxel (NPC208553) | |
| NCT02455141 | A Study Assessing the Safety and Efficacy of Adding Ipatasertib to Paclitaxel Treatment in Participants With Breast Cancer That Has Spread Beyond the Initial Site, and the Cancer Does Not Have Certain Hormonal Receptors | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT02611700 | Nimotuzumab in Combined With Paclitaxel and Cisplatin for Treatment of Metastatic Esophageal Squamous Cell Carcinoma | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00584909 | A Phase II Study of Paclitaxel and Carboplatin in Patients With an Elevated-Risk Cancer of the Uterus | uterine cancer | Paclitaxel (NPC208553) | |
| NCT00111787 | Study Of Lapatinib In Combination With Paclitaxel In The Treatment Of Newly Diagnosed Inflammatory Breast Cancer | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT00563784 | TARCEVA (Erlotinib) in Combination With Chemoradiation in Patients With Stage IIIA/B Non-Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00151034 | Trastuzumab (Herceptin), Paclitaxel, Carboplatin and Gemcitabine in Advanced Urothelial Cancer | urogenital neoplasm | Paclitaxel (NPC208553) | |
| NCT01534585 | Safety and Efficacy Study of Icotinib With Intensity-modulated Radiotherapy in Nasopharyngeal Carcinoma | nasopharyngeal neoplasm | Paclitaxel (NPC208553) | |
| NCT03928314 | Study of ORIC-101 in Combination With Anticancer Therapy | neoplasm | Paclitaxel (NPC208553) | |
| NCT00728845 | Hydroxychloroquine, Carboplatin, Paclitaxel, and Bevacizumab in Recurrent Advanced Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00980954 | Chemotherapy and Pelvic Radiation Therapy With or Without Additional Chemotherapy in Treating Patients With High-Risk Early-Stage Cervical Cancer After Radical Hysterectomy | cervical cancer | Paclitaxel (NPC208553) | |
| NCT01755845 | Trial of Postoperative Chemoradiotherapy With or Without Consolidation Chemotherapy for Cervical Cancer Patients | cervical cancer | Paclitaxel (NPC208553) | |
| NCT03435289 | A Study of CPI-613 With Gemcitabine and Nab-paclitaxel for Patients With Advanced or Metastatic Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00178256 | Pulsed Paclitaxel And Daily Thoracic Radiotherapy For Inoperable (Stage I/II) Or Unresectable Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT03169777 | QUILT-3.050: NANT Colorectal Cancer (CRC) Vaccine: Combination Immunotherapy in Subjects With Recurrent or Metastatic CRC | colorectal carcinoma | Paclitaxel (NPC208553) | |
| NCT05017012 | A Study to Evaluate the Bioavailability of Pembrolizumab (MK-3475) Via Subcutaneous (SC) Injection of MK-3475A (Pembrolizumab Formulated With MK-5180) In Advanced Solid Tumors (MK-3475A-C18) | neoplasm | Paclitaxel (NPC208553) | |
| NCT02047500 | Phase I TH-302 Plus Gemcitabine Plus Nab-Paclitaxel in Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00512096 | Neoadjuvant Chemotherapy for Patients With Squamous Cell Carcinoma of the Penis | penile cancer | Paclitaxel (NPC208553) | |
| NCT04257448 | Safety and Tolerance of Epigenetic and Immunomodulating Drugs Combined With Chemotherapeutics in Patients Suffering From Advanced Pancreatic Cancer | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT04826679 | Neoadjuvant Camrelizumab in Combination With Cisplatin and Nab-paclitaxel in Resectable HNSCC | head and neck squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00006248 | S0007 - Combination Chemotherapy in Treating Patients With Metastatic or Recurrent Head and Neck Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT04767295 | A Study of Camrelizumab Combined With Chemotherapy as Neoadjuvant Therapy in Adcanced Esophageal Squamous Cell Carcinoma (ESCC) | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT01100931 | A Phase I/II Study of Paclitaxel, Carboplatin and YM155 (Survivin Suppressor) in Subjects With Solid Tumors (Phase I) and Advanced Non-Small Cell Lung Carcinoma (Phase II) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00495170 | Concurrent Proton and Chemotherapy in Locally Advanced Stage IIIA/B Non-Small Cell Lung Cancer (NSCLC) | lung cancer | Paclitaxel (NPC208553) | |
| NCT03805399 | FUSCC Refractory TNBC Umbrella (FUTURE) | triple-negative breast cancer | Paclitaxel (NPC208553) | |
| NCT00003088 | Combination Chemotherapy in Treating Patients With Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00024310 | Paclitaxel, Folic Acid, and Lometrexol in Treating Patients With Locally Advanced or Metastatic Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT00005838 | Combination Chemotherapy Plus Radiation Therapy With or Without AE-941 in Treating Patients With Stage III Non-small Cell Lung Cancer That Cannot Be Removed By Surgery | lung adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00002894 | Platinum-based Chemotherapy With or Without Paclitaxel in Treating Patients With Relapsed Ovarian Cancer | ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT02389985 | Paclitaxel/Pazopanib for Platinum Resistant/Refractory Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00006257 | SU5416 and Paclitaxel in Treating Patients With Advanced Cancer | neoplasm | Paclitaxel (NPC208553) | |
| NCT00984464 | Study of REOLYSIN® in Combination With Paclitaxel and Carboplatin in Patients With Metastatic Melanoma | metastatic melanoma | Paclitaxel (NPC208553) | |
| NCT00273507 | Neoadjuvant Chemotherapy in Non Small Cell Lung Cancer (NSCLC) Stages IB, IIA, IIB, and IIIA/T3 | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01930292 | Debio 1143 in Combination With Carboplatin and Paclitaxel in Patient With Advanced Solid Malignancies | neoplasm | Paclitaxel (NPC208553) | |
| NCT00006012 | Combination Chemotherapy and Radiation Therapy in Treating Patients With Limited-Stage Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT04766359 | Paclitaxel (Albumin-bound) Combined With Radiotherapy for the Treatment of Early Stage Nasopharyngeal Carcinoma | nasopharyngeal neoplasm | Paclitaxel (NPC208553) | |
| NCT01970722 | Surgery and Chemotherapy With or Without Chemotherapy After Surgery in Treating Patients With Ovarian, Fallopian Tube, Uterine, or Peritoneal Cancer | Fallopian Tube Carcinoma;ovarian carcinoma;uterine neoplasm | Paclitaxel (NPC208553) | |
| NCT02272790 | Adavosertib Plus Chemotherapy in Platinum-Resistant Epithelial Ovarian, Fallopian Tube, or Primary Peritoneal Cancer | Fallopian Tube Carcinoma;ovarian carcinoma | Paclitaxel (NPC208553) | |
| NCT03807999 | Nab-Paclitaxel Plus Gemcitabine Versus Gemcitabine For The First Line Treatment of Pancreas Cancer | pancreatic neoplasm | Paclitaxel (NPC208553) | |
| NCT00785291 | Paclitaxel, Nab-paclitaxel, or Ixabepilone With or Without Bevacizumab in Treating Patients With Stage IIIC or Stage IV Breast Cancer | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT00642759 | Carboplatin, Abraxane, and Bevacizumab in Previously Untreated Patients With Advanced Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00009802 | Calcitriol Plus Paclitaxel in Treating Patients With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT03777462 | Borderline Resectable Pancreatic Cancer Neoadjuvant Chemoradiotherapy Clinicaltrial-1 | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT03806049 | Study of Relacorilant in Combination With Nab-Paclitaxel for Patients With Recurrent Platinum-Resistant Ovarian, Fallopian Tube, or Primary Peritoneal Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT03989336 | Apatinib With Albumin-bound Paclitaxel in Patients With Platinum-resistant Recurrent Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT05052931 | Nab-paclitaxel Combined With Oxaliplatin and S-1 for Perioperative Treatment of Advanced Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT03257033 | Intra-arterial Gemcitabine vs. IV Gemcitabine and Nab-Paclitaxel Following Radiotherapy for LAPC | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00009828 | Paclitaxel Combined With Fluorouracil-Uracil and Leucovorin in Treating Patients With Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT03649321 | Clinical Trial of Chemotherapy and Bemcentinib for Metastatic Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00011986 | Combination Chemotherapy in Treating Patients With Stage III or Stage IV Ovarian Epithelial Cancer or Primary Peritoneal Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00986674 | Carboplatin and Paclitaxel Combined With Cetuximab and/or IMC-A12 in Patients With Advanced Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT04047862 | Study of Ociperlimab (BGB-A1217) in Combination With Tislelizumab in Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT00034164 | Safety and Efficacy of S-8184 in Second Line Treatment of Relapsed Stage IIIB or IV Non Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01676649 | Ipilimumab With Carboplatin and Paclitaxel in Patients With Unresectable Stage III and Stage IV Melanoma | melanoma | Paclitaxel (NPC208553) | |
| NCT03834948 | AO-176 in Multiple Solid Tumor Malignancies | neoplasm | Paclitaxel (NPC208553) | |
| NCT00226746 | Efficacy Study of Gemcitabine, Paclitaxel, and Irradiation in the Treatment of Pancreatic Cancer | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT05092373 | Tumor Treating Fields Therapy in Combination With Chemotherapy for the Treatment of Advanced Solid Tumors Involving the Abdomen or Thorax | renal cell carcinoma;hepatocellular carcinoma;endometrial carcinoma;Fallopian Tube Carcinoma;ovarian carcinoma;breast carcinoma | Paclitaxel (NPC208553) | |
| NCT01137994 | A Randomized Trial Comparing EC-wPversus EP-wP as Adjuvant Therapy for Operable Breast Cancer Patients Less Than 40 Years Old | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT00003089 | Chemotherapy, Amifostine, and Radiation Therapy in Treating Patients With Non-small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00231868 | A Study of Radiation Therapy and Paclitaxel and Carboplatin in Patients With Uterine Papillary Serous Carcinoma | uterine cancer | Paclitaxel (NPC208553) | |
| NCT03830385 | Paclitaxel (Albumin Bound),Bleomycin And Cisplatin Or Carboplatin for Recurrent Or Metastatic Squamous Cell Carcinoma Of The Head And Neck | head and neck squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT02890511 | A Study DHP107, a Novel Oral Paclitaxel Formulation, in Patients With Advanced Solid Tumours or Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT04711161 | First-in-Human Evaluation of GRN-300 in Subjects With Recurrent Ovarian, Primary Peritoneal, and Fallopian Tube Cancers. | ovarian neoplasm | Paclitaxel (NPC208553) | |
| NCT00003402 | Peripheral Stem Cell Transplantation Plus Combination Chemotherapy in Treating Patients With Low-Grade Non-Hodgkin's Lymphoma or Chronic Lymphocytic Leukemia | leukemia;lymphoma | Paclitaxel (NPC208553) | |
| NCT03154294 | Evaluation of the Safety and Tolerability of TAK-228 With TAK-117 and Paclitaxel in Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT05158062 | Pembrolizumab and Bevacizumab With Chemotherapy Followed by Pembrolizumab, Bevacizumab and Olaparib in Recurrent Ovarian Cancer | ovarian carcinoma | Paclitaxel (NPC208553) | |
| NCT00768469 | Study Evaluating Safety And Tolerability, Solid Tumor | neoplasm | Paclitaxel (NPC208553) | |
| NCT03894540 | Dose Escalation and Dose Expansion Study of IPN60090 in Patients With Advanced Solid Tumours | neoplasm | Paclitaxel (NPC208553) | |
| NCT04180384 | A Safety Study of Oraxol (HM30181 + Oral Paclitaxel) in Cancer Patients | neoplasm | Paclitaxel (NPC208553) | |
| NCT01594099 | Concurrent Chemoradiotherapy for Cervical Cancer in Elderly Women | cervical cancer | Paclitaxel (NPC208553) | |
| NCT00003251 | Amifostine Plus Chemotherapy and Radiation Therapy in Treating Patients With Advanced, Unresectable Head and Neck Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT00394251 | Study of Dose-dense Adriamycin Plus Cytoxan (AC) Followed by Either ABI-007 (Abraxane) or Taxol With Bevacizumab as Adjuvant Therapy for Patients With Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00002734 | Radiolabeled Monoclonal Antibody, Paclitaxel, and Interferon Alfa in Treating Patients With Recurrent Ovarian Cancer | ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT00004174 | Combination Chemotherapy Followed By Peripheral Stem Cell Transplantation in Treating Patients With Advanced Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT03946969 | Sintilimab in Combination With Chemotherapy in Neoadjuvant Treatment of Potentially Resectable Esophageal Cancer | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00526890 | Carboplatin, Paclitaxel, Selenomethionine, and Radiation Therapy in Treating Patients With Stage III Non-Small Cell Lung Cancer That Cannot Be Removed by Surgery | lung cancer | Paclitaxel (NPC208553) | |
| NCT00019812 | Monoclonal Antibody Plus Chemotherapy in Treating Patients With Metastatic Breast Cancer That Overexpresses HER2 | breast cancer | Paclitaxel (NPC208553) | |
| NCT05047991 | Study of Irinotecan Liposome Injection-containing Regimens Versus Nab-paclitaxel Plus Gemcitabine in Patients With Previously Untreated, Metastatic Pancreatic Adenocarcinoma | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00458315 | Cisplatin/Paclitaxel/Gemcitabine +/- Avastin in Patients With Unknown Primary Tumor | neoplasm | Paclitaxel (NPC208553) | |
| NCT03328494 | Trial of ZW25 (Zanidatamab) in Patients With Advanced HER2-expressing Cancers | cancer | Paclitaxel (NPC208553) | |
| NCT00874848 | Efficacy/Safety of Imprime PGG With Cetuximab & Paclitaxel/Carboplatin Therapy in Pts With Untreated Advanced Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00654836 | Carboplatin, Paclitaxel, and Bevacizumab in Treating Patients With Locally Recurrent or Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00005649 | Paclitaxel and Capecitabine in Treating Women With Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00028769 | S0032, Combination Chemotherapy Plus Hormone Therapy in Treating Patients With Metastatic Prostate Cancer | prostate cancer | Paclitaxel (NPC208553) | |
| NCT01693276 | Gemcitabine/Abraxane Chemotherapy and Dose Escalated Radiotherapy for Locally Advanced, Unresectable Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT04449549 | Rapid Analysis and Response Evaluation of Combination Anti-Neoplastic Agents in Rare Tumors (RARE CANCER) Trial: RARE 1 Nilotinib and Paclitaxel | neoplasm | Paclitaxel (NPC208553) | |
| NCT04339738 | Testing the Addition of Nivolumab to Chemotherapy in Treatment of Soft Tissue Sarcoma | angiosarcoma | Paclitaxel (NPC208553) | |
| NCT00005806 | Combination Chemotherapy in Treating Patients With Advanced Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00600340 | 2-arm Trial of Paclitaxel Plus Bevacizumab vs. Capecitabine Plus Bevacizumab | breast cancer | Paclitaxel (NPC208553) | |
| NCT04603846 | A Study of Anti-PD-L1 Antibody ZKAB001 Combined With Albumin-bound Paclitaxel in Advanced Urothelial Carcinoma | urothelial carcinoma | Paclitaxel (NPC208553) | |
| NCT01978184 | Randomized Phase II Trial of Pre-Operative Gemcitabine and Nab Paclitacel With or With Out Hydroxychloroquine | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT01461915 | Phase 2 Study to Assess the Efficacy & Safety of Gemcitabine + Nab Paclitaxel With or Without Dociparstat in Metastatic Pancreatic Cancer Patients | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT02383251 | Study of Metformin With Carboplatin/Paclitaxel Chemotherapy in Patients With Advanced Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT02256436 | A Study of Pembrolizumab (MK-3475) Versus Paclitaxel, Docetaxel, or Vinflunine for Participants With Advanced Urothelial Cancer (MK-3475-045/KEYNOTE-045) | urogenital neoplasm | Paclitaxel (NPC208553) | |
| NCT00546156 | Preoperative Dose-dense Doxorubicin and Cyclophosphamide Followed by Paclitaxel With Bevacizumab in Operable Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00300885 | A Randomized Controlled Trial Comparing Safety and Efficacy of Carboplatin and Paclitaxel Plus or Minus Sorafenib (BAY 43-9006) in Chemonaive Patients With Stage IIIB-IV Non-Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02822157 | Circulating Tumor DNA Guiding (Olaparib) Lynparza® Treatment in Ovarian Cancer | malignant epithelial tumor of ovary | Paclitaxel (NPC208553) | |
| NCT01196741 | Neoadjuvant Therapy for Ovarian Cancer | fallopian tube cancer;ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00003206 | Carboplatin and Paclitaxel in Treating Patients With Locally Recurrent or Metastatic Nasopharyngeal Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT02501902 | Dose-Escalation Study Of Palbociclib + Nab-Paclitaxel In mPDAC | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00937560 | ZD4054 (Zibotentan) or Placebo Plus Chemotherapy in Patients With Advanced Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00005819 | Combination Chemotherapy in Treating Patients With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT00014573 | Chemotherapy and Vaccine Therapy Followed by Bone Marrow or Peripheral Stem Cell Transplantation and Interleukin-2 in Treating Patients With Recurrent or Refractory Brain Cancer | Central Nervous System Neoplasm | Paclitaxel (NPC208553) | |
| NCT00003927 | Combination Chemotherapy, Amifostine, and Peripheral Stem Cell Transplantation in Treating Patients With Stage II, Stage III, or Stage IV Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00025688 | Paclitaxel With or Without Carboplatin in Treating Women With Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00003064 | Combination Chemotherapy and Peripheral Stem Cell Transplantation in Treating Patients With Recurrent or Persistent Epithelial Ovarian Cancer, Fallopian Tube, or Primary Peritoneal Cancer | fallopian tube cancer;ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT00881101 | Clinical Study of Liposomal Paclitaxel in Chinese Patients | neoplasm | Paclitaxel (NPC208553) | |
| NCT00729612 | Paclitaxel Albumin-Stabilized Nanoparticle Formulation and Carboplatin in Treating Patients With Stage IIIB, Stage IV, or Recurrent Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00629278 | Combination Chemotherapy and Trastuzumab in Treating Women With Stage I, Stage II, or Stage III HER2-Positive Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT01113476 | Nab-paclitaxel, Gemcitabine, and Bevacizumab in Advanced Malignancies | cancer | Paclitaxel (NPC208553) | |
| NCT00006378 | Combination Chemotherapy Plus Radiation Therapy in Treating Patients With Stage III Non-small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT03852979 | Neo-Adjuvant Chemotherapy and Conservative Surgery in Cervical Cancer to Preserve Fertility | cervical cancer | Paclitaxel (NPC208553) | |
| NCT00091377 | Phenoxodiol Combined With Either Cisplatin or Paclitaxel in Patients With Recurrent Ovarian Epithelial, Fallopian Tube, or Primary Peritoneal Cancer | fallopian tube cancer;ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT03393884 | Neo-adjuvant Pembrolizumab in Primary Stage IV Ovarian Cancer | fallopian tube cancer | Paclitaxel (NPC208553) | |
| NCT02588443 | Study of Neo-adjuvant RO7009789 Alone or Neo-adjuvant RO7009789 Plus Nab-Paclitaxel and Gemcitabine Followed by Adjuvant RO7009789 Plus Nab-Paclitaxel and Gemcitabine for Patients With Newly Diagnosed Resectable Pancreatic Carcinoma | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00004137 | S9914: Combination Chemotherapy Plus Filgrastim in Untreated Extensive-Stage Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00008047 | Combination Chemotherapy Combined With Radiation Therapy in Treating Patients Who Have Stage II or Stage III Cancer of the Esophagus | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00014222 | Combination Chemotherapy With or Without Colony-stimulating Factors in Treating Women With Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT04949256 | Efficacy and Safety of Pembrolizumab (MK-3475) Plus Lenvatinib (E7080/MK-7902) Plus Chemotherapy in Participants With Metastatic Esophageal Carcinoma (MK-7902-014/E7080-G000-320/LEAP-014) | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT01303497 | Efficacity of Weekly Paclitaxel in Association or Not With Bevacizumab in Metastatic or Locally Advanced Angiosarcomas | angiosarcoma | Paclitaxel (NPC208553) | |
| NCT04000295 | An Open-label, Phase I/II Study of the Pan-immunotherapy in Patients With Relapsed/Refractory Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT03725436 | ALRN-6924 and Paclitaxel in Treating Patients With Advanced, Metastatic, or Unresectable Solid Tumors | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT03721744 | A Study of Napabucasin in Combination With Weekly Paclitaxel and Low-dose Gemcitabine in Patients With Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00174434 | Study Of SU011248 In Combination With Paclitaxel In Patients With Metastatic Breast Cancer | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT04516616 | Pd-1 Antibody Combined Neoadjuvant Chemotherapy for Locally Advanced Cervical Cancer | cervical adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00334763 | Radiation Therapy, Chemotherapy, and Bevacizumab in Treating Patients With Recurrent, Unresectable or Stage III or Stage IV Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00002568 | Combination Chemotherapy With or Without Surgery in Treating Patients With Stage III Ovarian Epithelial Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT01881230 | Evaluate Risk/Benefit of Nab Paclitaxel in Combination With Gemcitabine and Carboplatin Compared to Gemcitabine and Carboplatin in Triple Negative Metastatic Breast Cancer (or Metastatic Triple Negative Breast Cancer) | triple-negative breast cancer | Paclitaxel (NPC208553) | |
| NCT01873326 | Paclitaxel, Ifosfamide and Cisplatin (TIP) Versus Bleomycin, Etoposide and Cisplatin (BEP) for Patients With Previously Untreated Intermediate- and Poor-risk Germ Cell Tumors | germ cell tumor | Paclitaxel (NPC208553) | |
| NCT00002953 | Epirubicin and Cyclophosphamide Compared With Epirubicin and Paclitaxel in Treating Women With Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT04879368 | RegoNivo vs Standard of Care Chemotherapy in AGOC | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT02873598 | A Dose Escalation Trial of SBRT After Induction Chemotherapy for Locally Advanced Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00392704 | Treatment of Head & Neck Cancer With Chemotherapy and Radiation | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT00199758 | Study of an Early Change of a Chemotherapeutic Doublet Versus Four Cycles of Chemotherapy in Advanced Non Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00119262 | Bevacizumab and Combination Chemotherapy in Patients With Lymph Node Positive Breast Cancer | male breast carcinoma | Paclitaxel (NPC208553) | |
| NCT04864782 | QL1604 Plus Chemotherapy Versus Chemotherapy in Subjects With Stage ⅣB, Recurrent, or Metastatic Cervical Cancer | cervical cancer | Paclitaxel (NPC208553) | |
| NCT00262847 | Carboplatin and Paclitaxel With or Without Bevacizumab in Treating Patients With Stage III or Stage IV Ovarian Epithelial, Primary Peritoneal, or Fallopian Tube Cancer | endometrioid carcinoma | Paclitaxel (NPC208553) | |
| NCT00838656 | Intraperitoneal Paclitaxel Albumin-Stabilized Nanoparticle Formulation in Treating Patients With Advanced Cancer of the Peritoneal Cavity | ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT02718417 | Phase I Low Dose WART in Combination With Weekly Paclitaxel for Platinum Resistant Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT03912415 | Efficacy and Safety of BCD-100 (Anti-PD-1) in Combination With Platinum-Based Chemotherapy With and Without Bevacizumab as First-Line Treatment of Subjects With Advanced Cervical Cancer (FERMATA) | cervical cancer | Paclitaxel (NPC208553) | |
| NCT01253681 | A Study of the Addition of Avastin (Bevacizumab) to Carboplatin and Paclitaxel Therapy in Patients With Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT02101021 | Gemcitabine and Nab-paclitaxel Combined With Momelotinib in Participants With Previously Untreated Metastatic Pancreatic Ductal Adenocarcinoma | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00004094 | Combination Chemotherapy Plus Radiation Therapy in Treating Patients With Previously Untreated Advanced Cancer of the Mouth, Pharynx, or Larynx | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT01763645 | A Safety and Efficacy Study of BCD-021 With Paclitaxel and Carboplatin Compared to Avastin With Paclitaxel and Carboplatin in Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00614484 | Chemotherapy and Proton Radiation for the Treatment of Locally Advanced Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00583349 | Phase I & II Trial of Intravesicular Abraxane for Treatment-refractory Bladder Cancer | urinary bladder cancer | Paclitaxel (NPC208553) | |
| NCT00003880 | Paclitaxel Plus Carboplatin With or Without SCH-58500 in Treating Patients With Newly Diagnosed Stage III Ovarian or Stage III Primary Peritoneal Cancer | ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT00009763 | Monoclonal Antibody Therapy, Cyclosporine, and Paclitaxel in Treating Patients With Recurrent or Refractory Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT01107665 | Pazopanib and Paclitaxel as First-Line Treatment for Subjects With Unresectable Stage III and Stage IV Melanoma | melanoma | Paclitaxel (NPC208553) | |
| NCT02033993 | Nab-Paclitaxel to Paclitaxel in Advanced Urothelial Cancer Progressing on or After Platinum Containing Regimen. | urogenital neoplasm | Paclitaxel (NPC208553) | |
| NCT00014599 | Paclitaxel in Treating Patients With Locally Advanced, Metastatic, or Recurrent Cancer of the Vulva | vulva cancer | Paclitaxel (NPC208553) | |
| NCT03519308 | A Pilot Study of Perioperative Nivolumab and Paricalcitol to Target the Micoenvironment in Resectable Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT05189483 | Albumin-bound Paclitaxel Plus Camrelizumab for Advanced Soft Tissue Sarcoma. | soft tissue sarcoma | Paclitaxel (NPC208553) | |
| NCT01641783 | Efficacy and Safety Study of ABI-007 Plus Capecitabine as First-line Chemotherapy for Advanced Gastric Cancer Patients | gastric adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT04731038 | Combination Therapy for First Line Treatment of Advanced Cervical Cancer | cervical cancer | Paclitaxel (NPC208553) | |
| NCT00189566 | Taxol® in Monotherapy or in Combination With Topotecan or Carboplatin in Patients With Epithelial Ovarian Cancer in Early Relapse | fallopian tube cancer;ovarian cancer;peritoneal neoplasm | Paclitaxel (NPC208553) | |
| NCT00680758 | Cisplatin, Paclitaxel, and Everolimus in Treating Patients With Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT03829722 | Radiotherapy, Carboplatin/Paclitaxel and Nivolumab for High Risk HPV-related Head and Neck Cancer | oropharynx squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00006472 | Combination Chemotherapy Plus Radiation Therapy Followed By Surgery in Treating Patients With Stage I, Stage II, or Stage III Esophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00005059 | Carboplatin and Paclitaxel in Treating Older Patients With Metastatic or Recurrent Unresectable Non-small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00752115 | Combination Chemotherapy With Sildenafil Plus Carboplatin and Paclitaxel in Patients With Advanced Non-small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT04802980 | A Study of HB002.1T Plus Chemotherapy in Subjects With Solid Tumor | neoplasm | Paclitaxel (NPC208553) | |
| NCT02845908 | POF Versus FOLFOX Versus FOLFOX Plus ip Paclitaxel in AGC | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00003899 | Chemotherapy Plus Bone Marrow or Peripheral Stem Cell Transplantation in Treating Patients With Recurrent or Refractory Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT00198432 | Chemoradiotherapy of NSCLC Stage IIIB | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00035152 | Study Comparing Weekly Taxol and Carboplatin vs Standard Taxol and Carboplatin Regimen for Stage IIIB or IV Non-Small-Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02403895 | AZD2014 and Weekly Paclitaxel in Squamous NSCLC | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00373256 | Combination Of Lapatinib With Carboplatin, Paclitaxel, and With or Without Trastuzumab In Metastatic Breast Cancer. | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT02366143 | A Study of Atezolizumab in Combination With Carboplatin Plus (+) Paclitaxel With or Without Bevacizumab Compared With Carboplatin+Paclitaxel+Bevacizumab in Participants With Stage IV Non-Squamous Non-Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01755897 | A Multicenter, Prospective, Randomized Trial of Adjuvant Chemotherapy for Early-Stage Cervical Cancer Patients | cervical adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT01024101 | Early Phase II Study of Weekly Paclitaxel (BMS-181339) in Patient With Advanced or Recurrent Head and Neck Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT00284752 | Phase II Trial of Abraxane in Front Line Therapy of Hormone Refractory Metastatic Prostate Cancer | prostate cancer | Paclitaxel (NPC208553) | |
| NCT00287937 | Vorinostat, Paclitaxel, and Carboplatin in Treating Patients With Advanced or Refractory Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT01225302 | A Study of Linifanib (ABT-869) in Combination With Carboplatin/Paclitaxel in Japanese Subjects With Non-Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00499603 | Paclitaxel Followed by FEC Versus Paclitaxel and RAD001 Followed by FEC In Women With Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00034151 | Safety and Efficacy of S-8184 in Second Line Treatment of Stage III or IV Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT03026881 | A Study of Fluzoparib Given in Combination With Apatinib and Paclitaxel in Gastric Cancer Patients | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00005028 | Paclitaxel and Bryostatin 1 in Treating Patients With Metastatic Prostate Cancer | prostate cancer | Paclitaxel (NPC208553) | |
| NCT00093756 | Bortezomib, Paclitaxel, Carboplatin and Radiation Therapy for Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01684878 | ICON8: Weekly Chemotherapy in Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT04238988 | Carboplatin-Paclitaxel-Pembrolizumab in Neoadjuvant Treatment of Locally Advanced Cervical Cancer | cervical cancer | Paclitaxel (NPC208553) | |
| NCT03197571 | QUILT-3.048: NANT Urothelial Cancer Vaccine: Combination Immunotherapy in Subjects With Urothelial Cancer Who Have Progressed on or After Chemotherapy and PD-1/PD-L1 Therapy | urothelial carcinoma | Paclitaxel (NPC208553) | |
| NCT02583477 | Phase Ib/II Study of MEDI4736 Evaluated in Different Combinations in Metastatic Pancreatic Ductal Carcinoma | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT02525653 | Albumin-Bound Paclitaxel and Gemcitabine in Patients With Untreated Stage IV or Recurrent Squamous Cell Lung Cancers | lung cancer | Paclitaxel (NPC208553) | |
| NCT00006484 | Combination Chemotherapy With or Without Tirapazamine in Treating Patients With Stage IIIB or Stage IV Non-small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT01836432 | Immunotherapy Study in Borderline Resectable or Locally Advanced Unresectable Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT02632071 | ACY-1215 + Nab-paclitaxel in Metastatic Breast Cancer | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT00228319 | Treatment of Newly Diagnosed Ovarian Cancer With Antioxidants | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00033696 | Combination Chemotherapy and Radiation Therapy in Treating Patients With Limited-Stage Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT02264990 | Study Comparing Veliparib Plus Carboplatin and Paclitaxel Versus Investigator's Choice of Standard Chemotherapy in Adults Receiving First Cytotoxic Chemotherapy for Metastatic or Advanced Non-Squamous Non-Small Cell Lung Cancer (NSCLC) and Who Are Current or Former Smokers | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT03586869 | QUILT-3.080: NANT Pancreatic Cancer Vaccine | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00686114 | Concurrent Chemoradiotherapy Containing Paclitaxel&Cisplatin With/Without Tarceva in Locally Advanced Esophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT02570711 | Phase 2, Nab Paclitaxel/Gemcitabine Alone and in Combination With ACP-196 in Subjects With Metastatic Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT01802749 | Phase I/Ib Study of Paclitaxel in Combination With VS-6063 in Patients With Advanced Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00668616 | Adjuvant Treatment of Breast Cancer With 1-3 Aflicted Lymph Nodes | breast cancer | Paclitaxel (NPC208553) | |
| NCT01264328 | Study of the Combination of Panitumumab With Paclitaxel as First-line Treatment of Subjects With Head and Neck Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT01244789 | Chemotherapy or Observation in Stage I-II Intermediate or High Risk Endometrial Cancer | endometrial cancer | Paclitaxel (NPC208553) | |
| NCT03002103 | A Trial Evaluating the Efficacy and Safety of EndoTAG®-1 in Combination With Paclitaxel and Gemcitabine Compared With Paclitaxel and Gemcitabine as First-line Therapy in Patients With Visceral Metastatic Triple-negative Breast Cancer | triple-negative breast cancer | Paclitaxel (NPC208553) | |
| NCT00756470 | Phase II Neoadjuvant in Inflammatory Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00052351 | Vaccine Therapy Plus Sargramostim and Chemotherapy in Treating Women With Stage II or Stage III Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT02560038 | Trial of Paclitaxel Plus Gemcitabine and Cisplatin in Bladder Cancer | bladder tumor | Paclitaxel (NPC208553) | |
| NCT00566540 | Ph 2 Intensification Regimen for Previously Untreated, Resectable, Advanced Squamous Cell Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT04973904 | Toripalimab Combined With Chemotherapy and Bevacizumab as First-Line Treatment in Patients With Advanced Cervical Cancer | cervical cancer | Paclitaxel (NPC208553) | |
| NCT00280787 | Induction Chemotherapy Using Paclitaxel, Carboplatin, CPT-11 With Pegfilgrastim | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00336791 | Randomized Clinical Trial to Evaluate the Predictive Accuracy of a Gene Expression for Stage I-II Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT03641183 | Folfirinox or Gemcitabine-Nab Paclitaxel Followed by Stereotactic Body Radiotherapy for Locally Advanced Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT01285466 | A Trial of Oral BEZ235 and BKM120 in Combination With Paclitaxel With or Without Trastuzumab | neoplasm | Paclitaxel (NPC208553) | |
| NCT00154700 | Twice Weekly TP-HDFL for Recurrent or Metastatic Esophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00021372 | Paclitaxel and Estramustine in Treating Patients With Relapsed or Refractory Non-Hodgkin's Lymphoma | lymphoma | Paclitaxel (NPC208553) | |
| NCT02036164 | Adjuvant Chemotherapy for Locally Advanced Cervical Cancer | cervical cancer | Paclitaxel (NPC208553) | |
| NCT01525602 | Safety Study of PLX3397 and Paclitaxel in Patients With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT00001272 | A Phase I Study of Taxol, Cisplatin, Cyclophosphamide and Granulocyte Colony-Stimulating Factor (G-CSF) in Previously Nontreated Ovarian Cancer Patients | ovarian neoplasm | Paclitaxel (NPC208553) | |
| NCT02615730 | PI3Kβ Selective Inhibitor With Paclitaxel, Advanced Gastric Adenocarcinoma | gastric adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT01325441 | A Study of BBI608 Administered With Paclitaxel in Adult Patients With Advanced Malignancies | cancer | Paclitaxel (NPC208553) | |
| NCT00003050 | Radiation Therapy Plus Paclitaxel in Treating Patients With Stage IIB or Stage III Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00006004 | Comparison of Two Combination Chemotherapy Regimens in Treating Non-small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT05000892 | Neoadjuvant Sintilimab in Combination With Carboplatin and Nab-paclitaxel in Untreated Salivary Gland Malignant Neoplasms | salivary gland cancer | Paclitaxel (NPC208553) | |
| NCT00874107 | Efficacy/Safety of Imprime PGG® Injection With Bevacizumab and Paclitaxel/Carboplatin in Patients With Untreated Advanced Non-Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00295789 | Trial of TP Versus TC Regimens for Stage IVb, Persistent, or Recurrent Cervical Cancer (JCOG0505, CC-TPTC-P3) | cervical adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00038545 | A Phase II Study of Paclitaxel and Topotecan With Filgrastim-SD/01 Support For Relapsed and Refractory Aggressive Non-Hodgkin's Lymphoma | non-Hodgkins lymphoma | Paclitaxel (NPC208553) | |
| NCT03367871 | Combination Pembrolizumab, Chemotherapy and Bevacizumab in Patients With Cervical Cancer | cervical cancer | Paclitaxel (NPC208553) | |
| NCT00318136 | A Study of Bevacizumab Plus Carboplatin and Paclitaxel in Subjects With Advanced, Previously Untreated, Squamous Non-Small Cell Lung Cancer (BRIDGE) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00998192 | A Study of REOLYSIN® in Combination With Paclitaxel and Carboplatin in Patients With Squamous Cell Carcinoma of the Lung | squamous cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT05449483 | Conversion of Tislelizumab Combined With Chemotherapy in Unresectable Esophageal Squamous Cell Carcinoma | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00102622 | Intraperitoneal tgDCC-E1 and Intravenous Paclitaxel in Women With Platinum-Resistant Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT01192165 | Safety and Tolerability Study of GSK1120212, a MEK Inhibitor, in Combination With Docetaxel, Erlotinib, Pemetrexed, Pemetrexed + Carboplatin, Pemetrexed + Cisplatin, or Nab-Paclitaxel | cancer | Paclitaxel (NPC208553) | |
| NCT03714555 | Disulfiram-Copper Gluconate in Met Pancreas Cancer w Rising CA19-9 on Abraxane-Gemzar, FOLFIRINOX or Gemcitabine | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT04085276 | Toripalimab in Combination With Nab-Paclitaxel For Patients With Metastatic or Recurrent Triple-Negative Breast Cancer (TNBC) With or Without Systemic Treatment | triple-negative breast cancer | Paclitaxel (NPC208553) | |
| NCT00042809 | Erlotinib, Trastuzumab, and Paclitaxel in Treating Patients With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT02328105 | LCI-LUN-ABR-001: Carbo With Nab-Paclitaxel in Patients With Advanced NSCL Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT04692051 | A Phase II Study for Nab-paclitaxel Plus Cisplatin vs Gemcitabine Plus Cispatin as First Line Chemotherapy in Advanced Biliary Tract Cancer | biliary tract cancer | Paclitaxel (NPC208553) | |
| NCT00825201 | First Line Tx Stage III Ovarian Cancer | peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT00268970 | Satraplatin and Paclitaxel in Patients With Advanced Non-small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02436993 | Phase II Breast Ca Carboplatin + Paclitaxel With Pertuzumab + Trastuzumab or Bevacizumab | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT00465907 | Study of Weekly Paclitaxel, Carboplatin and Irinotecan to Treat Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT01838538 | Abraxane/Bevacizumab | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT02769832 | Nab-Paclitaxel With Gemcitabine for Relapsed Small Cell Cancer | small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT04604132 | Derazantinib Alone or in Combination With Paclitaxel, Ramucirumab or Atezolizumab in Gastric Adenocarcinoma | gastric adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT03387098 | QUILT-3.070:Pancreatic Cancer Vaccine: Subjects With Pancreatic Cancer Who Have Progressed on or After Standard-of-care Therapy | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00691379 | Weekly Paclitaxel/Carboplatin/Bevacizumab as First Line Therapy for Triple Negative Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00997009 | Study of Adding Cetuximab to Chemotherapy for the Treatment of Advanced and/or Recurrent Cervical Cancer | cervical cancer | Paclitaxel (NPC208553) | |
| NCT01332656 | Study of AMG 386 in Combination With Paclitaxel and Carboplatin in Subjects With Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT05485766 | Novel Neoadjuvant and Adjuvant Strategy for Germline BRCA 1/2 Mutated Triple Negative Breast Cancer | Hereditary breast and ovarian cancer syndrome | Paclitaxel (NPC208553) | |
| NCT00006018 | Paclitaxel and BMS-214662 in Treating Patients With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT01711697 | An Alternative Radiation Fractionation Strategy for Locally Advanced Non-Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00095914 | Paclitaxel and ABI-007 in Treating Patients With Locally Advanced or Metastatic Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT00334321 | Pelvic IMRT With Tomotherapy in Post-Hysterectomy Endometrial Cancer Patients | endometrial cancer | Paclitaxel (NPC208553) | |
| NCT00003943 | Combination Chemotherapy Plus Peripheral Stem Cell Transplantation in Treating Patients With Metastatic Cancer. | lung cancer;carcinoma | Paclitaxel (NPC208553) | |
| NCT03430843 | A Study of Tislelizumab (BGB-A317) Versus Chemotherapy as Second Line Treatment in Participants With Advanced Esophageal Squamous Cell Carcinoma | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT04308837 | A Phase II Trial of Neoadjuvant Laparoscopic Hyperthermic Intraperitoneal Chemotherapy (HIPEC) With Chemoradiation | gastric cancer | Paclitaxel (NPC208553) | |
| NCT05177796 | Panitumumab and Pembrolizumab in Combination With Neoadjuvant Chemotherapy for the Treatment of Stage III-IV Triple Negative Breast Cancer | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT02124707 | Weekly Carboplatin, Paclitaxel and Cetuximab Treatment for Patients With Recurrent or Metastatic SCCHN | head and neck squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00003944 | Combination Chemotherapy Plus Peripheral Stem Cell Transplantation in Treating Patients With Stage III or Stage IV Ovarian, Fallopian Tube, or Primary Peritoneal Cancer | fallopian tube cancer;ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT05489848 | Chemotherapy vs Chemoradiotherapy for Post-operative Endometrial Cancer Patients With P53-mutation | endometrial cancer | Paclitaxel (NPC208553) | |
| NCT00533585 | BAY 43-9006 in Previously Untreated Patients With Non-Small Cell Lung Cancer (NSCLC) | lung cancer | Paclitaxel (NPC208553) | |
| NCT03167177 | QUILT-3.046: NANT Melanoma Vaccine: Combination Immunotherapy in Subjects With Melanoma Who Have Progressed On or After Chemotherapy and PD-1/PD-L1 Therapy | melanoma | Paclitaxel (NPC208553) | |
| NCT00730925 | Single-arm Trial of BIBW 2992 (Afatinib) in Demographically and Genotypically Selected NSCLC Patients | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02474173 | Onalespib and Paclitaxel in Treating Patients With Advanced Triple Negative Breast Cancer | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT00254592 | Neoadjuvant Treatment of Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00161278 | Pilot Study for the Determination of Tumor Response to Chemotherapy in Advanced NSCLC Through Gene Expression Profiling | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02142738 | Study of Pembrolizumab (MK-3475) Compared to Platinum-Based Chemotherapies in Participants With Metastatic Non-Small Cell Lung Cancer (MK-3475-024/KEYNOTE-024) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00072319 | Neoadjuvant or Adjuvant Epirubicin, Cyclophosphamide, and Paclitaxel in Treating Women With Stage I, Stage II, or Stage III Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT03030287 | A Phase 1b Study of OMP-305B83 Plus Paclitaxel in Subjects With Ovarian, Peritoneal or Fallopian Tube Cancer | Fallopian Tube Carcinoma;ovarian carcinoma;peritoneal neoplasm | Paclitaxel (NPC208553) | |
| NCT00061646 | Safety and Effectiveness of ABT-510 Plus Combination Chemotherapy in Subjects With Non-Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT03515798 | Study of Immunotherapy in Combination With Chemotherapy in HER2-negative Inflammatory Breast Cancer | inflammatory breast carcinoma | Paclitaxel (NPC208553) | |
| NCT01608009 | [18F]Fluciclatide-PET, Pazopanib and Paclitaxel in Ovarian Cancer | ovarian neoplasm | Paclitaxel (NPC208553) | |
| NCT04579224 | Comparing the New Anti-cancer Drug Eribulin With or Without Chemotherapy Against the Usual Chemotherapy Alone in Metastatic Urothelial Cancer | urethra cancer | Paclitaxel (NPC208553) | |
| NCT00031876 | Capecitabine and Paclitaxel in Treating Patients With Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT04683939 | Safety, Pharmacokinetics, Pharmacodynamics, and Preliminary Efficacy Trial of BNT141 in Patients With Unresectable or Metastatic CLDN18.2-positive Gastric, Pancreatic, Ovarian and Biliary Tract Tumors | cholangiocarcinoma;gastric cancer;pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT03432598 | Anti-PD-1 in Combination With Chemotherapy as First-Line Treatment to Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT05400915 | Two-Part, Phase Ib/II, Open Label, Single-Arm, Multi-center Study to Evaluate the Safety and Efficacy of Varlitinib in Combination With Weekly Paclitaxel in EGFR/HER2 Co-expressing Advanced or Metastatic Gastric Cancer Patients | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00004159 | Gemcitabine Plus Paclitaxel in Treating Patients With Advanced Non-small Cell Lung Cancer or Other Solid Tumor | lung cancer | Paclitaxel (NPC208553) | |
| NCT00001499 | Phase II Neoadjuvant Trial of a Continuous Infusion of Paclitaxel Plus Cisplatin Followed by Chest Radiotherapy for Patients With Stage III Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT04149015 | Neoadjuvant POF in the Treatment of Locally Advanced Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT01732640 | A Phase I/II Study Afatinib/Carboplatin/Paclitaxel Induction Chemotherapy In HPV-Negative HNSCC. | head and neck squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT01769391 | A Study of Necitumumab and Chemotherapy in Participants With Stage IV Squamous Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00041119 | Four Versus Six Cycles of Cyclophosphamide/Doxorubicin or Paclitaxel in Adjuvant Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00201734 | Capecitabine, Carboplatin and Weekly Paclitaxel for Patients With Solid Tumors and Adenocarcinoma of Unknown Primary | adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00542451 | Adjuvant Paclitaxel and Trastuzumab for Node-Negative HER2-Positive Breast Cancer | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT05422794 | Testing the Addition of Anti-Cancer Drug, ZEN003694 (ZEN-3694) and PD-1 Inhibitor (Pembrolizumab), to Standard Chemotherapy (Nab-Paclitaxel) Treatment in Patients With Advanced Triple-Negative Breast Cancer | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT03603756 | SHR-1210 in Combination With Apatinib and Chemotherapy in Patients With Advanced Esophageal Squamous Cell Cancer | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00004921 | High-Dose Chemotherapy Compared With Standard Chemotherapy in Treating Patients With Stage III or Stage IV Ovarian Epithelial Cancer That Has Been Removed During Surgery | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00616031 | Paclitaxel and Carboplatin With or Without Nitroglycerin in Treating Patients With Previously Untreated Stage III or Stage IV Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00005026 | Combination Chemotherapy in Treating Patients With Stage III or Stage IV Ovarian Epithelial or Primary Peritoneal Cancer | ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT00535119 | Veliparib, Carboplatin, and Paclitaxel in Treating Patients With Advanced Solid Cancer | Hereditary breast and ovarian cancer syndrome | Paclitaxel (NPC208553) | |
| NCT00479674 | Phase II Study With Abraxane, Bevacizumab and Carboplatin in Triple Negative Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00062062 | Gefitinib With or Without Carboplatin and Paclitaxel in Treating Older Patients With Unresectable or Metastatic Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT04258657 | Neoadjuvant Chomotherapy With Paclitaxel-albumin and S-1 for Advanced Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00807612 | QUILT-2.017: Phase 1b/2 Study of AMG 479 in Combination With Paclitaxel and Carboplatin for 1st Line Treatment of Advanced Squamous Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02506803 | Phase I Study of Neoadjuvant Chemotherapy for Patients With Borderline Resectable Pancreatic Cancer. | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00603538 | Study of CP-751,871 in Combination With Carboplatin and Paclitaxel in Advanced Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT04182724 | Camrelizumab, Apatinib and Nab-paclitaxel as Second-line Treatment in Advanced Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT01275677 | Chemotherapy With or Without Trastuzumab After Surgery in Treating Women With Invasive Breast Cancer | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT00181701 | Intraperitoneal Paclitaxel and Carboplatin in the Treatment of Women With Carcinoma of Mullerian Origin | ovarian carcinoma | Paclitaxel (NPC208553) | |
| NCT01146795 | Alisertib (MLN8237) in Participants With Ovarian, Fallopian Tube or Peritoneal Cancer Preceded by Phase 1 Study of MLN8237 Plus Paclitaxel Treatment of Ovary or Breast Cancer | fallopian tube cancer | Paclitaxel (NPC208553) | |
| NCT05095467 | HIPEC-AS in Patients With Peritoneal Metastasis of the Stomach or Esophagogastric Junction | stomach neoplasm | Paclitaxel (NPC208553) | |
| NCT00735696 | A Study of Ramucirumab (IMC-1121B) With Paclitaxel and Carboplatin in Non-small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00803556 | Clinical Trial of the Combination of Intravenous Alvespimycin (KOS-1022), Trastuzumab With or Without Paclitaxel in Patients With Advanced Solid Tumor Malignancies or Her2 Positive Metastatic Breast Cancer Who Have Previously Failed Trastuzumab Therapy | breast cancer | Paclitaxel (NPC208553) | |
| NCT00003742 | Combination Chemotherapy in Treating Patients With Locally Advanced or Metastatic Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT02481635 | A Study of Gemcitabine/Nab-paclitaxel and Radiation Therapy Followed by Surgery in Patients With Advanced Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT04608409 | Apatinib Combined With Abraxane and Carboplatin or Cisplatinum as First-line Treatment for Epithelial Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT01200342 | Genasense, Carboplatin, Paclitaxel (GCP) Combination in Uveal Melanoma | melanoma | Paclitaxel (NPC208553) | |
| NCT01820858 | The Efficacy and Safety of the Postoperative Adjuvant Treatment in Patients With High-risk Stage I Endometrial Carcinoma | endometrial neoplasm | Paclitaxel (NPC208553) | |
| NCT02392637 | Gemcitabine Hydrochloride, Cisplatin, and Nab-Paclitaxel in Treating Patients With Advanced or Metastatic Biliary Cancers | gallbladder cancer;intrahepatic cholangiocarcinoma | Paclitaxel (NPC208553) | |
| NCT00124943 | Use of Nanoparticle Paclitaxel (ABI-007) for the Prevention of In-Stent Restenosis | Coronary Restenosis | Paclitaxel (NPC208553) | |
| NCT00003377 | Radiation Therapy, Paclitaxel, and Cisplatin in Treating Patients With Cancer of the Cervix | cervical cancer | Paclitaxel (NPC208553) | |
| NCT03116152 | Study of IBI308 With Advanced/Metastatic Esophageal Squamous Cell Carcinoma After Failure of First-line Treatment | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00807859 | Safety Study of AMG 386 to Treat HER2-positive Locally Recurrent or Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00618826 | A Study of Biweekly Gemcitabine, Paclitaxel, and Avastin in Patients With Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00002679 | Adjuvant High-Dose, Sequential Chemotherapy in Treating Patients With Resected Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT01014351 | Study of Everolimus With Paclitaxel and Carboplatin in Patients With Metastatic Melanoma | metastatic melanoma | Paclitaxel (NPC208553) | |
| NCT00003394 | Combination Chemotherapy in Treating Patients With Advanced Prostate Cancer | prostate cancer | Paclitaxel (NPC208553) | |
| NCT01666418 | Pilot Study Investigating the Metabolic Activity and Transcriptional Profiling in Vivo in Tumor Biopsies in Melanoma Patients During Treatment With Pazopanib Alone and in Combination With Paclitaxel | melanoma | Paclitaxel (NPC208553) | |
| NCT02251951 | Study of Afuresertib Combined With Paclitaxel in Gastric Cancer | cancer | Paclitaxel (NPC208553) | |
| NCT05322720 | HR Positive, HER2 Negative Advanced Breast Cancer With Progression After Endocrine Therapy | neoplasm | Paclitaxel (NPC208553) | |
| NCT00795340 | Cediranib, Paclitaxel, and Carboplatin in Treating Patients With Stage IIIB or Stage IV Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00544232 | Preoperative Epirubicin Paclitaxel Aranesp Study (PREPARE) | breast cancer | Paclitaxel (NPC208553) | |
| NCT02250599 | Efficacy and Safety of TC+AVASTIN Versus TC in Patients With Metastatic Nasopharyngeal Carcinoma | nasopharyngeal neoplasm | Paclitaxel (NPC208553) | |
| NCT00004013 | Paclitaxel With or Without Trastuzumab Following Peripheral Stem Cell Transplantation in Treating Patients With Refractory Stage IV Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00003378 | Combination Chemotherapy in Treating Patients With Previously Untreated Stage III or Stage IV Ovarian or Primary Peritoneal Cancer | ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT02155088 | BYL719 in Combination With Gemcitabine and (Nab)-Paclitaxel in Locally Advanced and Metastatic Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00054392 | Combination Chemotherapy in Treating Patients With Stage IIIB, Stage IV, or Recurrent Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT03884101 | Pembrolizumab (MK-3475) Plus Lenvatinib (E7080/MK-7902) Versus Chemotherapy for Endometrial Carcinoma (ENGOT-en9 / MK-7902-001) | endometrial neoplasm | Paclitaxel (NPC208553) | |
| NCT05174156 | Study on the Biomarker of First-line Immunotherapy Combined With Chemotherapy in Advanced Esophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT03502343 | Efficacy and Safety of Modified Nab-Paclitaxel Plus Gemcitabine Chemotherapy for Metastatic Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT02297217 | Chemoradiotherapy for Advanced Esophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT03475615 | A Study of Intraperitoneal Paclitaxel in Combination With SOX Compared With SOX Alone in Gastric Cancer With Malignant Ascites | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00536939 | Trial of Paclitaxel, Bevacizumab, and Enzastaurin Versus Paclitaxel, Bevacizumab and Placebo for Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00873119 | Belinostat, Carboplatin and Paclitaxel (BelCaP) Compared to Carboplatin and Paclitaxel in Patients With Cancer of Unknown Primary | carcinoma | Paclitaxel (NPC208553) | |
| NCT03544567 | A Study of Oraxol in Subjects With Cutaneous Angiosarcoma | angiosarcoma | Paclitaxel (NPC208553) | |
| NCT05042128 | The ASCEND Study: Gemcitabine and Nab-Paclitaxel With CEND-1 or Placebo in Patients With Untreated Metastatic Pancreatic Ductal Adenocarcinoma | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT02038647 | Phase 2 Study of Alisertib (MLN8237) in Combination With Paclitaxel Versus Placebo in Combination With Paclitaxel as Second Line Therapy for Small Cell Lung Cancer (SCLC) | small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT04148911 | A Study of Atezolizumab Plus Nab-Paclitaxel in the Treatment of Unresectable Locally Advanced or Metastatic PD-L1-Positive Triple-Negative Breast Cancer | triple-negative breast cancer | Paclitaxel (NPC208553) | |
| NCT01383148 | Phase IIB/III Of TG4010 Immunotherapy In Patients With Stage IV Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00191256 | Efficacy Trial of Gemcitabine Containing Regimens As Preoperative Chemotherapy in Non Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT04602117 | ISPY-P1.01:Evaluating the Safety of Weekly Paclitaxel With Trastuzumab Duocarmazine (SYD985) in Patients With Metastatic Cancer | carcinoid tumor;endometrium neoplasm;gastric cancer;ovarian carcinoma;urinary bladder cancer | Paclitaxel (NPC208553) | |
| NCT00193206 | Neo-adjuvant Gemcitabine, Epirubicin, ABI-007 (GEA) in Locally Advanced or Inflammatory Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT04589234 | Saltikva for Metastatic Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT02210559 | A Study of Gemcitabine Plus Nab-paclitaxel With or Without FG-3019 in Participants With Locally Advanced, Unresectable Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00146549 | Trastuzumab in Combination With Vinorelbine or Taxane-Based Chemotherapy in Patients With Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT03723967 | FRAIL-IMMUNE (GORTEC 2018-03) - Combination of Durvalumab With Carboplatin/Paclitaxel | head and neck squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00003160 | Weekly Infusions of Paclitaxel in Treating Women With Stage III or Stage IV Ovarian Cancer Refractory to Paclitaxel and Platinum | fallopian tube cancer;ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT00369070 | A Phase 2 Trial of AMG 706 or Bevacizumab in Combination With Chemo for Advanced NSCLC | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00002888 | Combination Chemotherapy in Treating Patients With Advanced Head and Neck Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT05002686 | Safety and Efficacy of Sintilimab in Combination With Chemoradiothrapy Followed by D2 Surgical Resection in Patients With Advanced Gastric Cancer With Retroperitoneal Lymph Node Metastasis | gastric cancer | Paclitaxel (NPC208553) | |
| NCT02125344 | A Phase III Trial Comparing Two Dose-dense, Dose-intensified Approaches (ETC and PM(Cb)) for Neoadjuvant Treatment of Patients With High-risk Early Breast Cancer (GeparOcto) | inflammatory breast carcinoma | Paclitaxel (NPC208553) | |
| NCT02112552 | Paclitaxel and Intraperitoneal Carboplatin Followed by Radiation Therapy in Treating Patients With Stage IIIC-IV Uterine Cancer | uterine neoplasm | Paclitaxel (NPC208553) | |
| NCT00460317 | MONET1-MOtesanib NSCLC Efficacy and Tolerability Study | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00637897 | Paricalcitol and Chemotherapy in Treating Women With Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT05189730 | Selected Chemotherapy Combined Immunotherapy Treated High Risk Patient After NCRT in Resected Locally Advanced ESCC | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00003612 | Combination Chemotherapy and Trastuzumab in Treating Women With Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT03286244 | Second-line CM082 Combined With Paclitaxel For Patients With Advanced Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00006120 | Docetaxel or Paclitaxel in Treating Women With Unresectable Locally Advanced or Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00003413 | Combination Chemotherapy Plus Peripheral Stem Cell Transplantation in Treating Patients With Stage III or Stage IV Ovarian Epithelial Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00003881 | Trastuzumab Plus Chemotherapy in Treating Patients With Advanced Non-small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00003128 | Ifosfamide With or Without Paclitaxel in Treating Patients With Advanced, Refractory, or Recurrent Cancer of the Uterus | sarcoma | Paclitaxel (NPC208553) | |
| NCT01281150 | Veliparib in Combination With Carboplatin and Paclitaxel in Treating Patients With Locally Advanced or Metastatic Solid Tumors | male breast carcinoma | Paclitaxel (NPC208553) | |
| NCT00933426 | Lenalidomide and Paclitaxel in Prostate Cancer | prostate cancer | Paclitaxel (NPC208553) | |
| NCT01219777 | TRINOVA-1: A Study of AMG 386 or Placebo, in Combination With Weekly Paclitaxel Chemotherapy, as Treatment for Ovarian Cancer, Primary Peritoneal Cancer and Fallopian Tube Cancer | fallopian tube cancer;ovarian cancer | Paclitaxel (NPC208553) | |
| NCT04134468 | MRI Effects of Pegvorhyaluronidase Alfa (PEGPH20) in Pancreatic Ductal Adenocarcinoma | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT01402271 | A Study of MK-1775 in Combination With Paclitaxel and Carboplatin Versus Paclitaxel and Carboplatin Alone for Participants With Platinum-Sensitive Ovarian Tumors With the P53 Gene Mutation (MK-1775-004) | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00004863 | Paclitaxel and GEM 231 in Treating Patients With Recurrent or Refractory Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT02723331 | Perioperative Therapy for Resectable and Borderline-Resectable Pancreatic Adenocarcinoma With Molecular Correlates | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT04177108 | A Study Of Ipatasertib in Combination With Atezolizumab and Paclitaxel as a Treatment for Participants With Locally Advanced or Metastatic Triple-Negative Breast Cancer. | triple-negative breast cancer | Paclitaxel (NPC208553) | |
| NCT01649336 | A Phase IIclinical Trial of Carboplatin and Paclitaxel or Carboplatin and Gemcitabine in Platinum-sensitive, Recurrent Ovarian, Fallopian Tube, and Primary Peritoneal Cancer | fallopian tube cancer | Paclitaxel (NPC208553) | |
| NCT04027764 | Toripalimab Combined With S1 and Albumin Paclitaxel in Patients With Advanced Biliary Tract Cancer | biliary tract cancer | Paclitaxel (NPC208553) | |
| NCT00129922 | Fluorouracil, Epirubicin, and Cyclophosphamide Alone or Followed by Paclitaxel for Early Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT03328234 | Radiotherapy With or Without Concurrent Chemotherapy for Extensive Lymphatic Metastasis of Esophageal Cancer - 3JECROG P-03 | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00453323 | Paclitaxel and Capecitabine in Patients With Metastatic/Recurrence Esophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT01405235 | Study on Paclitaxel Plus Topotecan in Comparison With Topotecan Plus Cisplatin in Recurrent or Persistent Cervical Carcinoma | cervical cancer | Paclitaxel (NPC208553) | |
| NCT03563170 | QUILT-3.072: NANT Hepatocellular Carcinoma (HCC) Vaccine | hepatocellular carcinoma | Paclitaxel (NPC208553) | |
| NCT02861690 | Liposomal Paclitaxel Combined Nedaplatin in Treatment of Advanced or Recurrent Esophageal Carcinoma | esophageal carcinoma | Paclitaxel (NPC208553) | |
| NCT02048943 | Dovitinib Lactate, Gemcitabine Hydrochloride, and Paclitaxel Albumin-Stabilized Nanoparticle Formulation in Treating Patients With Advanced Solid Tumors or Pancreatic Cancer | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00657878 | Efficacy Study of Chemotherapy to Treat Ovarian Cancer Recurrence by Prolonging the Platinum Free Interval | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT01083537 | Study to Compare the Efficacy and Safety of Olaparib When Given in Combination With Carboplatin and Paclitaxel, Compared With Carboplatin and Paclitaxel in Patients With Advanced Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT04652076 | GYNecological Cancers Treated With NETrin mAbs in Combination With Chemotherapy and /or Pembrolizumab | cervical carcinoma;endometrial carcinoma | Paclitaxel (NPC208553) | |
| NCT01938846 | BI 860585 Dose Escalation Single Agent and in Combination With Exemestane or With Paclitaxel in Patients With Various Advanced and/or Metastatic Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT04883281 | A Prospective Study of Daily Adaptive Radiotherapy to Better Organ-at-Risk Doses in Head and Neck Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT00730353 | Sutent + Taxol for Advanced Esophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT02514031 | ARQ-761 Treatment With Gemcitabine/Nab-Paclitaxel Chemotherapy In Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00063999 | Doxorubicin Hydrochloride, Cisplatin, and Paclitaxel or Carboplatin and Paclitaxel in Treating Patients With Stage III-IV or Recurrent Endometrial Cancer | uterine neoplasm | Paclitaxel (NPC208553) | |
| NCT00559845 | A Study of Avastin (Bevacizumab) in Patients With Inflammatory or Locally Advanced Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT03990103 | S1+ Paclitaxel (IV&IP) + Bevacizumab (IP) Versus S1+Oxaliplatin as First-line Treatment in Gastric Cancer With Malignant Ascites | gastric adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT02446652 | Evaluation of TRANSKRIP ® Plus Chemotherapy in Recurrent-Persistent Cervical Cancer | cervical cancer | Paclitaxel (NPC208553) | |
| NCT02625623 | Avelumab in Third-Line Gastric Cancer (JAVELIN Gastric 300) | gastric adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00762034 | A Study of Pemetrexed, Carboplatin and Bevacizumab in Participants With Nonsquamous Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00686959 | Chemotherapy and Radiation in Treating Participants With Stage 3 Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00023907 | Paclitaxel in Treating Patients With Ovarian Epithelial Cancer or Primary Peritoneal Cancer | ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT00104676 | Combination Chemotherapy in Treating Patients With Stage II or Stage III Germ Cell Tumors | teratoma;testicular neoplasm | Paclitaxel (NPC208553) | |
| NCT02420314 | Pharmacological Ascorbate for Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00193596 | Gemcitabine/Irinotecan/ZD1839 vs Paclitaxel/Carboplatin/Etoposide/ZD1839 in Carcinoma of Unknown Primary Site | neoplasm | Paclitaxel (NPC208553) | |
| NCT00983424 | Cyclosporine A in Combination With Nab-Paclitaxel in Patients With Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00050960 | Evaluation of Efficacy, Safety and Tolerability of Targretin Capsules in Patients With Advanced or Metastatic Non-small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT04674956 | A Prospective, Randomized, Double-blinded, Multi-center Clinical Trial to Evaluate the Efficiency and Safety of Anti-PD1 Antibody (Camrelizumab) Combined With Paclitaxel(Albumin Bound) and Gemcitabine as First-line Therapy in Patients With Metastatic Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT01757288 | Phase I/II NabPaclitaxel, Paclitaxel&Carboplatin w RTX Followed by Consolidation in Patients w Favorable Prognosis Inoperable Stage IIIA/B NSCLC | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02039674 | A Study of Pembrolizumab (MK-3475) in Combination With Chemotherapy or Immunotherapy in Participants With Non-small Cell Lung Cancer (MK-3475-021/KEYNOTE-021) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02226757 | Paclitaxel-trastuzumab in EGFR-mutated NSCLC Patients | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02794571 | Safety and Pharmacokinetics (PK) of Escalating Doses of Tiragolumab as a Single Agent and in Combination With Atezolizumab and/or Other Anti-Cancer Therapies in Locally Advanced or Metastatic Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT00609791 | Paclitaxel Albumin-Stabilized Nanoparticle Formulation in Treating Patients of Different Ages With Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00997906 | Cisplatin and RT With or Without Gemcitabine, Carboplatin, and Paclitaxel in Treating Patients With Locally Advanced NPC | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT01039844 | Study of Weekly LOC-paclitaxel Injection for Melanoma | melanoma | Paclitaxel (NPC208553) | |
| NCT02312661 | Lower Dose Decitabine (DAC)-Primed TC (Carboplatin-Paclitaxel) Regimen in Ovary Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT01248403 | A Randomized, Double Blind Study Evaluating Paclitaxel With and Without RAD001 in Patients With Gastric Carcinoma After Prior Chemotherapy | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00005021 | Combination Chemotherapy With or Without Biological Therapy in Treating Patients With Refractory Solid Tumor or Lymphoma | lymphoma | Paclitaxel (NPC208553) | |
| NCT01205217 | Phase I/II Study of Lapatinib in Combination With Paclitaxel as 1L Chemotherapy for ErbB2-positive MBC | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT01756170 | Postoperative Chemoradiation v.s Radiotherapy for Lymph Node Negative Cervical Cancer Patients | cervical cancer | Paclitaxel (NPC208553) | |
| NCT01374620 | Weekly Paclitaxel and Cyclophosphamide in Metronomic Administration : Dose Escalation Study of Weekly Paclitaxel | cancer | Paclitaxel (NPC208553) | |
| NCT02551432 | Pembrolizumab and Paclitaxel in Refractory Small Cell Lung Cancer | small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT03977220 | Nab-paclitaxel Combined With S-1 Treating Diffuse Type of Stage Ⅲ Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00226915 | Trial of Tri-weekly TJ Versus Weekly TJ for Stage II-IV Mullerian Carcinoma | fallopian tube cancer;ovarian cancer | Paclitaxel (NPC208553) | |
| NCT03029611 | Two Therapeutic Strategies in First-line in Patients With Epithelial Advanced Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00540514 | Albumin-bound Paclitaxel (ABI-007) for Patients With Advanced Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT03564691 | Study of MK-4830 as Monotherapy and in Combination With Pembrolizumab (MK-3475) in Participants With Advanced Solid Tumors (MK-4830-001) | neoplasm | Paclitaxel (NPC208553) | |
| NCT05067530 | Study Assessing the Efficacy and Safety of Alpelisib + Nab-paclitaxel in Subjects With Advanced TNBC Who Carry Either a PIK3CA Mutation or Have PTEN Loss | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT01820754 | Evaluation of Circulating T Cells and Tumor Infiltrating Lymphocytes (TILs) During / After Pre-Surgery Chemotherapy in Non-Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01821859 | Bevacizumab Beyond Progression in Platinum Sensitive Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00193310 | Preoperative or Postoperative Therapy of Patients With Stages IB, II, IIIA Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT01739894 | Feasibility Study of Intraperitoneal Paclitaxel | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00093795 | Combination Chemotherapy in Treating Women Who Have Undergone Surgery for Node-Positive Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT01253681 | Study of Intraperitoneal Carboplatin With IV Paclitaxel and Bevacizumab in Untreated Ovarian Cancer | fallopian tube cancer | Paclitaxel (NPC208553) | |
| NCT04278287 | Chemoradiotherapy in Unresectable Esophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT01843829 | A Feasibility Study of Chemo-radiotherapy to Treat Operable Oesophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00979212 | Chemotherapy and Radiation Therapy With or Without Panitumumab in Treating Patients With Stage IIIA Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT03507491 | A Phase II Randomized Study Comparing the Efficacy and Safety of Targeted Therapy or Cancer Immunotherapy Versus Platinum-Based Chemotherapy in Patients With Cancer of Unknown Primary Site | cancer | Paclitaxel (NPC208553) | |
| NCT03697239 | High Dose Ascorbic Acid (AA) + Nanoparticle Paclitaxel Protein Bound + Cisplatin + Gemcitabine (AA NABPLAGEM) in Patients Who Have Metastatic Pancreatic Cancer | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT01696032 | Pertuzumab in Platinum-Resistant Low Human Epidermal Growth Factor Receptor 3 (HER3) Messenger Ribonucleic Acid (mRNA) Epithelial Ovarian Cancer (PENELOPE) | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT01953445 | A Study of Pertuzumab in Combination With Trastuzumab (Herceptin) and a Taxane in First-Line Treatment in Participants With Human Epidermal Growth Factor 2 (HER2)-Positive Advanced Breast Cancer | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT01419548 | ABT-888, Carboplatin, and Paclitaxel for Cancer With Liver or Kidney Problems | neoplasm | Paclitaxel (NPC208553) | |
| NCT01303926 | Quality of Life Comparison in Advanced Non-squamous Non Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02930902 | Pembrolizumab and Paricalcitol With or Without Chemotherapy in Patients With Pancreatic Cancer That Can Be Removed by Surgery | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00176254 | Paclitaxel, Carboplatin and Radiotherapy as Induction Therapy in Locally Advanced Head and Neck Cancer | squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00979082 | Glutathione in Preventing Peripheral Neuropathy Caused by Paclitaxel and Carboplatin in Patients With Ovarian Cancer, Fallopian Tube Cancer, and/or Primary Peritoneal Cancer | peripheral neuropathy | Paclitaxel (NPC208553) | |
| NCT03081143 | Ramucirumab Plus FOLFIRI Versus Ramucirumab Plus Paclitaxel in Patients With Advanced or Metastatic Gastric Cancer, Who Failed One Prior Line of Palliative Chemotherapy | gastric carcinoma | Paclitaxel (NPC208553) | |
| NCT00887575 | Neoadjuvant Sunitinib With Paclitaxel/Carboplatin in Patients With Triple-Negative Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT01632306 | A Study of LY2090314 and Chemotherapy in Participants With Metastatic Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT01822496 | Erlotinib Hydrochloride or Crizotinib and Chemoradiation Therapy in Treating Patients With Stage III Non-small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00154804 | CCRT With Twice Weekly Paclitaxel and Cisplatin Followed by Surgery for Locally Advanced Esophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT05322499 | Phase II Clinical Study of Camrelizumab Combined With Chemotherapy or Anlotinib in Advanced Esophageal Squamous Cell Cancer | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT04999969 | Safety, Pharmacokinetics and Clinical Activity of AZD0171 in Combination With Durvalumab and Chemotherapy in Locally Advanced or Metastatic Solid Tumours | neoplasm | Paclitaxel (NPC208553) | |
| NCT01138046 | A Phase II, Randomized, Open-label Study of Lapatinib Plus Chemotherapy Versus Trastuzumab Plus Chemotherapy in HER2-positive and p95HER2-positive Metastatic Breast Cancer | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT00466986 | Abraxane Plus Carboplatin for Recurrent Platinum-Sensitive Ovarian Cancer | fallopian tube cancer;ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00003622 | Vinorelbine Plus Paclitaxel in Treating Patients With Metastatic Prostate Cancer That Is Refractory to Hormone Therapy | prostate cancer | Paclitaxel (NPC208553) | |
| NCT00288041 | Bortezomib, Paclitaxel, and Carboplatin in Treating Patients With Metastatic Melanoma | melanoma | Paclitaxel (NPC208553) | |
| NCT00002913 | Paclitaxel, Cisplatin, and Topotecan With or Without Filgrastim in Treating Patients With Newly Diagnosed Stage III or Stage IV Epithelial Ovarian Cancer | ovarian serous cystadenocarcinoma;endometrioid carcinoma | Paclitaxel (NPC208553) | |
| NCT03884556 | TTX-030 Single Agent and in Combination With Immunotherapy or Chemotherapy for Patients With Advanced Cancers | lymphoma | Paclitaxel (NPC208553) | |
| NCT00891605 | Safety Study of ABT-263 in Combination With Paclitaxel in Subjects With Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT00038246 | Study of Paclitaxel, Estramustine Phosphate and Thalidomide for Patients With Metastatic Androgen-Independent Prostate Carcinoma | prostate cancer | Paclitaxel (NPC208553) | |
| NCT03240016 | Abraxane With Anti-PD1/PDL1 in Patients With Advanced Urothelial Cancer | urothelial carcinoma | Paclitaxel (NPC208553) | |
| NCT02172846 | Hypofractionated Proton Beam Radiation Therapy, Paclitaxel, and Carboplatin in Treating Patients With Stage II-III Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00110019 | Carboplatin and Paclitaxel With or Without Sorafenib Tosylate in Treating Patients With Stage III or Stage IV Melanoma That Cannot Be Removed by Surgery | cutaneous melanoma | Paclitaxel (NPC208553) | |
| NCT00933803 | Hydroxychloroquine, Carboplatin, Paclitaxel, and Bevacizumab in Recurrent Advanced Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00003386 | Vaccine Therapy in Treating Patients With Stage III or Stage IV Ovarian Epithelial Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT01711970 | SGI-110 in Combination With Carboplatin in Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT03601897 | A Phase 1b/2 Study of Rebastinib (DCC-2036) in Combination With Paclitaxel in Patients With Advanced or Metastatic Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT02762474 | Trial of Concurrent Chemoradiotherapy for Locally Advanced Esophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT01636622 | Study of Vemurafenib, Carboplatin, and Paclitaxel | cancer | Paclitaxel (NPC208553) | |
| NCT04858009 | Hyperthermic Intraperitoneal Chemotherapy for the Treatment of Pancreatic Cancer and Peritoneal Metastasis | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT04958785 | Study of Magrolimab Combination Therapy in Patients With Non-Surgically Removable Locally Advanced or Metastatic Triple-Negative Breast Cancer | triple-negative breast cancer | Paclitaxel (NPC208553) | |
| NCT00069953 | Combination Chemotherapy Followed By Chemoradiotherapy, With or Without Surgery, in Treating Patients With Resectable Locally Advanced Cancer of the Esophagus or Gastroesophageal Junction | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00016900 | PNU-93914 in Treating Patients With Locally Advanced or Metastatic Cancer of the Esophagus | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT03604965 | GP Induction Chemotherapy us TPF Adjuvant Chemotherapy Combined With DDP Concurrent Chemoradiotherapy in the Treatment of Locally Advanced NPC | nasopharyngeal neoplasm | Paclitaxel (NPC208553) | |
| NCT01165216 | Japanese Study of Ipilimumab Administered in Combination With Paclitaxel/Carboplatin in Patients With Nonsmall-cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT05459129 | A Study Evaluating Efficacy and Safety of Multiple Treatment Combinations in Patients With Locally Advanced Squamous Cell Carcinoma of the Head and Neck (Morpheus-Head and Neck Cancer) | head and neck squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT02834975 | Ruxolitinib Phosphate, Paclitaxel, and Carboplatin in Treating Patients With Stage III-IV Epithelial Ovarian, Fallopian Tube, or Primary Peritoneal Cancer | fallopian tube cancer | Paclitaxel (NPC208553) | |
| NCT03348904 | Nivolumab and Epacadostat With Platinum Doublet Chemotherapy Versus Platinum Doublet Chemotherapy in Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT05449366 | Intraperitoneal Paclitaxel for Patients With Primary Malignant Peritoneal Mesothelioma | malignant peritoneal mesothelioma | Paclitaxel (NPC208553) | |
| NCT00370552 | A Trial of 2 Schedules of Ixabepilone Plus Bevacizumab and Paclitaxel Plus Bevacizumab for Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT01199055 | CS-7017 in Combination With Carboplatin/Paclitaxel in Subjects With Stage IIIb/IV Non-small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT04481204 | New and Emerging Therapies for the Treatment of Resectable, Borderline Resectable, or Locally Advanced Pancreatic Cancer, PIONEER-Panc Study | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00003042 | Chemotherapy and Stem Cell Transplantation in Treating Patients With Stage IIIB Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT04794699 | Study of IDE397 in Participants With Solid Tumors Harboring MTAP Deletion | neoplasm | Paclitaxel (NPC208553) | |
| NCT02109445 | Study Of PF-03084014 In Combination With Gemcitabine And Nab-Paclitaxel In Patients With Metastatic Pancreatic Adenocarcinoma Not Previously Treated With Anticancer Therapies | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT02050009 | GANNET53: Ganetespib in Metastatic, p53-mutant, Platinum-resistant Ovarian Cancer | fallopian tube cancer | Paclitaxel (NPC208553) | |
| NCT00655850 | Lower Dose Chemotherapy Given More Frequent With Avastin to Treat Advanced Non-Squamous Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00100139 | Comparison of Liposome Entrapped Paclitaxel Easy to Use (LEP-ETU) and Taxol® Pharmacokinetics in Patients With Advanced Cancer | neoplasm | Paclitaxel (NPC208553) | |
| NCT03810872 | An Explorative Study of Afatinib in the Treatment of Advanced Cancer Carrying an EGFR, a HER2 or a HER3 Mutation | small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT04084158 | A Study of Toripalimab Combined With Concurrent Chemoradiotherapy for Locally Advanced Esophageal Squamous Cell Carcinoma. | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00004916 | Ifosfamide, Teniposide, and Paclitaxel in Treating Patients With Relapsed Non-Hodgkin's Lymphoma | lymphoma | Paclitaxel (NPC208553) | |
| NCT05312372 | S095033 in Combination With Paclitaxel as 2nd- or 3rd-line Treatment in Participants With Advanced or Metastatic ESCC | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT04563975 | Efficacy and Safety of Toripalimab Combined With Docetaxel or Nab-paclitaxel in Patients With Advanced Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00437749 | A Study of CBT-1 and Paclitaxel With Carboplatin in Patients With Advanced Inoperable Non-small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT03421353 | AZD9150 Plus Durvalumab Alone or in Combination With Chemotherapy in Patients With Advanced, Solid Tumours and in Patients With Non-Small-Cell Lung Cancer | neoplasm | Paclitaxel (NPC208553) | |
| NCT00691912 | Therapy of Metastatic Breast Cancer With Paclitaxel and Liposomal Doxorubicin | breast cancer | Paclitaxel (NPC208553) | |
| NCT00434252 | A Study of Bevacizumab With Carboplatin and Paclitaxel Chemotherapy for the First-Line Treatment of Patients With Metastatic Melanoma | melanoma | Paclitaxel (NPC208553) | |
| NCT01705483 | A Study of MK-8109 (Vintafolide) Given Alone or With Chemotherapy in Participants With Advanced Cancers (MK-8109-001) | cancer | Paclitaxel (NPC208553) | |
| NCT00005051 | Combination Chemotherapy in Treating Patients With Advanced Ovarian Epithelial Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00004924 | Paclitaxel and Irinotecan in Treating Patients With Advanced Non-small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT04225364 | Efficacy of Neoadjuvant PD-1 Blockade Plus Chemotherapy for Esophageal Squamous Cell Carcinoma | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT02440425 | A Study of CRLX101(NLG207) in Combination With Weekly Paclitaxel in Patients With Recurrent or Persistent Epithelial Ovarian, Fallopian Tube or Primary Peritoneal Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT04342910 | Study to Evaluate the Efficacy and Safety of Camrelizumab and Apatinib in Patients With GC/GEJC | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00008021 | Monoclonal Antibody Therapy, Chemotherapy, and Peripheral Stem Cell Transplantation in Treating Patients With Refractory Non-Hodgkin's Lymphoma | lymphoma | Paclitaxel (NPC208553) | |
| NCT01413750 | Carboplatin and Paclitaxel With or Without Vorinostat in Treating Patients With Advanced Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00049257 | Paclitaxel and Carboplatin in Treating Patients With Metastatic Prostate Cancer That Has Not Responded to Hormone Therapy | prostate cancer | Paclitaxel (NPC208553) | |
| NCT00006118 | Cisplatin, Paclitaxel, and Gemcitabine in Treating Patients With Progressive Unresectable Regional or Metastatic Bladder Cancer | urinary bladder cancer | Paclitaxel (NPC208553) | |
| NCT03322566 | A Study of Pembrolizumab Plus Epacadostat With Platinum-based Chemotherapy Versus Pembrolizumab Plus Platinum-based Chemotherapy Plus Placebo in Metastatic Non-Small Cell Lung Cancer (KEYNOTE-715-06/ECHO-306-06) | lung cancer | Paclitaxel (NPC208553) | |
| NCT03783442 | A Study of Tislelizumab (BGB-A317) in Combination With Chemotherapy as First Line Treatment in Participants With Advanced Esophageal Squamous Cell Carcinoma | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT04077255 | EGFR-targeted Therapy for Gastric Cancer | gastric adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00003644 | Carboplatin Plus Paclitaxel With or Without Continued Low-Dose Paclitaxel in Treating Patients With Early-Stage Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT03825328 | A Trial of NS/GEMOX Chemotherapy in Patients With Untreated Pancreatic Cancer ( HZ-NS/GEMOX-PC ) | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00482391 | Doxorubicin and Cyclophosphamide Followed by Paclitaxel, Trastuzumab, and Lapatinib in Treating Patients With HER2/Neu-Overexpressed Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00583830 | A Study of Mapatumumab in Combination With Paclitaxel and Carboplatin in Subjects With Non-small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT04921527 | A Phase III Study of BD0801 Combined With Chemotherapy in Recurrent, Platinum-resistant Epithelial Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT05025033 | Chemotherapy Combined With Apatinib and PD-1 Antibody | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00550537 | Proteomic Profiling in Predicting Response in Patients Receiving Erlotinib for Stage IIIB, Stage IV, or Recurrent Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT05189184 | Study of Camrelizumab in Combination With Apatinib Mesylate Plus Albumin-bound Paclitaxel and Cisplatin as the First Line Treatment of Recurrent or Metastatic, UnresectableHead and Neck Squamous Cell Carcinoma | head and neck squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00532857 | Phase II Study of Primary Chemotherapy With Paclitaxel, Gemcitabine, and Trastuzumab | breast cancer | Paclitaxel (NPC208553) | |
| NCT00652119 | Intraperitoneal Paclitaxel and Carboplatin With IV Avastin Therapy in Patients With Carcinomas of Mullerian Origin | fallopian tube cancer;ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT00653939 | A Safety and Efficacy Study of Carboplatin, Paclitaxel, Bevacizumab and CA4P in Non-Small Cell Lung Cancer | neoplasm | Paclitaxel (NPC208553) | |
| NCT00732836 | Hepatic Arterial Infusion (HAI) of Abraxane | liver cancer | Paclitaxel (NPC208553) | |
| NCT00002711 | Combination Chemotherapy and Radiation Therapy in Treating Patients With Cancer of the Esophagus | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00295893 | Combination Chemotherapy With or Without Trastuzumab in Treating Patients With Stage II or Stage III Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT05410847 | Nab-paclitaxel Plus Camrelizumab and S-1 Conversion Therapy for Gastric Cancer With Positive Exfoliative Cancer Cells | gastric cancer | Paclitaxel (NPC208553) | |
| NCT01650376 | A Study of MEK162 and Paclitaxel in Patients With Epithelial Ovarian, Fallopian Tube or Peritoneal Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT05081609 | A Study Evaluating Targeted Therapies in Participants Who Have Advanced Solid Tumors With Genomic Alterations or Protein Expression Patterns Predictive of Response | cancer | Paclitaxel (NPC208553) | |
| NCT01994031 | Dose Escalation and Pharmacokinetic Study of Paclitaxel Liposome Injection in Treating Patients With Advanced Solid Tumor After Failure From Conventional Treatments | neoplasm | Paclitaxel (NPC208553) | |
| NCT02301143 | Phase 2 Nab® -Paclitaxel (Abraxane®) Plus Gemcitabine in Subjects With Locally Advanced Pancreatic Cancer (LAPC) | pancreatic neoplasm | Paclitaxel (NPC208553) | |
| NCT02272738 | A Study of Chemoradiotherapy Using Gem Plus Nab-paclitaxel for Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT01196234 | Paclitaxel/Carboplatin (PC) Followed by Gefitinib Versus PC in Advanced Non-small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00005087 | Paclitaxel, Cisplatin, and Filgrastim Combined With Radiation Therapy in Treating Patients With Locally Recurrent Head and Neck Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT00832819 | E7080 in Combination With Carboplatin and Paclitaxel in Patients With Non-small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01454102 | Study of Nivolumab (BMS-936558) in Combination With Gemcitabine/Cisplatin, Pemetrexed/Cisplatin, Carboplatin/Paclitaxel, Bevacizumab Maintenance, Erlotinib, Ipilimumab or as Monotherapy in Subjects With Stage IIIB/IV Non-small Cell Lung Cancer (NSCLC) (CheckMate 012) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00770809 | Paclitaxel and Trastuzumab With or Without Lapatinib in Treating Patients With Stage II or Stage III Breast Cancer That Can Be Removed by Surgery | male breast carcinoma | Paclitaxel (NPC208553) | |
| NCT01057264 | HAI Abraxane With Gemcitabine and Bevacizumab | cancer | Paclitaxel (NPC208553) | |
| NCT01721746 | A Study to Compare BMS-936558 to the Physician's Choice of Either Dacarbazine or Carboplatin and Paclitaxel in Advanced Melanoma Patients That Have Progressed Following Anti-CTLA-4 Therapy (CheckMate 037) | metastatic melanoma | Paclitaxel (NPC208553) | |
| NCT00480831 | A Study of PRO95780 in Patients With Previously Untreated, Advanced-Stage Non-Small Cell Lung Cancer (APM4074g) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02979392 | Phase I Study of TENPA in Advanced Solid Cancer | neoplasm | Paclitaxel (NPC208553) | |
| NCT02725268 | A Study of Sapanisertib, Combination of Sapanisertib With MLN1117, Paclitaxel and Combination of Sapanisertib With Paclitaxel in Women With Endometrial Cancer | endometrial neoplasm | Paclitaxel (NPC208553) | |
| NCT00005847 | Chemotherapy With or Without Biological Therapy in Treating Patients With Metastatic Prostate Cancer That Has Not Responded to Hormone Therapy | prostate cancer | Paclitaxel (NPC208553) | |
| NCT00486954 | Lapatinib in Combination With Weekly Paclitaxel in Patients With ErbB2 Amplified Advanced Gastric Cancer | neoplasm | Paclitaxel (NPC208553) | |
| NCT01091428 | Alisertib (MLN8237) in Participants With Ovarian, Fallopian Tube or Peritoneal Cancer Preceded by Phase 1 Study of MLN8237 Plus Paclitaxel Treatment of Ovary or Breast Cancer | ovarian carcinoma;breast carcinoma | Paclitaxel (NPC208553) | |
| NCT02741856 | Study of Chemoradiotherapy in Oesophageal Cancer Including PET Response and Dose Escalation | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00002717 | Paclitaxel and Cisplatin in Treating Patients With Stage III or Stage IV Ovarian Cancer or Primary Peritoneal Cancer | ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT05281003 | Pembrolizumab Plus Chemo in Neoadjuvant Treatment of Esophageal Squamous Cell Carcinoma | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00672295 | PH I SRC Kinase, Dasatinib Combo Paclitaxel & Carboplatin in Pts w Ovarian, Peritoneal, & Tubal Cancer | fallopian tube cancer;ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT02259621 | Neoadjuvant Nivolumab, or Nivolumab in Combination With Ipilimumab, in Resectable NSCLC | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02040454 | Paclitaxel for the Treatment of Distal Radial Artery Arteriovenous Access Fistula Stenosis (PaciFIST-2) | stricture | Paclitaxel (NPC208553) | |
| NCT05193604 | A Clinical Trial to Evaluate Tolerability and Security of TQB2858 Injection in Subjects With Advanced Pancreatic Carcinoma | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00709761 | Neo ALTTO (Neoadjuvant Lapatinib and/or Trastuzumab Treatment Optimisation) Study | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT00667251 | Chemotherapy and Lapatinib or Trastuzumab in Treating Women With HER2/Neu-Positive Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00251472 | A Phase II Trial of Abraxane™ Given Weekly as a Single Agent in First-line Treatment of Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT05222204 | DDP ip Combined With AG in PDAC With Peritoneal Metastasis | pancreatic neoplasm | Paclitaxel (NPC208553) | |
| NCT00003992 | Chemotherapy Plus Monoclonal Antibody Therapy in Treating Women With Stage II or Stage IIIA Breast Cancer That Overexpresses HER2 | breast cancer | Paclitaxel (NPC208553) | |
| NCT01933061 | Study to Determine Efficacy and Safety of CC-486 With Nab-Paclitaxel Versus Nab-Paclitaxel in Patients With Chemotherapy naïve Metastatic Melanoma | metastatic melanoma | Paclitaxel (NPC208553) | |
| NCT00097227 | Trial of Carboplatin/Paclitaxel/Cetuximab in Stage IIIB/IV Non-Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00343083 | Evaluation of Cetuximab (ERBITUX) and Concurrent Carboplatin, Paclitaxel & Radiotherapy in the Management of Patients With Advanced Locoregional Squamous Cell Carcinomas of the Head and Neck (GCC 0442) | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT01852292 | Study of Efficacy and Safety of Buparlisib (BKM120) Plus Paclitaxel Versus Placebo Plus Paclitaxel in Recurrent or Metastatic Head and Neck Cancer Previously Pre-treated With a Platinum Therapy | head and neck squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00478361 | Gemcitabine, Paclitaxel, Doxorubicin in Metastatic or Unresectable Bladder Cancer With Decreased Kidney Function | urethra cancer;urinary bladder carcinoma | Paclitaxel (NPC208553) | |
| NCT03492047 | N-acetyl Cysteine Effect in Peripheral Neuropathy in Cancer Patients | peripheral neuropathy | Paclitaxel (NPC208553) | |
| NCT00789581 | A Randomized Trial of Ixempra Versus Taxol in Adjuvant Therapy of Triple Negative Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00002632 | Paclitaxel in Treating Patients With Metastatic or Recurrent Salivary Gland Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT00278122 | Paclitaxel and GM-CSF in Treating Patients With Stage III or Stage IV Melanoma That Cannot Be Removed By Surgery | cutaneous melanoma | Paclitaxel (NPC208553) | |
| NCT03574779 | A Study to Evaluate the Efficacy and Safety of Novel Treatment Combinations in Participants With Ovarian Cancer | ovarian neoplasm | Paclitaxel (NPC208553) | |
| NCT04698941 | Combined Simvastatin and Albumin Paclitaxel in Treating ES-SCLC Patients Relapsed From 1st Chemotherapy | small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01063517 | Efficacy Study of Olaparib With Paclitaxel Versus Paclitaxel in Gastric Cancer Patients | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00531687 | Trial of Paclitaxel, Gemcitabine and Cisplatin in Patients With Relapsing Germ Cell Cancer | testicular carcinoma | Paclitaxel (NPC208553) | |
| NCT00334802 | Combination Chemotherapy of Gemcitabine and Paclitaxel for Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00003379 | Radiation Therapy Plus Paclitaxel and Cisplatin in Treating Patients With Cervical Cancer | cervical cancer | Paclitaxel (NPC208553) | |
| NCT03693612 | GSK3359609 Plus Tremelimumab for the Treatment of Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT04135781 | Nab-paclitaxel Combined With S-1 as Adjuvant Chemotherapy for Stage Ⅲ Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00499083 | Paclitaxel, Cyclophosphamide, and Doxorubicin Followed by Autologous Dendritic Cells and Surgery With or Without Radiation Therapy and/or Hormone Therapy in Treating Women With Stage II or Stage III Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT02451956 | Study of AZD5363 in Combination With Paclitaxel, in Advanced Gastric Adenocarcinoma Patients Harboring PIK3CA Mutation and/or PIK3CA Amplification as a Second-line Chemotherapy | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00686322 | Concurrent Chemoradiotherapy (CCRT) With Paclitaxel Plus Cisplatin in LA Non-small-cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02047513 | Neoadjuvant Plus Adjuvant or Only Adjuvant Nab- Paclitaxel Plus Gemcitabine for Resectable Pancreatic Cancer | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT03410030 | Trial of Ascorbic Acid (AA) + Nanoparticle Paclitaxel Protein Bound + Cisplatin + Gemcitabine (AA NABPLAGEM) | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT01969578 | Androgen Deprivation Therapy in Advanced Salivary Gland Cancer | salivary gland cancer | Paclitaxel (NPC208553) | |
| NCT02756013 | Weight-based Dosing for Dense Weekly Paclitaxel and Carboplatin in Overweight Patients w/ Body Surface Area (BSA) > 2.0 | urogenital neoplasm | Paclitaxel (NPC208553) | |
| NCT03136055 | Pembrolizumab-based Therapy in Previously Treated High Grade Neuroendocrine Carcinomas | neuroendocrine carcinoma | Paclitaxel (NPC208553) | |
| NCT02252796 | Phase I Hypofractionated Stereotactic Boost (Radiotherapy) for Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT05298020 | Envafolimab Combined With Endostar and Chemotherapy for First-line Treatment of Advanced Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00483782 | Carboplatin and Paclitaxel With or Without Bevacizumab in Treating Patients With Newly Diagnosed Ovarian Epithelial Cancer, Fallopian Tube Cancer, or Primary Peritoneal Cavity Cancer | fallopian tube cancer;ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT00473720 | Phase I Study of the Combination of Satraplatin and Abraxane in Advanced Cancers | cancer | Paclitaxel (NPC208553) | |
| NCT00340184 | A Safety and Efficacy of CCRT With Paclitaxel/Carboplatin as Adjuvant Therapy to Post-operative Cervical Cancer Patients | cervical cancer | Paclitaxel (NPC208553) | |
| NCT00006469 | Combination Chemotherapy and Radiation Therapy Followed By Surgery in Treating Patients With Stage IIB or Stage IIIA Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT01912625 | Trametinib, Combination Chemotherapy, and Radiation Therapy in Treating Patients With Stage III Non-small Cell Lung Cancer That Cannot Be Removed by Surgery | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01372202 | CHFR Methylation Status Esophageal Cancer Study | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00003235 | Combination Chemotherapy Plus Radiation Therapy in Treating Patients With Stage III Non-small Cell Lung Cancer That Can Not Be Surgically Removed | lung cancer | Paclitaxel (NPC208553) | |
| NCT04472429 | Carboplatin-paclitaxel With Retifanlimab or Placebo in Participants With Locally Advanced or Metastatic Squamous Cell Anal Carcinoma (POD1UM-303/InterAACT 2). | squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00003392 | High-Dose Chemotherapy and Peripheral Stem Cell Transplantation in Treating Patients With Recurrent or Refractory Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00882973 | Trial to Determine the Maximum Tolerated Dose of Genexol-PM Plus Gemcitabine and Evaluate Efficacy and Safety of Genexol-PM Regimens in Subjects With Advanced Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00004093 | Combination Chemotherapy Plus Radiation Therapy in Treating Patients With Stage III Non-small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT02317874 | Testing the Addition of the Anti-Cancer Drug Talazoparib to the Combination of Carboplatin and Paclitaxel for the Treatment of Advanced Cancer | neoplasm | Paclitaxel (NPC208553) | |
| NCT02573493 | Nab-Paclitaxel and Cisplatin or Nab-paclitaxel as Induction Therapy for Locally Advanced Squamous Cell Carcinoma of the Head and Neck (HNSCC) | head and neck squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT02570893 | A Phase III Trail of Adjuvant Chemoradiotherapy Versus Adjuvant Radiotherapy in Esophageal Squamous Cell Carcinoma | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00003281 | Topotecan and Paclitaxel in Treating Patients With Recurrent or Refractory Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT01057342 | Paclitaxel, Carboplatin, and Dimethylxanthenone Acetic Acid in Treating Patients With Extensive-Stage Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00310011 | Gemcitabine, Paclitaxel, and Cisplatin in Treating Patients With Advanced Cancer of the Urothelium | urinary bladder cancer | Paclitaxel (NPC208553) | |
| NCT05383196 | Onvansertib + Paclitaxel In TNBC | inflammatory breast carcinoma | Paclitaxel (NPC208553) | |
| NCT03579784 | Biomarker-oriented Study of Durvalumab (MEDI4736) in Combination With Olaparib and Paclitaxel in Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT04060459 | Paclitaxel-binding Albumin and Cisplatin as Neoadjuvant Chemotherapy in Patients With Muscle-Invasive Bladder Cancer | urinary bladder cancer | Paclitaxel (NPC208553) | |
| NCT00733408 | Nab-Paclitaxel and Bevacizumab Followed By Bevacizumab and Erlotinib in Metastatic Breast Cancer | triple-negative breast cancer | Paclitaxel (NPC208553) | |
| NCT05283226 | Study to Evaluate the Safety and Efficacy of Oral NRC-2694-A in Combination With Paclitaxel in Patients With Recurrent and/or Metastatic Head and Neck Squamous Cell Carcinoma, Who Progressed on or After Immune Checkpoint Inhibitor Therapy | head and neck squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00003130 | Paclitaxel in Treating Women With Recurrent Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT02632305 | A Study to See the Effects That a New Combination of the Three Drugs, Nab-paclitaxel, Gemcitabine, and Cisplatin Has on Biliary Tract Cancer | biliary tract cancer | Paclitaxel (NPC208553) | |
| NCT02157870 | Neoadjuvant Chemotherapy in Epithelial Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00003317 | Combination Chemotherapy With or Without Radiation Therapy in Treating Patients With Stage IIIA Non-small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00004253 | Combination Chemotherapy and Radiation Therapy in Treating Patients With Stage II or Stage III Non-small Cell Lung Cancer That Cannot Be Removed By Surgery | lung cancer | Paclitaxel (NPC208553) | |
| NCT00077220 | Paclitaxel, Carboplatin, and Radiation Therapy With or Without Adjuvant Paclitaxel and Carboplatin in Treating Patients With Stage II or Stage III Unresectable Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT03935256 | Adjuvant Sequential & Concurrent CarboTaxol + Radiotherapy for High Risk Endometrial Cancer | endometrial cancer | Paclitaxel (NPC208553) | |
| NCT02013453 | A Phase II Trial of Proton Pump Inhibitors With Chemotherapy in Patients With Metastatic Head and Neck Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT00002627 | Addition of Paclitaxel to High-Dose Combination Chemotherapy in Treating Patients With High-Risk Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00087802 | Gemcitabine/Oxaliplatin (GEMOX) vs Carboplatin/Paclitaxel (CP) in Non-Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00550771 | Lapatinib +/- Trastuzumab In Addition To Standard Neoadjuvant Breast Cancer Therapy. | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT01488552 | Gemcitabine+Nab-paclitaxel and FOLFIRINOX and Molecular Profiling for Patients With Advanced Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT02412722 | Study of REGN2810 (Anti-PD-1) in Patients With Advanced Malignancies | cancer | Paclitaxel (NPC208553) | |
| NCT02350517 | Endostar Combined With Paclitaxel and Nedaplatin in Treating Patients With Recurrent or Metastatic Esophageal Cancer | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00004067 | Doxorubicin and Cyclophosphamide Plus Paclitaxel With or Without Trastuzumab in Treating Women With Node-Positive Breast Cancer That Overexpresses HER2 | breast cancer | Paclitaxel (NPC208553) | |
| NCT00020449 | Liposomal Doxorubicin and Interleukin-12 in Treating Patients With AIDS-Related Kaposi's Sarcoma | sarcoma | Paclitaxel (NPC208553) | |
| NCT00256243 | Neoadjuvant Biweekly Treatment Followed by Weekly Treatment of Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00544648 | Ph I/II Nab-Paclitaxel & Carboplatin w/Concurrent Radiation Therapy for Unresectable Stg III NSCLC | lung cancer | Paclitaxel (NPC208553) | |
| NCT00390611 | Paclitaxel and Carboplatin With Or Without Sorafenib In The First-Line Treatment Of Patients With Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT01285609 | Phase 3 Trial in Squamous Non Small Cell Lung Cancer Subjects Comparing Ipilimumab Versus Placebo in Addition to Paclitaxel and Carboplatin | lung cancer | Paclitaxel (NPC208553) | |
| NCT00016276 | Combination Chemotherapy, Surgery, and Radiation Therapy With or Without Dexrazoxane and Trastuzumab in Treating Women With Stage III or Stage IV Breast Cancer | inflammatory breast carcinoma | Paclitaxel (NPC208553) | |
| NCT03600623 | Folfirinox or Gemcitabine-Nab Paclitaxel Followed by Stereotactic Body Radiotherapy for Locally Advanced Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT04982237 | A Study of AK104 Plus Platinum-containing Chemotherapy±Bevacizumab as First-line Treatment for Persistent, Recurrent, or Metastatic Cervical Cancer | cervical cancer | Paclitaxel (NPC208553) | |
| NCT04850235 | Nab-paclitaxel Based TPX Neoadjuvant Chemotherapy for NPC Patients: a Dose-escalation Study | nasopharyngeal neoplasm | Paclitaxel (NPC208553) | |
| NCT03539328 | Study on Decitabine Plus Carboplatin Versus Physician's Choice Chemotherapy in Recurrent, Platinum-resistant Ovarian Cancer. | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00337376 | A Study of the mTOR Inhibitor Rapamycin (Rapamune, Sirolimus) in Combination With Abraxane in Advanced Solid Cancers | cancer | Paclitaxel (NPC208553) | |
| NCT00002852 | Surgery With or Without Chemotherapy in Treating Patients With Stage I Non-small Cell Lung Cancer | lung adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00499525 | Phase 2b Study of Taxol Plus Sorafenib or Placebo in Patients With Advanced Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT01146795 | Trial of Best Supportive Care and Either Cisplatin or Paclitaxel to Treat Patients With Primary Ovarian Cancer, Primary Peritoneal Cancer or Fallopian Tube Cancer and Inoperable Malignant Bowel Obstruction | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT01612351 | Multimodality Risk Adapted Tx Including Induction Chemo for SCCHN Amenable to Transoral Surgery | head and neck squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT02453282 | Phase III Open Label First Line Therapy Study of MEDI 4736 (Durvalumab) With or Without Tremelimumab Versus SOC in Non Small-Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT04016142 | Carboplatin-Paclitaxel Adjuvant Chemotherapy in the Treatment of Locally Advanced Cervical Cancer | cervical cancer | Paclitaxel (NPC208553) | |
| NCT03292822 | Effects of (Licochalcone A) and Paclitaxel on Human Oral Squamous Cell Carcinoma Cell Line | squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00809133 | Trial Exploring Afatinib (BIBW 2992) + Paclitaxel (Part A), Afatinib + Paclitaxel + Bevacizumab (Part B), Afatinib + Carboplatin (Part C) and Afatinib+ Paclitaxel +Carboplatin(Part D) in Patients With Advanced Solid Tumours | neoplasm | Paclitaxel (NPC208553) | |
| NCT02083679 | Sym004 in Subjects With Stage IV Non-small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02289456 | Phase II Safety and Tolerability Trial With Nab-Paclitaxel Plus Carboplatin Followed by Nab-Paclitaxel for First Line Treatment of NSCLC Subjects With ECOG PS 2 | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00042302 | A Double-Blind, Randomized, Placebo-Controlled, Multicenter, Phase III Study of Tariquidar + Paclitaxel/Carboplatin as First-Line Therapy in Non-Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT04046887 | Study of Lonsurf in Combination With Gemcitabine and Nab-Paclitaxel in Patients With Advanced (PDAC) | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT01431794 | Gemcitabine + Nab-paclitaxel With LDE-225 (Hedgehog Inhibitor) as Neoadjuvant Therapy for Pancreatic Adenocarcinoma | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT04004234 | A Phase I/II Study of the Pan-immunotherapy in Patients With Local Advanced/Metastatic BTC | biliary tract cancer | Paclitaxel (NPC208553) | |
| NCT03487016 | First-line Therapy in Metastatic PDAC | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00069901 | Phase II CT-2103/Carboplatin in Ovarian Cancer | ovarian neoplasm | Paclitaxel (NPC208553) | |
| NCT01821859 | A Study of MEK162 and Paclitaxel in Patients With Epithelial Ovarian, Fallopian Tube or Peritoneal Cancer | peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT02978495 | Neoadjuvant Carboplatin in Triple Negative Breast Cancer | Hereditary breast and ovarian cancer syndrome | Paclitaxel (NPC208553) | |
| NCT02672475 | Galunisertib and Paclitaxel in Treating Patients With Metastatic Androgen Receptor Negative (AR-) Triple Negative Breast Cancer | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT00666991 | Pharmacokinetic, Safety and Efficacy Study of Nanoparticle Paclitaxel in Patients With Peritoneal Cancers | peritoneal neoplasm | Paclitaxel (NPC208553) | |
| NCT01159418 | LBH589 Oral in Combination With Carboplatin and Paclitaxel in Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT00003691 | Combination Chemotherapy With or Without G-CSF in Treating Patients With Stage III, Stage IV, or Recurrent Endometrial Cancer | endometrial cancer | Paclitaxel (NPC208553) | |
| NCT04241276 | Phase IIb Randomised Trial of ATRA in a Novel Drug Combination for Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00533936 | Trastuzumab or Observation After Combination Chemotherapy and Trastuzumab in Treating Patients Undergoing Surgery for Stage II or Stage III Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT04286711 | Clinical Study of Albumin-paclitaxel Combined With Apatinib and Camrelizumab in Advanced Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT02039791 | Study of Nimotuzumab in Combination With Neoadjuvant Chemotherapy for Cervical Cancer | cervical cancer | Paclitaxel (NPC208553) | |
| NCT00622466 | Sorafenib and Paclitaxel in Treating Patients With Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT01924533 | Efficacy and Safety Study of Olaparib in Combination With Paclitaxel to Treat Advanced Gastric Cancer. | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00373620 | A Safety and Efficacy of CCRT With Paclitaxel as Adjuvant Therapy to Post-Operative Advanced Endometrial Cancer Patients | endometrial cancer | Paclitaxel (NPC208553) | |
| NCT00024414 | DHA-Paclitaxel in Treating Patients With Metastatic Prostate Cancer | prostate cancer | Paclitaxel (NPC208553) | |
| NCT00003717 | Paclitaxel Plus Estramustine in Treating Patients With Metastatic Prostate Cancer | prostate cancer | Paclitaxel (NPC208553) | |
| NCT00011921 | Chemotherapy Followed by Peripheral Stem Cell or Bone Marrow Transplant Compared With Chemotherapy Alone in Treating Patients With Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00009750 | Monoclonal Antibody Therapy Plus Chemotherapy Followed by Peripheral Stem Cell Transplantation in Treating Patients With Metastatic Prostate Cancer That Has Not Responded to Hormone Therapy | prostate cancer | Paclitaxel (NPC208553) | |
| NCT01496742 | A Study of Onartuzumab (MetMAb) in Combination With Bevacizumab (Avastin) Plus Platinum And Paclitaxel or With Pemetrexed Plus Platinum in Patients With Non-Squamous Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02834975 | Phase II: Pembrolizumab/Carboplatin/Taxol in Epithelial Ovary Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00034190 | Safety and Efficacy of S-8184 in Second Line Treatment of Stage III or IV Colorectal Adenocarcinoma | colorectal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT04764227 | Phase II Study of Postoperative Concurrent Chemoradiotherapy for Esophageal Squamous Cell Carcinoma (ESO- Shanghai 17) | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT02162719 | Safety and Efficacy Study of Vintafolide and Vintafolide Plus Paclitaxel Compared to Paclitaxel Alone in Participants With Triple Negative Breast Cancer (TNBC) (MK-8109-004) | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT00900627 | Phase I/II AZD8931/Paclitaxel in Treatment of Advanced Solid Tumours (Phase I) and Advanced Breast Cancer (Phase II) | breast cancer | Paclitaxel (NPC208553) | |
| NCT00944047 | Evaluate Trastuzumab Plus Standard Chemotherapy Given Before Surgery in Breast Cancer Patients With Low HER 2 Expression | breast cancer | Paclitaxel (NPC208553) | |
| NCT04729387 | Trial on NIraparib-TSR-042 (Dostarlimab) vs Physician's Choice CHEmotherapy in Recurrent, Ovarian, Fallopian Tube or Primary Peritoneal Cancer Patients Not Candidate for Platinum Retreatment | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT01279291 | Study of Anti-HB-EGF Antibody KHK2866 in Subjects With Advanced Solid Tumors and Ovarian Cancer | Fallopian Tube Carcinoma;ovarian neoplasm;peritoneal neoplasm | Paclitaxel (NPC208553) | |
| NCT03197935 | A Study to Investigate Atezolizumab and Chemotherapy Compared With Placebo and Chemotherapy in the Neoadjuvant Setting in Participants With Early Stage Triple Negative Breast Cancer | triple-negative breast cancer | Paclitaxel (NPC208553) | |
| NCT02262897 | The Efficacy and Safety of Nab-paclitaxel in Pretreated Patients With Extensive Disease of Small Cell Lung Cancer | small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00011999 | Chemotherapy and Radiation Therapy Following Surgery in Treating Patients With Head and Neck Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT00750386 | An Efficacy and Safety Study of MORAb-003 in Platinum-Resistant or Refractory Relapsed Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT04035473 | A Study to Determine the Bioequivalence of Oraxol in Cancer Patients Treated With Intravenous Paclitaxel | neoplasm | Paclitaxel (NPC208553) | |
| NCT00004859 | Carboplatin, Paclitaxel, and Radiation Therapy With or Without Thalidomide in Patients With Stage III Non-small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT01730833 | Pertuzumab, Trastuzumab, and Paclitaxel Albumin-Stabilized Nanoparticle Formulation in Treating Patients With HER2-Positive Advanced Breast Cancer | inflammatory breast carcinoma | Paclitaxel (NPC208553) | |
| NCT00001426 | A Multi-Institutional Phase II Study of Cyclophosphamide, Paclitaxel, Cisplatin With G-CSF for Patients With Newly Diagnosed Advanced Stage Ovarian Cancer | ovarian neoplasm | Paclitaxel (NPC208553) | |
| NCT00016913 | Chemotherapy, Hormone Therapy, and Radiation Therapy in Treating Patients With Locally Advanced Prostate Cancer | prostate cancer | Paclitaxel (NPC208553) | |
| NCT04004871 | Induction Chemotherapy With Nab-paclitaxel, Cisplatin and Fluorouracil for Locoregionally Advanced Nasopharyngeal Carcinoma | nasopharyngeal neoplasm | Paclitaxel (NPC208553) | |
| NCT00655876 | Paclitaxel, Cisplatin, and Radiation Therapy With or Without Cetuximab in Treating Patients With Locally Advanced Esophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT01402271 | Alisertib (MLN8237) in Participants With Ovarian, Fallopian Tube or Peritoneal Cancer Preceded by Phase 1 Study of MLN8237 Plus Paclitaxel Treatment of Ovary or Breast Cancer | peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT01872403 | Neoadjuvant Chemotherapy of Nanoparticle Albumin-bound Paclitaxel/Carboplatin vs. Paclitaxel /Carboplatin in Stage Ⅱ B and IIIA Squamous Cell Carcinoma of the Lung | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00768131 | A Study to Determine Whether EGFR Status by FISH Can Predict Results in Non Small Cell Lung Cancer (NSCLC) Patients Treated With Cetuximab, Carboplatin and Paclitaxel | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00003084 | Combination Chemotherapy With Ketoconazole in Treating Patients With Prostate Cancer | prostate cancer | Paclitaxel (NPC208553) | |
| NCT00635050 | Therapy for Locally Advanced Breast Cancer Using Doxil, Paclitaxel, and Cyclophosphamide With Avastin | breast cancer | Paclitaxel (NPC208553) | |
| NCT00022217 | Cisplatin-Epinephrine Injectable Gel Plus Paclitaxel and Carboplatin in Treating Patients With Recurrent Head and Neck Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT02272790 | Metformin Hydrochloride, Carboplatin, and Paclitaxel in Treating Patients With Recurrent Ovarian, Fallopian Tube, or Primary Peritoneal Cancer | peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT00071188 | ZD6474 Alone or in Combination With Paclitaxel and Carboplatin in Subjects With Previously Untreated Locally Advanced or Metastatic Non-small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00989651 | Carboplatin, Paclitaxel, Bevacizumab, and Veliparib in Treating Patients With Newly Diagnosed Stage II-IV Ovarian Epithelial, Fallopian Tube, or Primary Peritoneal Cancer | endometrioid carcinoma;undifferentiated carcinoma | Paclitaxel (NPC208553) | |
| NCT03316599 | Study of Gemcitabine, Nab-paclitaxel, and Ficlatuzumab (AV-299) in Patients With Advanced Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00003587 | S9806: Combination Chemotherapy in Treating Patients With Stage IIIB or Stage IV Non-small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00840450 | Two Different Schedules of Carboplatin, Paclitaxel, Gemcitabine, and Surgery in Treating Patients With Newly Diagnosed Stage IIIC or Stage IV Primary Epithelial Ovarian Cancer, Fallopian Tube Cancer, or Primary Peritoneal Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT03964753 | Neoadjuvant Therapy With Nab-paclitaxel and Cisplatin for Locally Advanced Esophageal Squamous Cell Carcinoma | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00434356 | A Study of Sunitinib in Combination With Bevacizumab and Paclitaxel in Previously Untreated Patients With Metastatic Breast Cancer (SABRE-B) | breast cancer | Paclitaxel (NPC208553) | |
| NCT00136539 | Neoadjuvant Therapy With Herceptin and Taxol for Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00508326 | Paclitaxel Administered by HAI to Patients With Advanced Cancer and Dominant Liver Involvement | cancer | Paclitaxel (NPC208553) | |
| NCT04694183 | The Conversion Therapy of Chemotherapy Plus Camrelizumab in Metastatic Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT02730416 | Combination Chemotherapy With Nintedanib / Placebo in Endometrial Cancer | endometrial cancer | Paclitaxel (NPC208553) | |
| NCT04561817 | To Evaluate the Safety and Efficacy of Ipatasertib (GDC-0068) in Combination With Paclitaxel in Platinum-resistant Recurrent Epithelial Ovarian Cancer | ovarian neoplasm | Paclitaxel (NPC208553) | |
| NCT02041533 | An Open-Label, Randomized, Phase 3 Trial of Nivolumab Versus Investigator's Choice Chemotherapy as First-Line Therapy for Stage IV or Recurrent PD-L1+ Non-Small Cell Lung Cancer (CheckMate 026) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00533949 | High-Dose or Standard-Dose Radiation Therapy and Chemotherapy With or Without Cetuximab in Treating Patients With Newly Diagnosed Stage III Non-Small Cell Lung Cancer That Cannot Be Removed by Surgery | lung cancer | Paclitaxel (NPC208553) | |
| NCT02311907 | Glutathione in Preventing Peripheral Neuropathy Caused by Paclitaxel and Carboplatin in Patients With Ovarian Cancer, Fallopian Tube Cancer, and/or Primary Peritoneal Cancer | neuropathy;pain | Paclitaxel (NPC208553) | |
| NCT01739218 | A Phase I/II Trial of VB-111 and Paclitaxel for Recurrent Platinum-Resistant Müllerian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00846027 | A Study of Avastin (Bevacizumab) in Combination With Taxane-based Chemotherapy as First Line Treatment in Patients With HER-2 Negative Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT02182245 | Dose Escalation Study of BIBF 1120 in Patients With Advanced Gynaecological Malignancies | Genital neoplasm, female | Paclitaxel (NPC208553) | |
| NCT00003322 | Combination Chemotherapy in Treating Patients With Primary Peritoneal or Stage III Epithelial Ovarian Cancer | ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT03400306 | A Study Evaluating the Bioavailability and Food Effect of Veliparib Tablets Followed by an Extension in Subjects With Ovarian Cancer | ovarian carcinoma | Paclitaxel (NPC208553) | |
| NCT00093119 | Trial of ABI-007 in Previously Treated Patients With Metastatic Melanoma | melanoma | Paclitaxel (NPC208553) | |
| NCT03885219 | Nab-paclitaxel and S-1 in Patients With Locally Advanced Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00768859 | Trastuzumab in a Neo-adjuvant Regimen for HER2+ Breast Cancer - the TRAIN Study | breast cancer | Paclitaxel (NPC208553) | |
| NCT01470417 | Gemcitabine With Abraxane and Other Investigational Therapies in Neoadjuvant Treatment of Pancreatic Adenocarcinoma | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT02715531 | A Study of the Safety and Efficacy of Atezolizumab Administered in Combination With Bevacizumab and/or Other Treatments in Participants With Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT03866993 | A Study of Anti-PD-1 AK105 in Patients With Metastatic Squamous Non-small Cell Lung Cancer | squamous cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT03336216 | A Study of Cabiralizumab Given With Nivolumab With and Without Chemotherapy in Patients With Advanced Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT03126812 | Pembrolizumab, Paclitaxel, and Carboplatin in Patients With Advanced Stage Epithelial Ovarian Cancer (EOC). | peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT01435018 | Three Chemo Regimens as an Adjunct to ART for Treatment of Advanced AIDS-KS | HIV-1 infection | Paclitaxel (NPC208553) | |
| NCT00001383 | A Phase I Study of Infusional Paclitaxel With the P-Glycoprotein Antagonist PSC 833 | renal cell carcinoma;lymphoma;ovarian cancer;breast cancer | Paclitaxel (NPC208553) | |
| NCT04974944 | First-line Treatment With Camrelizumab + Apatinib Versus Chemotherapy + Bevacizumab in Advanced Cervical Cancer | cervical cancer | Paclitaxel (NPC208553) | |
| NCT02315196 | Pegylated Liposomal Doxorubicin Hydrochloride and Carboplatin Followed by Surgery and Paclitaxel in Treating Patients With Triple Negative Stage II-III Breast Cancer | triple-negative breast cancer | Paclitaxel (NPC208553) | |
| NCT04006041 | Combination of Toripalimab and Neoadjuvant Chemoradiotherapy in Esophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00006027 | Comparison of Radiation Therapy With or Without Combination Chemotherapy Following Surgery in Treating Patients With Stage I or Stage II Endometrial Cancer | endometrial cancer | Paclitaxel (NPC208553) | |
| NCT00453167 | Weekly Paclitaxel Plus Gemcitabine as Second-line in Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00144053 | A Phase Ⅲ Randomized Study of Mitomycin/Vindesine/Cisplatin Versus Irinotecan/Carboplatin Versus Paclitaxel/Carboplatin With Concurrent Thoracic Radiotherapy for Unresectable Stage Ⅲ Non-Small-Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00194753 | Adjuvant Therapy for High-Risk Breast Cancer With Wkly Adriamycin & Oral Cytoxan With G-CSF for 12 Wks | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT01889420 | Phase I Trial of Everolimus, Pomalidomide and Dexamethasone in Patients With Relapsed/Refractory Multiple Myeloma | multiple myeloma | Paclitaxel (NPC208553) | |
| NCT03117933 | Ribociclib (Ribociclib (LEE-011)) With Platinum-based Chemotherapy in Recurrent Platinum Sensitive Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00388076 | A Study Of SU011248 Plus Paclitaxel Versus Bevacizumab Plus Paclitaxel In Patients With Advanced Breast Cancer | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT03038100 | IGFBP-2 Vaccine and Combination Chemotherapy in Treating Patients With Stage III-IV Ovarian, Fallopian Tube, or Primary Peritoneal Cancer Undergoing Surgery | fallopian tube cancer;ovarian cancer | Paclitaxel (NPC208553) | |
| NCT02186847 | Chemotherapy and Radiation Therapy With or Without Metformin Hydrochloride in Treating Patients With Stage III Non-small Cell Lung Cancer | bronchoalveolar adenocarcinoma;large cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00970996 | Cisplatin, Temozolomide, Abraxane, With Interleukin-2 and Interferon for Metastatic Melanoma | melanoma | Paclitaxel (NPC208553) | |
| NCT04644250 | Combination of Toripalimab and Neoadjuvant Chemoradiotherapy for Locally Advanced Esophageal Squamous Cell Carcinoma | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT03345810 | Durvalumab (MEDI4736) in Frail and Elder Patients With Metastatic NSCLC (DURATION) | large cell lung carcinoma;lung adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT01362374 | Safety and Clinical Pharmacology of GDC-0068 in Combination With Docetaxel, Fluoropyrimidine Plus Oxaliplatin, Paclitaxel, or Enzalutamide in Participants With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT00607438 | A Phase II Study Of Abraxane and Nexavar in the First-Line Treatment of Locally Advanced or Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT02976909 | Efficacy and Safety of TPF Induction Chemotherapy for Borderline-resectable Esophageal Squamous Cell Carcinoma | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00003298 | Chemotherapy, Surgery, and Radiation Therapy in Treating Patients With Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00294762 | Erlotinib (Tarceva) as a Single Agent or Intercalated With Combination Chemotherapy in Patients With EGFR Positive NSCLC | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00020007 | Paclitaxel and Hyperthermic Perfusion in Treating Patients With Lung Cancer or Lung Metastases That Cannot Be Removed By Surgery | lung cancer | Paclitaxel (NPC208553) | |
| NCT00702299 | Alimta® Plus Cisplatin & Paclitaxel Given Intraperitonelly | fallopian tube cancer;ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT03766607 | Trastuzumab Beyond Progression in HER2 Positive Metastatic Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00466960 | Sargramostim and Paclitaxel Albumin-Stabilized Nanoparticle Formulation in Treating Patients With Advanced Ovarian Cancer, Fallopian Tube Cancer, or Primary Peritoneal Cancer That Did Not Respond to Previous Chemotherapy | ovarian serous cystadenocarcinoma;endometrioid carcinoma;fallopian tube cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT00024401 | DHA-Paclitaxel in Treating Patients With Metastatic Colorectal Cancer | colorectal carcinoma | Paclitaxel (NPC208553) | |
| NCT00073723 | Trial of ABI-007 Administered Weekly in Chemotherapy Naive Patients With Advanced Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01493505 | EP-100 Plus Paclitaxel Versus Paclitaxel Alone in Patients With Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00002969 | S9714: Paclitaxel in Treating Patients With Stage IIIB or Stage IV Non-small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00350025 | Study of Cetuximab With and Without Weekly Paclitaxel for Patients With Previously Treated Advanced Urothelial Cancer | neoplasm | Paclitaxel (NPC208553) | |
| NCT03430882 | Sapanisertib, Carboplatin, and Paclitaxel in Treating Patients With Recurrent or Refractory Malignant Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT00085501 | S0342: Paclitaxel, Carboplatin, and Cetuximab in Treating Patients With Stage IIIB or Stage IV Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT01386385 | Veliparib With or Without Radiation Therapy, Carboplatin, and Paclitaxel in Patients With Stage III Non-small Cell Lung Cancer That Cannot Be Removed by Surgery | squamous cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00513383 | Panitumumab Chemoradiotherapy Chemotherapy for Squamous Cancer of the Head and Neck | head and neck squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT04548440 | Efficacy and Safety of Preoperative Sintilimab Plus Nab-paclitaxel and Cisplatin in BR-ESCC Patients | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00004054 | Hormone Therapy Plus Radiation Therapy With or Without Combination Chemotherapy in Treating Patients With Prostate Cancer | prostate cancer | Paclitaxel (NPC208553) | |
| NCT00626405 | Bevacizumab and Temozolomide or Bevacizumab and Paclitaxel Albumin-Stabilized Nanoparticle Formulation and Carboplatin in Treating Patients With Stage IV Malignant Melanoma That Cannot Be Removed by Surgery | cutaneous melanoma | Paclitaxel (NPC208553) | |
| NCT03887442 | Paclitaxel vs Paclitaxel + Cetuximab in Recurrent - Metastatic Head & Neck Carcinoma After Failure of a 1º Chemotherapy | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT02367794 | A Study of Atezolizumab in Combination With Carboplatin + Paclitaxel or Carboplatin + Nab-Paclitaxel Compared With Carboplatin + Nab-Paclitaxel in Participants With Stage IV Squamous Non-Small Cell Lung Cancer (NSCLC) [IMpower131] | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02308553 | Efficacy and Safety of Nintedanib Combined With Paclitaxel Chemotherapy for Patients With BRAF wt Metastatic Melanoma | cutaneous melanoma | Paclitaxel (NPC208553) | |
| NCT04572100 | Pilot Study of Chemotherapy for HPV-Associated Oropharyngeal Cancer | oropharynx squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00798252 | Ascending Multiple-Dose Study of Brivanib Alaninate in Combination With Chemotherapeutic Agents in Subjects With Advanced Cancers | cancer | Paclitaxel (NPC208553) | |
| NCT02012192 | A Study of MEK162 and Paclitaxel in Patients With Epithelial Ovarian, Fallopian Tube or Peritoneal Cancer | fallopian tube cancer | Paclitaxel (NPC208553) | |
| NCT00889382 | Paclitaxel-Loaded Polymeric Micelle and Carboplatin as First-Line Therapy in Treating Patients With Advanced Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00002772 | S9623, Combination Chemotherapy Plus Peripheral Stem Cell Transplantation in Treating Women With Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT03056833 | Ribociclib (Ribociclib (LEE-011)) With Platinum-based Chemotherapy in Recurrent Platinum Sensitive Ovarian Cancer | peritoneal neoplasm | Paclitaxel (NPC208553) | |
| NCT00618657 | Carboplatin+Nab-paclitaxel, Plus Trastuzumab (HER2+) or Bevacizumab (HER2-) in the Neoadjuvant Setting | breast cancer | Paclitaxel (NPC208553) | |
| NCT03169790 | QUILT-3.052: NANT Non-Hodgkin Lymphoma (NHL) Vaccine: Combination Immunotherapy in Subjects With Relapsed CD20-positive NHL | non-Hodgkins lymphoma | Paclitaxel (NPC208553) | |
| NCT05441254 | Camrelizumab Combined With Intraperitoneal Infusion of Nab-paclitaxel, Intravenous Chemotherapy and S-1 in the Treatment of Advanced Gastric Cancer With Peritoneal Metastasis:Single-arm, Prospective Clinical Study | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00064077 | Comparison of Four Combination Chemotherapy Regimens Using Cisplatin in Treating Patients With Stage IVB, Recurrent, or Persistent Cancer of the Cervix | cervical adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT04046016 | A Study Using Whole-body PET After Oral Microdose of 18F-labeled Liporaxel in Patients With Solid Tumor | neoplasm | Paclitaxel (NPC208553) | |
| NCT00616967 | Carboplatin and Nab-Paclitaxel With or Without Vorinostat in Treating Women With Newly Diagnosed Operable Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT02392507 | A Study of Nab-Paclitaxel and Carboplatin Plus Necitumumab (LY3012211) in Participants With Stage IV Squamous NSCLC | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00442260 | Doxorubicin HCL Liposome Injection (DOXIL) in Combination With Abraxane in Patients With Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00004126 | Paclitaxel and Oxaliplatin in Treating Patients With Recurrent or Advanced Non-small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00753038 | Phase 2 Study of REOLYSIN® in Combination With Paclitaxel and Carboplatin in Patients With Head and Neck Carcinoma | squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT03524898 | NAPAGE: NAb-PAclitaxel and GEmcitabine in Advanced Soft Tissue Sarcoma | soft tissue sarcoma | Paclitaxel (NPC208553) | |
| NCT04592861 | Carbon Ion RT for Locally Advanced Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT03490292 | Avelumab With Chemoradiation for Stage II/III Resectable Esophageal and Gastroesophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00003401 | Peripheral Stem Cell Transplantation Plus Combination Chemotherapy in Treating Patients With Multiple Myeloma | multiple myeloma | Paclitaxel (NPC208553) | |
| NCT03818282 | Trial Comparing Niraparib-bevacizumab-Dostarlimab and Niraparib-bevacizumab to Standard of Care in Recurrent Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00773695 | A Study of Bevacizumab (Avastin) in Combination With Neoadjuvant Treatment Regimens in Participants With Primary Human Epidermal Growth Factor Receptor 2 (HER2) Negative Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00404404 | ABI-007 and Bevacizumab in Treating Women With Recurrent or Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00951665 | A Study of Trastuzumab Emtansine, Paclitaxel, and Pertuzumab in Patients With HER2-Positive, Locally Advanced or Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00662597 | ASA404 or Placebo in Combination With Paclitaxel and Carboplatin as First-Line Treatment for Stage IIIb/IV Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT03008512 | A Phase II Study of Weekly Genexol-PM in Patients With Hepatocelluar Carcinoma After Failure of Sorafenib | hepatocellular carcinoma | Paclitaxel (NPC208553) | |
| NCT03314740 | Best Approach in Recurrent-Ovarian-Cancer-with Cediranib-Olaparib | ovarian neoplasm | Paclitaxel (NPC208553) | |
| NCT00136175 | Paclitaxel, Carboplatin and Gemcitabine in the Treatment of Patients With Advanced Transitional Cell Cancer of the Bladder | urinary bladder cancer | Paclitaxel (NPC208553) | |
| NCT03393884 | A Study of Apatinib Treatment in for Advanced Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT02711137 | Open-Label Safety and Tolerability Study of INCB057643 in Subjects With Advanced Malignancies | neoplasm | Paclitaxel (NPC208553) | |
| NCT00028860 | Combination Chemotherapy Following Surgery in Treating Patients With Urinary Tract Cancer | urinary bladder cancer | Paclitaxel (NPC208553) | |
| NCT03517176 | CEND-1 in Combination With Nabpaclitaxel and Gemcitabine in Metastatic Pancreatic Cancer | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00462397 | Paclitaxel and Carboplatin Followed by Cisplatin and Radiation Therapy in Treating Patients With Stage IB, Stage II, Stage III, or Stage IVA Cervical Cancer | cervical cancer | Paclitaxel (NPC208553) | |
| NCT04426955 | Study of Camrelizumab (SHR-1210) in Combination With Concurrent Chemoradiotherapy in Locally Advanced Esophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT02121990 | Metformin Hydrochloride, Carboplatin, and Paclitaxel in Treating Patients With Recurrent Ovarian, Fallopian Tube, or Primary Peritoneal Cancer | fallopian tube cancer | Paclitaxel (NPC208553) | |
| NCT00048893 | Vaccine and Chemotherapy for Previously Untreated Metastatic Breast Cancer | metastasis;breast neoplasm | Paclitaxel (NPC208553) | |
| NCT02617849 | Pembrolizumab With Carboplatin/Paclitaxel in Patients With Metastatic Melanoma | melanoma | Paclitaxel (NPC208553) | |
| NCT02125513 | Dose-Escalation Study of Intraperitoneal (IP) Cisplatin, IV/IP Paclitaxel, IV Bevacizumab, and Oral Olaparib for Newly Diagnosed Ovarian, Primary Peritoneal, and Fallopian Tube Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT03554044 | T-VEC With Chemotherapy or Endocrine Therapy in Treating Participants With HER2- Negative Breast Cancer | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT05427383 | KN026 in Combination With Chemotherapy in the Second Line Treatment of HER-2 Positive Advanced or Metastatic Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT04460066 | A Study of Anti-PD-L1 Antibody in Neoadjuvant Chemotherapy of Esophageal Squamous Cell Carcinoma. | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00731861 | Phase I, Pharmacokinetic, Pharmacodynamic Trial of PTK787 and Paclitaxel in Combination for Advanced Solid Tumors | metastasis;carcinoma | Paclitaxel (NPC208553) | |
| NCT01402271 | Study of AMG 386 in Combination With Paclitaxel and Carboplatin in Subjects With Ovarian Cancer | fallopian tube cancer | Paclitaxel (NPC208553) | |
| NCT02221999 | Weekly Paclitaxel and Cisplatin to Treat Hormone Receptor Positive and Triple Negative Breast Cancer Patients | inflammatory breast carcinoma | Paclitaxel (NPC208553) | |
| NCT00626561 | Bevacizumab and Paclitaxel for Neuroendocrine Tumors of the Cervix | cervical cancer | Paclitaxel (NPC208553) | |
| NCT03189446 | Vaginal Cuff Brachytherapy Followed by Chemotherapy in Endometrial Cancer | endometrial cancer | Paclitaxel (NPC208553) | |
| NCT02117024 | A Phase 2 Study of Viagenpumatucel-L (HS-110) in Patients With Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01351350 | Dose Escalation Study of MLN0128 in Combination With Paclitaxel, With/Without Trastuzumab, in Subjects With Advanced Solid Malignancies | cancer | Paclitaxel (NPC208553) | |
| NCT00003092 | Paclitaxel in Treating Older Patients With Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT03309150 | ATRi Transition Rollover Study | neoplasm | Paclitaxel (NPC208553) | |
| NCT01179269 | Pazopanib and Paclitaxel for Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02250326 | Safety and Efficacy Study of Nab®-Paclitaxel With CC-486 or Nab®-Paclitaxel With Durvalumab, and Nab®-Paclitaxel Monotherapy as Second/Third-line Treatment for Advanced Non-small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00816634 | Efficacy Comparison Study of Combination Regimens to Treat Advanced Esophageal Squamous Cell Carcinoma | squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00003812 | Chemotherapy Plus Radiation Therapy in Treating Patients With Limited-Stage Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT03057600 | Study of CB-839 in Combination w/ Paclitaxel in Participants of African Ancestry and Non-African Ancestry With Advanced Triple Negative Breast Cancer (TNBC) | triple-negative breast cancer | Paclitaxel (NPC208553) | |
| NCT00003539 | Paclitaxel Plus Monoclonal Antibody Therapy in Treating Women With Recurrent or Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00524303 | EndoTAG-1 in Triple Receptor Negative Breast Cancer Patients | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT00032032 | Combination Chemotherapy Plus Radiation Therapy in Treating Patients With Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00151060 | Estramustine, Etoposide and Paclitaxel Treatment for Hormonally Responsive Adenocarcinoma of the Prostate | prostate cancer | Paclitaxel (NPC208553) | |
| NCT00281658 | ErbB2 Over-expressing Metastatic Breast Cancer Study Using Paclitaxel, Trastuzumab, and Lapatinib | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT00955305 | Paclitaxel, Carboplatin, and Bevacizumab With or Without Cixutumumab in Treating Patients With Stage IV or Recurrent Non-small Cell Lung Cancer | large cell lung carcinoma;lung adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00110695 | Therapy With Abraxane and 5-Fluorouracil, Epirubicin, Cyclophosphamide (FEC) for Patients With Breast Cancer | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT00289263 | Maintenance Chemotherapy in Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT04158635 | Gemcitabine, Nab-Paclitaxel, and Bosentan for the Treatment of Unresectable Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00129727 | Carboplatin Taxol Avastin in Ovarian Cancer (OVCA) | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT02103062 | Phase 2 Study With Abraxane (Nab®Paclitaxel) in Metastatic Colorectal Cancer | colorectal neoplasm | Paclitaxel (NPC208553) | |
| NCT03830606 | The Efficacy and Safety of Nab-paclitaxel Plus S-1 in First-line Treatment of Advanced Biliary Tract Adenocarcinoma | biliary tract cancer | Paclitaxel (NPC208553) | |
| NCT00547443 | Sorafenib and High-Dose Carboplatin, Paclitaxel, and External-Beam Radiation Therapy in Treating Patients With Stage III Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00112489 | Paclitaxel and Carboplatin in Treating Patients With Persistent or Recurrent Stage III or Stage IV Uterine Cancer | sarcoma | Paclitaxel (NPC208553) | |
| NCT00915603 | Trial of Paclitaxel/Bevacizumab +/- Everolimus for Patients With HER2-Negative Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT03464734 | Pembrolizumab and Nab Paclitaxel in Patients With Metastatic Urothelial Carcinoma | urothelial carcinoma | Paclitaxel (NPC208553) | |
| NCT02682693 | Denosumab as an add-on Neoadjuvant Treatment (GeparX) | inflammatory breast carcinoma | Paclitaxel (NPC208553) | |
| NCT00558636 | A Trial Comparing Safety and Efficacy of Carboplatin and Paclitaxel Plus or Minus Sorafenib (BAY43-9006) in Chemonaive Patients With Stage IIIB-IV Non-Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00025298 | Chemotherapy Plus Radiation Therapy With or Without Amifostine in Treating Patients With Locally Advanced Cancer of the Nasopharynx | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT02051751 | Paclitaxel and BKM120 Before Surgery in Treating Patients With Stage II or III Estrogen Receptor-Positive and HER2-Negative Breast Cancer | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT04213937 | Nab-paclitaxel Versus Topotecan As Second-Line Treatment for Patients With Extensive Stage Small Cell Lung Cancer | small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT03815461 | Nab-paclitaxel Combined With S-1 as Induction Therapy for Locally Advanced Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT02597036 | A Study of LY3127804 With Ramucirumab in Participants With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT02107937 | Phase II Study DCVAC/OvCa Added to First Line Carboplatin and Paclitaxel Newly Diagnosed Epithelial Ovarian Carcinoma | malignant epithelial tumor of ovary | Paclitaxel (NPC208553) | |
| NCT01968915 | Safety and Tolerability of Oral LCL161 in Japanese Adult Patients With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT00039546 | Combination Chemotherapy in Treating Women With Breast Cancer Who Have Undergone Surgery | breast cancer | Paclitaxel (NPC208553) | |
| NCT00561119 | Maintenance Versus Observation After 6 Cycles of Gemcitabine Plus Paclitaxel in Pts With Advanced Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT01288261 | Paclitaxel and Bavituximab in Treating Patients With HER2-Negative Metastatic Breast Cancer | male breast carcinoma | Paclitaxel (NPC208553) | |
| NCT05257993 | Study to Assess the Safety, Tolerability of JPI-547 in Combination With Modified FOLFIRINOX or Gemcitabine-nab-paclitaxel in Patients With Locally Advanced and Metastatic Pancreatic Cancer | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT02735239 | Study of Anti-PD-L1 in Combination With Chemo(Radio)Therapy for Oesophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT02379416 | Combination Nilotinib and Paclitaxel in Adults With Relapsed Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT02426281 | Nab-pacliatxel Plus Gemcitabine in Korean Patients With Metastatic Pancreatic Ductal Adenocarcinoma | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT02422563 | NeoAdjuvant Chemotherapy Followed by Radical Hysterectomy (OP) Versus Primary Chemo-RADiation in Cervical Cancer FIGO Stage IB2 and IIB | cervical cancer | Paclitaxel (NPC208553) | |
| NCT00070278 | Neoadjuvant Epirubicin, Cyclophosphamide, and Paclitaxel With or Without Gemcitabine in Treating Women Who Are Undergoing Surgery for Early Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT03537690 | FID-007 in Treating Participants With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT02684227 | Enzalutamide, Carboplatin, and Paclitaxel in Treating Patients With Stage III-IV or Recurrent Endometrioid Endometrial Cancer | uterine neoplasm | Paclitaxel (NPC208553) | |
| NCT01297452 | BKM120 + Carboplatin + Paclitaxel for Patients With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT03415802 | Efficacy and Safety of Nab-Paclitaxel Plus S-1 in the First-line Treatment of Advanced Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00004055 | Topotecan, Paclitaxel, and Filgrastim in Treating Patients With Previously Untreated Extensive-Stage Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT03036488 | Safety and Efficacy Study of Pembrolizumab (MK-3475) in Combination With Chemotherapy as Neoadjuvant Treatment for Participants With Triple Negative Breast Cancer (TNBC) (MK-3475-173/KEYNOTE-173) | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT02059967 | Phase I IGART Study Using Active Breathing Control and Simultaneous Boost for Patients With NSCLC | lung adenocarcinoma;squamous cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02106884 | Quality of Life in Locally Advanced or Metastatic Pancreatic Cancer Treated With Gemcitabine and Nab-paclitaxel | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT01763671 | Paclitaxel-bevacizumab in Advanced Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT05262335 | Anlotinib Plus Chemotherapy as First-line Therapy for Gastrointestinal Tumor Patients With Unresectable Liver Metastasis (ALTER-G-001) | neoplasm | Paclitaxel (NPC208553) | |
| NCT01204749 | Saracatinib and Paclitaxel in Platinum-resistant Ovarian Cancer | fallopian tube cancer;ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00848718 | A Phase I Study of MK-2206 in Combination With Standard Chemotherapy in Participants With Locally Advanced or Metastatic Solid Tumors (MK-2206-003) | neoplasm | Paclitaxel (NPC208553) | |
| NCT00912639 | A Clinical Trial of Paclitaxel Loaded Polymeric Micelle in Patients With Taxane-Pretreated Recurrent Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT02639650 | Study of Paclitaxel Plus Cisplatin as the First-line Chemotherapy in High Risk Gestational Trophoblastic Tumor | gestational trophoblastic neoplasm | Paclitaxel (NPC208553) | |
| NCT00287989 | Erlotinib, Paclitaxel, and Carboplatin in Treating Patients With Stage III, Stage IV, or Recurrent Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00016406 | S0012 Doxorubicin, Cyclophosphamide, and Paclitaxel With or Without Filgrastim in Treating Women With Inflammatory or Locally Advanced Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00084214 | STA-4783/Paclitaxel or Paclitaxel Alone in Melanoma | melanoma | Paclitaxel (NPC208553) | |
| NCT02270814 | Cisplatin, Nab-Paclitaxel, and Cetuximab (CACTUX) in Patients With Incurable Head and Neck Squamous Cell Carcinoma | head and neck squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT02412371 | A Study Evaluating the Efficacy and Tolerability of Veliparib in Combination With Paclitaxel/Carboplatin-Based Chemoradiotherapy Followed by Veliparib and Paclitaxel/Carboplatin Consolidation in Adults With Stage III Non-Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01704690 | Combination Treatment of S-1 With Paclitaxel in Advanced Esophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00970580 | A Study of BIIB022 in Combination With Paclitaxel and Carboplatin in Subjects With Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00084448 | Paclitaxel and Celecoxib in Treating Patients With Recurrent or Persistent Platinum-Resistant Ovarian Epithelial or Primary Peritoneal Cancer | ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT00002819 | Chemotherapy With or Without Peripheral Stem Cell Transplantation in Treating Patients With Persistent Ovarian Epithelial Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00320541 | A Trial of Paclitaxel and Bevacizumab vs. Gemcitabine, Paclitaxel, and Bevacizumab in Advanced Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00467012 | Phase Ⅱ Clinical Study of R435 (Bevacizumab) in Combination With Paclitaxel in Patients With Inoperable Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00818090 | Paclitaxel and Cisplatin for Thymic Neoplasm | Thymic Carcinoma;Thymoma | Paclitaxel (NPC208553) | |
| NCT00470301 | Tipifarnib and Combination Chemotherapy in Treating Patients With Stage II or Stage III Breast Cancer | male breast carcinoma | Paclitaxel (NPC208553) | |
| NCT00448305 | Neoadjuvant Study With Chemotherapy, Lapatinib And Trastuzumab In Breast Cancer | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT03853707 | Ipatasertib in Combination With Carboplatin, Carboplatin/Paclitaxel, or Capecitabine/Atezolizumab in Treating Patients With Metastatic Triple Negative Breast Cancer | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT00006377 | Doxorubicin, Paclitaxel, and Carboplatin in Treating Patients With Primary Stage III, Stage IV, or Recurrent Endometrial Cancer | endometrial cancer | Paclitaxel (NPC208553) | |
| NCT04592211 | Pembrolizumab, Olaparib, Recurrent/Advanced Gastric and Gastro-esophageal Junction(GEJ) Cancer With HRR Mutation and MSS | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00772798 | Paclitaxel and Carboplatin Combination as 1st Line Treatment in Ovarian Carcinomas | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT05344742 | A Study of Mitoxantrone Hydrochloride Liposome Injection Combination Therapy in Chinese Patients With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT00476827 | A Phase II Safety and Tolerability Study of Bevacizumab When Added to Single-agent Chemotherapy to Treat Patient With Breast Cancer Metastatic to Brain | breast cancer | Paclitaxel (NPC208553) | |
| NCT04581265 | Nab-PTX, Ifosfamide and Cisplatin in the Treatment of Pediatric Extracranial Germ Cell Tumor. | germ cell tumor | Paclitaxel (NPC208553) | |
| NCT00881166 | Safety of MP470 in Combination With Standard-of-Care Chemotherapy Regimens to Treat Solid Tumors | cancer | Paclitaxel (NPC208553) | |
| NCT00879359 | Trial of Vascular Endothelial Growth Factor (VEGF), Bevacizumab, in Combination With Cytotoxic Chemotherapy for Endometrial Cancer | endometrial carcinoma | Paclitaxel (NPC208553) | |
| NCT02467907 | Safety and Efficacy of Bevacizumab in Combination With Carboplatin and Paclitaxel for Metastatic, Recurrent or Persistent Cervical Cancer | cervical cancer | Paclitaxel (NPC208553) | |
| NCT02133612 | Adjuvant Chemotherapy With Paclitaxel and Cisplatin in Lymph Node-Positive Thoracic Esophageal Squamous Cell Carcinoma | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT03197584 | Neo-adjuvant Pembrolizumab in Primary Stage IV Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00800202 | A Study of Avastin (Bevacizumab) in Patients With Non-Squamous Non-Small Cell Lung Cancer With Asymptomatic Untreated Brain Metastasis | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00016315 | Gemcitabine, Carboplatin or Paclitaxel Plus Radiation Therapy in Treating Patients With Stage IIIA or IIIB Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT01463072 | Nab-Paclitaxel in Treating Older Patients With Locally Advanced or Metastatic Breast Cancer | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT01204996 | A Study of the Safety and Efficacy of CNTO 888 in Combination With SoC (Standard of Care) Chemotherapy in Patients With Solid Tumors | cancer | Paclitaxel (NPC208553) | |
| NCT04876313 | An Open Label, Single-arm, Phase 2 Study of Neoadjuvant Nivolumab and Nab-paclitaxel Before Radical Cystectomy for Patients With Muscle-invasive Bladder Cancer (NURE-Combo) | urinary bladder carcinoma | Paclitaxel (NPC208553) | |
| NCT00002814 | Combination Chemotherapy for Patients With Brain Cancer | Central Nervous System Neoplasm | Paclitaxel (NPC208553) | |
| NCT02858206 | Simultaneous Integrated Boost Radiotherapy and Concurrent Nimotuzumab or Chemotherapy for Esophageal Carcinoma | esophageal carcinoma | Paclitaxel (NPC208553) | |
| NCT01497470 | A Clinical Study in Cancer Patients to Investigate the Potential Impact of Custirsen, on the Blood Levels of the Chemotherapeutic Drug, Paclitaxel, When Given Together as Part of a Treatment Regimen | cancer | Paclitaxel (NPC208553) | |
| NCT01069328 | Dose Escalating Study With BAY43-9006 With Carboplatin, Paclitaxel and Bevacizumab in Untreated Stage IIIb Non-small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT04723030 | The Transformation of Locally Advanced Pancreatic Cancer. | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00736944 | Trial of 2 Cycles of Induction Chemo With Abraxane, Cetuximab, Cisplatin, & 5-FU for Advanced Head and Neck Cancer | head and neck squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00513695 | Sunitinib Malate, Paclitaxel, Doxorubicin Hydrochloride, and Cyclophosphamide Before Surgery in Treating Patients With Stage IIB-IIIC Breast Cancer | inflammatory breast carcinoma;male breast carcinoma | Paclitaxel (NPC208553) | |
| NCT01240655 | A Study of LCL161 in Combination With Weekly Paclitaxel in Adult Patients With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT00003399 | Peripheral Stem Cell Transplantation Plus Combination Chemotherapy in Treating Patients With Multiple Myeloma | multiple myeloma | Paclitaxel (NPC208553) | |
| NCT02779855 | Talimogene Laherparepvec in Combination With Neoadjuvant Chemotherapy in Triple Negative Breast Cancer | breast ductal carcinoma in situ | Paclitaxel (NPC208553) | |
| NCT01258192 | Neoadjuvant Chemotherapy With Nab-paclitaxel Plus Cisplatin for Stage Ⅱ-Ⅲ Esophageal Cancer Patients | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00002939 | Irinotecan and Paclitaxel in Treating Patients With Metastatic or Recurrent Cancer | neoplasm | Paclitaxel (NPC208553) | |
| NCT00576225 | CT-2103/Carboplatin vs Paclitaxel/Carboplatin for NSCLC in Women With Estradiol > 25 pg/mL | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01326767 | Central European Society for Anticancer Research (CESAR) Study of Paclitaxel Therapeutic Drug Monitoring | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01712919 | Chemoradiation Plus Weekly c225 Against Locoregionally Advanced Nasopharyngeal Carcinoma (NPC) | nasopharyngeal neoplasm | Paclitaxel (NPC208553) | |
| NCT05371301 | Effect of Albumin-bound Paclitaxel Plus Carboplatin Compared With Paclitaxel Plus Carboplatin in Patients Receiving Neoadjuvant Chemotherapy for Advanced Ovarian, Fallopian Tube or Peritoneal Cancer: A Phase 2 Single Center Clinical Trial | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT03087864 | PDL-1 Targeting in Resectable Oesophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00002662 | Paclitaxel or Docetaxel in Treating Women With Advanced Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00849472 | Phase II Lapatinib Plus Nab-Paclitaxel As First And Second Line Therapy In her2+ MBC | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT04568200 | Study of Anti-PD-L1 in Combination With Chemo(Radio)Therapy for Resectable Esophageal Squamous Cell Carcinoma | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT04221828 | Trial of NanoPac Focal Therapy for Prostate Cancer | prostate adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT01653912 | Phase Ib Study of Olaparib Plus Weekly Carboplatin and Paclitaxel in Relapsed Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT01154920 | Paclitaxel, Carboplatin and Cetuximab (PCC) With Cetuximab, Docetaxel, Cisplatin and Fluorouracil (C-TPF) in Previously Untreated Patients With Locally Advanced Head and Neck Squamous Cell Carcinoma | head and neck squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT04138719 | Nab-paclitaxel Plus Carboplatin Versus Nab-paclitaxel Plus Epirubicin in the Neoadjuvant Therapy for Breast Cancer | triple-negative breast cancer | Paclitaxel (NPC208553) | |
| NCT01034189 | Cetuximab/Paclitaxel/Cisplatin Concurrent Chemoradiotherapy Followed by Esophagectomy for Loco-regional Esophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT05497778 | A Phase 1b Study of Gemcitabine and Nab-paclitaxel in Combination With IM156 in Patients With Advanced Pancreatic Cancer. | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT04063683 | Paclitaxel and DDP Combined With Anlotinib in the First-line Treatment for Patients With Advanced Esophageal Squamous Cell Carcinoma(ESCC). | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT05446870 | Pembrolizumab With Chemotherapy and MK-4830 for Treating Participants With Ovarian Cancer (MK-4830-002) | ovarian carcinoma | Paclitaxel (NPC208553) | |
| NCT02204345 | A Study Evaluating RO5479599 in Combination With Carboplatin and Paclitaxel in Participants With Advanced or Metastatic Non-Small-Cell Lung Cancer (NSCLC) of Squamous Histology | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02689427 | Enzalutamide and Paclitaxel Before Surgery in Treating Patients With Stage I-III Androgen Receptor-Positive Triple-Negative Breast Cancer | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT05218889 | Surufatinib Plus Camrelizumab and AS in First Line Treatment of Advanced Metastatic Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00021060 | Combination Chemotherapy With or Without Bevacizumab in Treating Patients With Advanced, Metastatic, or Recurrent Non-Small Cell Lung Cancer | lung adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT04884906 | Camrelizumab Combined With Radiotherapy and Chemotherapy for the Treatment of Recurrent or Metastatic Cervical Cancer | cervical cancer | Paclitaxel (NPC208553) | |
| NCT00936702 | Carboplatin, Paclitaxel, and Everolimus in Treating Patients With Previously Untreated Cancer of Unknown Primary | carcinoma | Paclitaxel (NPC208553) | |
| NCT02092363 | GANNET53: Ganetespib in Metastatic, p53-mutant, Platinum-resistant Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00246571 | Weekly Taxol Plus Xeloda® vs Taxotere q3wk Plus Xeloda® in the Treatment of Metastatic BC | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT00003701 | Combination Chemotherapy in Treating Patients With Bladder Cancer | urinary bladder cancer | Paclitaxel (NPC208553) | |
| NCT00823186 | Dose-finding Study of Weekly Paclitaxel and Cisplatin in FIGO IB2 and Bulky IIA Cervical Cancer | cervical cancer | Paclitaxel (NPC208553) | |
| NCT00138151 | Isotretinoin, Interferon Alpha-2b, and Paclitaxel in Stage IV, Recurrent, or Persistent Cervical Cancer | cervical cancer | Paclitaxel (NPC208553) | |
| NCT03941093 | Evaluation of Efficacy and Safety of Neoadjuvant Treatment With Pamrevlumab in Combination With Chemotherapy (Either Gemcitabine Plus Nab-paclitaxel or FOLFIRINOX) in Participants With Locally Advanced Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00038168 | Intravenous Estramustine With Taxol in Hormone Refractory Prostate Adenocarcinoma | prostate cancer | Paclitaxel (NPC208553) | |
| NCT01653470 | Study to Evaluate Safety & Tolerability of BMS-906024 in Combination With Chemotherapy & to Define DLTs & MTD of BMS-906024 in Combination With One of the Following Chemotherapy Regimens | cancer | Paclitaxel (NPC208553) | |
| NCT02240212 | A Phase I Trial of the Combination of AZD2014 and Weekly Paclitaxel. | cancer | Paclitaxel (NPC208553) | |
| NCT00919880 | Comparison of Neo-adjuvant Weekly Paclitaxel With or Without Carboplatin in Early Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00788931 | A Trial l of Panobinostat Given in Combination With Trastuzumab and Paclitaxel in Adult Female Patients With HER2 Positive Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT03595059 | A Study With ABBV-155 Alone and in Combination With Taxane Therapy in Adults With Relapsed and/or Refractory Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT05493995 | A Study of Penpulimab and Anlotinib Plus Nab-paclitaxel and Gemcitabine for Metastatic Pancreatic Cancer. | pancreatic neoplasm | Paclitaxel (NPC208553) | |
| NCT04258644 | Camrelizumab in Combination With Apatinib Mesylate, Paclitaxel-albumin and S-1 for Translational Treatment of Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT03944304 | Paclitaxel in Patients With GIST With Low P-glycoprotein Expression After Failure of at Least Imatinib and Sunitinib, and Regorafenib. | gastrointestinal stromal tumor | Paclitaxel (NPC208553) | |
| NCT05013697 | TQB2450 Solution for Injection (TQB2450)+Paclitaxel+Cisplatin ± Anlotinib in the Treatment of Esophageal Cancer. | esophageal carcinoma | Paclitaxel (NPC208553) | |
| NCT05201547 | Endometrial Cancer Patientes MMR Deficient Comparing Chimiotherapy vs Dostarlimab in First Line | endometrial cancer | Paclitaxel (NPC208553) | |
| NCT01441349 | Irinotecan/Cisplatin With or Without Simvastatin in Chemo-naive Patients With Extensive Disease-small Cell Lung Cancer | small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00022633 | S0028, Gemcitabine and Paclitaxel in Treating Patients With Advanced or Recurrent Cancer of the Urinary Tract | urethra cancer;urinary bladder cancer | Paclitaxel (NPC208553) | |
| NCT03308552 | Radiotherapy With or Without Concurrent Chemotherapy for Limited Lymphatic Metastasis of Esophageal Cancer - 3JECROG P-02 | esophageal carcinoma | Paclitaxel (NPC208553) | |
| NCT01770171 | Carboplatin-Paclitaxel ± Bevacizumab in Advanced (Stage III-IV) or Recurrent Endometrial Cancer | endometrial cancer | Paclitaxel (NPC208553) | |
| NCT00431080 | Randomized Phase III Trial Comparing Sequential Administration of FE75C Followed by Docetaxel Versus Paclitaxel as Adjuvant Chemotherapy in Axillary Lymph Node (+) Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00006108 | Capecitabine, Paclitaxel, and Trastuzumab in Treating Patients With Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT05381909 | Study Investigating BGB-24714 as Monotherapy and in Combination With Chemotherapy in Participants With Advanced or Metastatic Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT03908333 | High Dose Ascorbic Acid and Nanoparticle Paclitaxel Protein Bound and Cisplatin and Gemcitabine (AA NABPLAGEM) in Patients Who Have Metastatic Pancreatic Cancer | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT02476955 | Veliparib With Carboplatin and Paclitaxel and as Continuation Maintenance Therapy in Adults With Newly Diagnosed Stage III or IV, High-grade Serous, Epithelial Ovarian, Fallopian Tube, or Primary Peritoneal Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00083057 | Gefitinib, Paclitaxel, and Radiation Therapy in Treating Patients With Advanced or Recurrent Squamous Cell Carcinoma of the Head and Neck | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT02138383 | Enzalutamide in Combination With Gemcitabine and Nab-Paclitaxel for the Treatment of Advanced Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT02027428 | Safety and Efficacy Study of Abraxane as Maintenance Treatment After Abraxane Plus Carboplatin in 1st Line Stage IIIB / IV Squamous Cell Non-small Cell Lung Cancer | squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00017407 | Carboplatin and Paclitaxel With or Without ISIS 3521 in Treating Patients With Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00903942 | Abraxane and RT for Non-Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02312804 | Ph Ib/BGJ398/Cervix and Other Solid Tumors | cervical carcinoma | Paclitaxel (NPC208553) | |
| NCT00025480 | Tipifarnib Plus Radiation Therapy After Combination Chemotherapy in Treating Patients With Stage III Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT01015222 | Dasatinib, Bevacizumab, Paclitaxel in Patients With Advanced Malignancies | cancer | Paclitaxel (NPC208553) | |
| NCT00329641 | Sorafenib, Carboplatin, and Paclitaxel in Treating Patients With Stage IV Melanoma of the Eye | melanoma | Paclitaxel (NPC208553) | |
| NCT03635567 | Efficacy and Safety Study of First-line Treatment With Pembrolizumab (MK-3475) Plus Chemotherapy Versus Placebo Plus Chemotherapy in Women With Persistent, Recurrent, or Metastatic Cervical Cancer (MK-3475-826/KEYNOTE-826) | cervical cancer | Paclitaxel (NPC208553) | |
| NCT00001387 | Phase I and Pharmacokinetic Trial of Paclitaxel (Taxol) Given as a 3-Hour Infusion in Pediatric Patients With Refractory Malignancy | neoplasm | Paclitaxel (NPC208553) | |
| NCT02389751 | Ganetespib in Combination With Paclitaxel, Carboplatin, and Radiation Therapy in Treating Patients With Stage II-III Esophageal Cancer | esophageal carcinoma | Paclitaxel (NPC208553) | |
| NCT00661193 | S0709: Erlotinib With or Without Carboplatin and Paclitaxel in Stage IIIB or Stage IV Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT01083537 | Intraperitoneal vs Intravenous Chemotherapy Following Neoadjuvant Chemotherapy in Ovarian Cancer | fallopian tube cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT04806945 | A Phase III Study to Evaluate Efficacy and Safety of First-Line Treatment With HLX10 + Chemotherapy in Patients With Advanced Cervical Cancer | cervical cancer | Paclitaxel (NPC208553) | |
| NCT00662129 | Paclitaxel Albumin-Stabilized Nanoparticle Formulation, Gemcitabine, and Bevacizumab in Treating Patients With Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT04908787 | PD-1 Antibody Combined Neoadjuvant Chemotherapy for Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT03322969 | Receiving Modified Chemotherapy Followed With Radical Resection After Neoadjuvant Chemotherapy | gastric cancer | Paclitaxel (NPC208553) | |
| NCT05214976 | A Trial of Camrelizumab for Injection in Combination With Famitinib Malate Capsule and Paclitaxel For Injection(Albumin Bound) in Advanced Solid Tumors of Patients | neoplasm | Paclitaxel (NPC208553) | |
| NCT02442362 | TOF Versus SOX in Metastatic Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT01702714 | A Study of RO5083945 in Combination With Cisplatin and Gemcitabine or Carboplatin and Paclitaxel in Patients With Advanced or Recurrent Non-Small Cell Lung Cancer of Squamous Histology Who Have Not Received Prior Chemotherapy for The Metastatic Disease | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00111904 | Paclitaxel in Treating Patients With Unresectable Locally Advanced or Metastatic Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT02761694 | Absorption, Metabolism, Excretion and Pharmacokinetics of a Single Dose [14C]AZD2014 Followed by a Multiple Dose Phase | cancer | Paclitaxel (NPC208553) | |
| NCT00005078 | Tirapazamine, Carboplatin, and Paclitaxel in Treating Patients With Advanced Malignant Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT04634877 | Study of Pembrolizumab (MK-3475) in Combination With Adjuvant Chemotherapy With or Without Radiotherapy in Participants With Newly Diagnosed Endometrial Cancer After Surgery With Curative Intent (MK-3475-B21 / KEYNOTE-B21 / ENGOT-en11 / GOG-3053) | endometrial neoplasm | Paclitaxel (NPC208553) | |
| NCT02506842 | Second-Line Adjuvant Therapy With Nab-Paclitaxel Plus Gemcitabine Versus Oxaliplatin Plus Folinic Acid and Fluorouracil for Gemcitabine-Refractory Pancreatic Cancer After Curative Resection | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT01340131 | CKD-828 (40/5mg) Pharmacokinetic Study | hypertension | Paclitaxel (NPC208553) | |
| NCT03412799 | Study of SBP-101 Combined With Nab-Paclitaxel and Gemcitabine in Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT03737643 | An uMbrella Study of BIomarker-driven Targeted Therapy In Patients With Platinum-resistant Recurrent OvariaN Cancer(AMBITION) | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT02038621 | The Maintenance Therapy of Capecitabine of Advanced Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00006374 | Combination Chemotherapy in Treating Patients With Extensive-Stage Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT04584112 | A Study of the Safety, Efficacy, and Pharmacokinetics of Tiragolumab in Combination With Atezolizumab and Chemotherapy in Participants With Triple-Negative Breast Cancer | triple-negative breast cancer | Paclitaxel (NPC208553) | |
| NCT00003732 | Combination Chemotherapy in Treating Patients With Stage II, Stage III, or Stage IV Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00003589 | Combination Chemotherapy in Treating Patients With Advanced Non-small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00513292 | Combination Chemotherapy and Paclitaxel Plus Trastuzumab in Treating Women With Palpable Breast Cancer That Can Be Removed by Surgery | breast cancer | Paclitaxel (NPC208553) | |
| NCT01604863 | A Study of HGS1036 in Combination With Chemotherapy in Subjects With Advanced Solid Malignancies | cancer | Paclitaxel (NPC208553) | |
| NCT04129996 | A Trial of Camrelizumab in Combination With Nab-paclitaxel and Famitinib as a First Line Treatment in Patients With Unresectable Locally Advanced or Metastatic Immunomodulatory Triple Negative Breast Cancer(FUTURE-C-PLUS) | triple-negative breast cancer | Paclitaxel (NPC208553) | |
| NCT04481009 | A Study to Evaluate YH003 in Combination With Toripalimab (Anti-PD-1 mAb) in Subjects With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT02903004 | Trial on Trabectedin (ET-743) vs Clinician's Choice Chemotherapy in Recurrent Ovarian, Primary Peritoneal or Fallopian Tube Cancers of BRCA Mutated or BRCAness Phenotype Patients | ovarian neoplasm | Paclitaxel (NPC208553) | |
| NCT04742036 | Capivasertib China PK Study | neoplasm | Paclitaxel (NPC208553) | |
| NCT04527900 | The UPPROACH (Upfront Intensity Modulated Proton Beam Therapy) Approach | endometrial cancer | Paclitaxel (NPC208553) | |
| NCT04868708 | A Study of AK104( an Anti-PD-1 and Anti-CTLA-4 Bispecific Antibody) in Recurrent or Metastatic Cervical Cancer | cervical cancer | Paclitaxel (NPC208553) | |
| NCT00198354 | Stage I/II NSCLC Perioperative Chemotherapy | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01026116 | Treatment With Pazopanib for Neoadjuvant Breast Cancer | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT00398086 | Gemcitabine Plus Albumin-bound Paclitaxel In Patients With Advanced Metastatic Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT01110603 | A Study of MK-4827 in Combination With Standard Chemotherapy in Participants With Advanced Solid Tumors (MK-4827-008 AM1) | cancer | Paclitaxel (NPC208553) | |
| NCT04164238 | Neoadjuvant Anti-PD-1 Antibody (Toripalimab) or Combined With Chemotherapy in HNSCC Patients | head and neck squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT03748134 | Sintilimab or Placebo With Chemotherapy in Esophageal Squamous Cell Carcinoma | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT02810418 | Mesothelin-Targeted Immunotoxin LMB-100 Alone or in Combination With Nab-Paclitaxel in People With Previously Treated Metastatic and/or Locally Advanced Pancreatic Ductal Adenocarcinoma and Mesothelin Expressing Solid Tumors | pancreatic neoplasm | Paclitaxel (NPC208553) | |
| NCT01752205 | Paclitaxel Plus Radiation With Erlotinib to Treat Esophageal Squamous Carcinoma | squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT05290935 | Immunotherapy for Recurrent Cervical Cancer Refractory to Platinum-based Chemotherapy: Multi-Center Trial | cervical carcinoma | Paclitaxel (NPC208553) | |
| NCT03763123 | Study of Chemotherapy With Pembrolizumab (MK-3475) Followed by Maintenance With Olaparib (MK-7339) for the First-Line Treatment of Women With BRCA Non-mutated Advanced Epithelial Ovarian Cancer (EOC) (MK-7339-001/KEYLYNK-001/ENGOT-ov43/GOG-3036) | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT02138812 | A Phase 1, Dose Escalation Study to Assess the Safety and Tolerability of ASP9853 With Either Docetaxel or Paclitaxel in Patients With Advanced Non-hematologic Malignancies | cancer | Paclitaxel (NPC208553) | |
| NCT00779714 | Comparative Study of Individualized Sensitivity-Directed Chemotherapy Versus DTIC | melanoma | Paclitaxel (NPC208553) | |
| NCT00045630 | S0219, Combination Chemotherapy Followed By Observation or Surgery in Patients With Stage II or Stage III Cancer of the Urothelium | urethra cancer;urinary bladder cancer | Paclitaxel (NPC208553) | |
| NCT00006049 | ZD 1839 Plus Chemotherapy in Treating Patients With Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT01183559 | A Trial of ZD6474, Paclitaxel, Carboplatin, 5-Fluorouracil, and Radiation Therapy Followed by Surgery | esophageal carcinoma;stomach neoplasm | Paclitaxel (NPC208553) | |
| NCT00828841 | Phase 2b Study of Cetuximab With Platinum-Based Chemo as First Line Treatment of Recurrent or Advanced NSCLC | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00959387 | Induction Chemotherapy for Advanced Head and Neck Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT00517621 | Use of FACT-GOG/NTX Questionnaire in Peripheral Neurotoxicity & Validation of a French Version of This Questionnaire | fallopian tube cancer;ovarian cancer;peritoneal neoplasm | Paclitaxel (NPC208553) | |
| NCT00002519 | Paclitaxel Plus Radiation Therapy in Treating Patients With Untreated Stage III Non-small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00003105 | Combination Chemotherapy in Treating Patients With Metastatic or Locally Advanced Bladder Cancer | urinary bladder cancer | Paclitaxel (NPC208553) | |
| NCT00319839 | Study of Albumin Bound-Paclitaxel for Treatment of Recurrent or Metastatic Head and Neck Cancer With Cetuximab | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT04249167 | Cryoablation, Atezolizumab/Nab-paclitaxel for Locally Advanced or Metastatic Triple Negative Breast Cancer | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT00502203 | Paclitaxel and Carboplatin in Women With Malignant Mixed Mullerian Tumors (MMMT) of the Uterus | mixed neoplasm | Paclitaxel (NPC208553) | |
| NCT02364999 | A Comparative Study Of PF-06439535 Plus Paclitaxel-Carboplatin And Bevacizumab Plus Paclitaxel-Carboplatin Patients With Advanced Non-Squamous NSCLC | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT03181100 | Atezolizumab With Chemotherapy in Treating Patients With Anaplastic or Poorly Differentiated Thyroid Cancer | thyroid carcinoma | Paclitaxel (NPC208553) | |
| NCT03797443 | High Dose Ascorbic Acid (AA) + Nanoparticle Paclitaxel Protein Bound + Cisplatin + Gemcitabine (AA NABPLAGEM) in Patients Who Have Metastatic Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00753909 | Paclitaxel, Carboplatin Plus Bevacizumab in Pretreated, Advanced or Metastatic Non Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02911350 | Safety Study of Combination of Hormone Therapy, Paclitaxel and Radiation Therapy to Treat Prostate Cancer | prostate cancer | Paclitaxel (NPC208553) | |
| NCT00272987 | Comparison of Safety and Efficacy of TOCOSOL(R) Paclitaxel Versus Taxol(R) for Treatment of Metastatic Breast Cancer | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT00508625 | A Study of AMG 951 [rhApo2L/TRAIL] in Subjects With Previously Untreated Non-Small Cell Lung Cancer (NSCLC) Treated With Chemotherapy +/- Bevacizumab | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT02581501 | Prospective Phase I Study of GAX for Metastatic Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT01310244 | MTD Study PXD-101 in Combination With Paclitaxel + Carboplatin in Chemotherapy-Naive Patients With Stage IV NSCLC | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT03756818 | TAK-659 and Paclitaxel in Treating Patients With Advanced Solid Tumors | ovarian carcinoma | Paclitaxel (NPC208553) | |
| NCT00603941 | A Phase 1/2 Study of CS7017, an Oral PPARγ Agonist, in Combination With Paclitaxel | thyroid cancer | Paclitaxel (NPC208553) | |
| NCT00408070 | Bevacizumab Study With Carboplatin & Paclitaxel in Ovarian, Fallopian Tube or Primary Peritoneal Cancer | fallopian tube cancer;ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT05390710 | PhI to Solid Tumors and PhII to Locally Advanced or mTNBC | triple-negative breast cancer | Paclitaxel (NPC208553) | |
| NCT03361319 | Combination Nab-paclitaxel (N-P) and Nintedanib or N-P and Placebo in Relapsed NSCLC Adenocarcinoma | lung adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT02651727 | Ph 1 Study of VS-4718, a FAK Inhibitor, in Combination With Nab-paclitaxel and Gemcitabine in Advanced Cancer Subjects | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00191646 | An Ovarian, Primary Peritoneal or Fallopian Tube Cancer Study for Patients That Have Not Received Prior Chemotherapy | Fallopian Tube Carcinoma;ovarian neoplasm;peritoneal neoplasm | Paclitaxel (NPC208553) | |
| NCT03281369 | A Study of Multiple Immunotherapy-Based Treatment Combinations in Patients With Locally Advanced Unresectable or Metastatic Gastric or Gastroesophageal Junction Cancer (G/GEJ) or Esophageal Cancer (Morpheus-Gastric and Esophageal Cancer) | esophageal carcinoma;gastric adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT04460352 | Chemoradiotherapy Followed by Planned Surgery or by Surveillance and Surgery Only When Needed for Oesophageal Cancer | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT04072263 | Apatinib and Etoposide Capsule Versus Weekly Paclitaxel in Patients With Platinum Resistant Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00807573 | Paclitaxel, Bevacizumab and Pemetrexed in Patients With Untreated, Advanced Non-Small Cell Lung Cancer Using Web-Based Data Collection, Patient Self-Reporting of Adverse Effects and Automated Response Assessment | lung cancer | Paclitaxel (NPC208553) | |
| NCT01364012 | A Study of Bevacizumab Versus Placebo in Combination With Carboplatin/Paclitaxel in Participants With Advanced or Recurrent Non-Squamous Non-Small Cell Lung Cancer Who Have Not Received Previous Chemotherapy | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT04551950 | Bintrafusp Alfa Combination Therapy in Participants With Cervical Cancer (INTR@PID 046) | cervical cancer | Paclitaxel (NPC208553) | |
| NCT00702975 | Study of Combination Therapy of Carboplatin -Gemcitabine Plus Bevacizumab Beyond Progression in Patients With Locally Advanced and/or Metastatic Non-small Cell Lung Cancer (NSCLC) Who Have Not Received Prior Systemic Therapy | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT03699449 | Efficacy and Safety Study of AVB-S6-500 in Patients With Platinum-Resistant Recurrent Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT05420636 | Iadademstat in Combination With Paclitaxel in Relapsed/Refractory SCLC and Extrapulmonary High Grade NET | small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00411138 | Randomized Trial of Radiation Therapy With or Without Chemotherapy for Endometrial Cancer | endometrial cancer | Paclitaxel (NPC208553) | |
| NCT00864253 | A Trial of ABI-007 Versus Dacarbazine in Previously Untreated Patients With Metastatic Malignant Melanoma | melanoma | Paclitaxel (NPC208553) | |
| NCT01281176 | High-Dose or Low-Dose Vorinostat in Combination With Carboplatin or Paclitaxel in Treating Patients With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT00003035 | Doxorubicin and Paclitaxel in Treating Women With Locally Advanced Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT04882462 | Sintilimab and Nimotuzumab Combined With Chemotherapy for the Treatment of R/M HNSCC. | head and neck squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00003008 | Paclitaxel in Treating Patients With AIDS-Related Kaposi's Sarcoma | sarcoma | Paclitaxel (NPC208553) | |
| NCT00993655 | AURELIA: A Study of Avastin (Bevacizumab) Added to Chemotherapy in Patients With Platinum-resistant Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT04844385 | Toripalimab Plus Neoadjuvant Chemotherapy Combined With Chemoradiotherapy for Locally Advanced Unresectable Esophageal Squamous Cell Carcinoma | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT04617067 | Paricalcitol Trial: Phase II, Open Label Clinical Trial of Paricalcitol in Combination With Gemcitabine/ Nab-Paclitaxel Therapy in Advanced Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00748553 | A Phase I/II Clinical Trial of Vidaza With Abraxane in Patients With Advanced/Metastatic Solid Tumors and Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT02717091 | Neoadjuvant FOLFIRINOX or Nab-paclitaxel With Gemcitabine for Borderline Resectable Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT04269200 | Durvalumab With or Without Olaparib as Maintenance Therapy After First-Line Treatment of Advanced and Recurrent Endometrial Cancer | endometrial neoplasm | Paclitaxel (NPC208553) | |
| NCT00825201 | A Phase I/II Study of Paclitaxel Plus Carboplatin Plus Vorinostat in Recurrent Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT02276560 | Cisplatin and Nab-paclitaxel for (N2) Defined NSCLC | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00003930 | Radiation Therapy and Combination Chemotherapy in Treating Patients With Stage II or Stage III Bladder Cancer | urinary bladder cancer | Paclitaxel (NPC208553) | |
| NCT03606967 | Testing the Addition of an Individualized Vaccine to Nab-Paclitaxel, Durvalumab and Tremelimumab and Chemotherapy in Patients With Metastatic Triple Negative Breast Cancer | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT00861627 | Phase 2 Study of REOLYSIN® in Combination With Paclitaxel and Carboplatin for Non-Small Cell Lung Cancer With KRAS or EGFR Activation | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00323583 | Weekly Dosing of an Integrative Chemotherapy Combination to Treat Advanced Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT03585062 | Trial of Neoadjuvant Chemotherapy With S1 Plus Paclitaxel-albumin on Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT02461043 | Adjuvant Chemotherapy With Paclitaxel and Cisplatin in Lymph Node-Positive Adjuvant Chemotherapy in the Treatment of the Lymph Node Positive Thoracic Esophageal Squamous Cell Carcinoma | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT02001272 | LDE225 and Paclitaxel in Solid Tumors | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT02607202 | A Study of Nab-paclitaxel-based Doublet as First Line Therapy in Patients With Cancer of Unknown Primary (CUP) | neoplasm | Paclitaxel (NPC208553) | |
| NCT00518284 | Prevention of Restenosis Following Revascularization | peripheral vascular disease | Paclitaxel (NPC208553) | |
| NCT04679064 | Efficacy and Safety of Paclitaxel (Albumin-bound) Combination With Carboplatin in Ovarian Cancer. | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00567554 | Bevacizumab, Everolimus (RAD001), and Lapatinib as Neoadjuvant Chemotherapy Regimes for Primary Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT01367002 | Evaluation of Carboplatin/Paclitaxel With and Without Trastuzumab (Herceptin) in Uterine Serous Cancer | endometrial cancer | Paclitaxel (NPC208553) | |
| NCT03394885 | Study of GEN-1 With NACT for Treatment of Ovarian Cancer (OVATION 2) | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00003327 | Paclitaxel in Treating Patients With Recurrent or Refractory Head and Neck Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT03279237 | A Pilot Study of FOLFIRINOX in Combination With Neoadjuvant Radiation for Gastric and GE Junction Cancers | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00004011 | S9900: Surgery With or Without Combination Chemotherapy in Treating Patients With Non-small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00050167 | Evaluation of Differing Taxanes/Taxane Combinations on the Outcome of Patients With Operable Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT04831320 | Nab-Paclitaxel in Combination With Nivolumab to Treat Recurrent or Metastatic Head and Neck Squamous-Cell Carcinoma That Progressed on a PD-1 or PD-L1 Inhibitor | head and neck squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT04581876 | The Safety and Efficacy of the Combination of Raltitrexed for Injection and Nab-Paclitaxel in Advanced Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT02060253 | Ganetespib, Paclitaxel, Trastuzumab and Pertuzumab for Metastatic Human Epidermal Growth Factor Receptor 2 Positive Breast Cancer | male breast carcinoma | Paclitaxel (NPC208553) | |
| NCT00276796 | Paclitaxel, Topotecan, and Cisplatin in Treating Patients With Advanced, Persistent, or Recurrent Cervical Cancer | cervical cancer | Paclitaxel (NPC208553) | |
| NCT00081042 | ABI-007 in Treating Patients With Inoperable Locally Recurrent or Metastatic Melanoma | cutaneous melanoma | Paclitaxel (NPC208553) | |
| NCT01081951 | Intraperitoneal vs Intravenous Chemotherapy Following Neoadjuvant Chemotherapy in Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00147680 | Uterine Papillary Serous Cancer (UPSC) Trial | uterine cancer | Paclitaxel (NPC208553) | |
| NCT02744898 | A Phase 2 Feasibility Study of Abraxane and Carboplatin in Epithelial Neoplasms of the Uterus | endometrial cancer | Paclitaxel (NPC208553) | |
| NCT00666692 | A Phase 1b Study With Volociximab in Combination With Carboplatin, Paclitaxel, and Bevacizumab in First-line, Advanced Non-small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00786110 | Sorafenib Plus Paclitaxel in Adreno-Cortical-Cancer Patients | adrenal cortex carcinoma | Paclitaxel (NPC208553) | |
| NCT00208936 | Phase II Study of Pre-Operative Chemotherapy in Patients With Resectable Local-Regional Carcinoma of Esophagus | gastrointestinal disease | Paclitaxel (NPC208553) | |
| NCT04804696 | Toripalimab With Paclitaxel and Cisplatin as Neoadjuvant Treatment for Esophageal Squamous Cell Carcinoma | squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00003111 | Combination Chemotherapy Followed by Surgery in Treating Patients With Stage IIIA Non-small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT01847677 | Neoadjuvant Therapy in Advanced Ovarian Cancer With Avastin | ovarian carcinoma | Paclitaxel (NPC208553) | |
| NCT02272699 | Neoadjuvant Treatment of Nimotuzumab With Chemotherapy or Radiotherapy in Resectable Esophageal Squamous Cell Carcinoma | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT03278626 | Immune Checkpoint Therapy With Nivolumab Esophageal Squamous Cell Carcinoma | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00971867 | Rollover Study of Weekly Paclitaxel (BMS-181339) in Patients With Head and Neck Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT00005831 | Trastuzumab and Combination Chemotherapy in Treating Patients With Locally Recurrent or Metastatic Urinary Tract Cancer | urinary bladder carcinoma | Paclitaxel (NPC208553) | |
| NCT00521781 | Study of Abraxane Plus Hormonal Therapy as Initial Treatment of Unresectable or Metastatic Prostate Cancer | prostate adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT04211012 | A Study on Toripalimab Plus Nab-Paclitaxel With or Without Cisplatin as First-line Treatment of Urothelial Carcinoma | urothelial carcinoma | Paclitaxel (NPC208553) | |
| NCT01293630 | A Dose-escalation Study of Ombrabulin in Combination With Paclitaxel and Carboplatin in Patients With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT00298415 | Chemotherapy of Elderly Patients With Non-Small-Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00449657 | Phase II Trial of Pulsed Taxol With Concurrent Thoracic Radiotherapy, & Adjuvant Chemo in Stage III NSCLC | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT05192798 | Albumin-Bound Paclitaxel Combined With Antiangiogenic Agents in First-line Treatment of Relapsed or Metastatic TNBC | triple-negative breast cancer | Paclitaxel (NPC208553) | |
| NCT02322281 | TIGER-3: Open Label, Multicenter Study of Rociletinib (CO-1686) Mono Therapy Versus Single-agent Cytotoxic Chemotherapy in Patients With Mutant EGFR NSCLC Who Have Failed at Least One Previous EGFR-Directed TKI and Platinum-doublet Chemotherapy | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00391118 | Comparing Two Treatments for Ovarian Cancer: Standard Chemotherapy Plus Enzastaurin, or Placebo ("Sugar Pill") | Fallopian Tube Carcinoma;ovarian cancer;peritoneal neoplasm | Paclitaxel (NPC208553) | |
| NCT00147537 | Combination Study Of CP-751,871 With Paclitaxel And Carboplatin In Advanced Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT05213312 | Study of Neoadjuvant Nivolumab or Placebo Plus Chemotherapy Followed by Surgery and Adjuvant Treatment in Subjects With Resectable ESCC | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00731380 | A Phase I Trial of Abraxane, Cisplatin and 5-Fluorouracil Along With Chemoradiotherapy in Advanced Head and Neck Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT00174356 | PH 1 Evaluation Of Oral CI-1033 In Combination With Paclitaxel/ Carboplatin As 1st Line Chemotherapy In NSCLC Patients | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00004077 | Paclitaxel, Ifosfamide, and Cisplatin in Treating Patients With Metastatic Testicular Cancer | testicular neoplasm | Paclitaxel (NPC208553) | |
| NCT02677597 | Cisplatin Combined With S-1 or Paclitaxel as First-line Treatment for Metastatic Esophageal Squamous Cell Carcinoma | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT04625543 | Neoadjuvant Immunotherapy to ESCC | esophageal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00384826 | Therapeutic Strategy in Advanced Bronchioloalveolar Carcinoma | bronchoalveolar adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00003342 | Combination Chemotherapy in Treating Patients With Advanced Bladder or Kidney Cancer | urinary bladder cancer | Paclitaxel (NPC208553) | |
| NCT04159155 | A Study of Various Treatments in Serous or p53 Abnormal Endometrial Cancer | endometrial cancer | Paclitaxel (NPC208553) | |
| NCT02593708 | Phase1 of Neratinib+Trastuzumab, Pertuzumab, Paclitaxel in Patients With Advanced Solid Tumors/HER2+ | neoplasm | Paclitaxel (NPC208553) | |
| NCT02429700 | TC or BEP in Treating Patients With Ovarian Malignant Sex Cord-Stromal Tumors | ovarian sex cord-stromal tumor | Paclitaxel (NPC208553) | |
| NCT00087217 | 17-N-Allylamino-17-Demethoxygeldanamycin and Paclitaxel in Treating Patients With Metastatic or Unresectable Solid Tumor | neoplasm | Paclitaxel (NPC208553) | |
| NCT02892123 | Vevorisertib (ARQ 751) as a Single Agent or in Combination With Other Anti-Cancer Agents, in Solid Tumors With PIK3CA / AKT / PTEN Mutations (MK-4440-001) | cancer | Paclitaxel (NPC208553) | |
| NCT00003592 | Methotrexate Compared With Paclitaxel in Treating Patients With Advanced Head and Neck Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT00038402 | Evaluation of the Addition of Herceptin to Standard Chemotherapy in the Neoadjuvant Setting for Operable Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00851877 | Nab-Paclitaxel, Cisplatin, and Cetuximab With Concurrent Radiation Therapy for Locally Advanced Head and Neck Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT00702572 | Carboplatin, Paclitaxel, Bevacizumab and Vorinostat for Patients With Advanced Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00189553 | Caelyx Plus Carboplatin Versus Paclitaxel Plus Carboplatin in Patients With Epithelial Ovarian Cancer in Late Relapse | fallopian tube cancer;ovarian cancer | Paclitaxel (NPC208553) | |
| NCT01593228 | Treatment Extension Study for Patients Who Have Previously Participated and Have Benefited From Iniparib in a Clinical Trial | neoplasm | Paclitaxel (NPC208553) | |
| NCT00856492 | S0800, Nab-Paclitaxel, Doxorubicin, Cyclophosphamide, and Pegfilgrastim With or Without Bevacizumab in Treating Women With Inflammatory or Locally Advanced Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00367471 | Lapatinib Combined With Paclitaxel For Patients With First-Line ErbB2-Amplified Metastatic Breast Cancer | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT02476955 | Open-label Phase 1b Study of ARQ 092 in Combination With Anastrozole | endometrial cancer | Paclitaxel (NPC208553) | |
| NCT01935492 | 8 Continuous vs 8 Intermittent Cycles in First and Second Line in HER2/Neu Neg Metastatic Breast Cancer | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT01820325 | Safety and Efficacy of Buparlisib (BKM120) in Patients With Untreated Squamous Non-small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00028587 | PS-341 and Combination Chemotherapy in Treating Patients With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT03179904 | FASN Inhibitor TVB-2640 and Trastuzumab in Combination With Paclitaxel or Endocrine Therapy for the Treatment of HER2 Positive Metastatic Breast Cancer | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT03330106 | A Study to Evaluate the Effects of Pevonedistat on the Corrected QT (QTc) Interval in Participants With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT04856137 | A Phase I/II Study of Diffuse Large B-cell Lymphoma | lymphoma | Paclitaxel (NPC208553) | |
| NCT01055028 | Paclitaxel + Bevacizumab (Avastin) for the Treatment of Metastatic or Unresectable Angiosarcoma | angiosarcoma | Paclitaxel (NPC208553) | |
| NCT05328336 | Perioperative Tislelizumab Combined With Nab-Paclitaxel for Muscle-invasive Urothelial Bladder Cancer: A Multicenter Study | urinary bladder cancer | Paclitaxel (NPC208553) | |
| NCT04336098 | Study of SRF617 in Patients With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT04390763 | Study of Efficacy and Safety of NIS793 (With and Without Spartalizumab) in Combination With SOC Chemotherapy in First-line Metastatic Pancreatic Ductal Adenocarcinoma (mPDAC) | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00551733 | Carboplatin With Either Paclitaxel Poliglumex or Paclitaxel in Treating Women With Stage IIIB, Stage IV, or Recurrent Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT01633970 | A Study of Atezolizumab Administered in Combination With Bevacizumab and/or With Chemotherapy in Participants With Locally Advanced or Metastatic Solid Tumors | cancer | Paclitaxel (NPC208553) | |
| NCT02283489 | Recombinant Human Endostatin Adenovirus Combined With Chemotherapy for Advanced Head and Neck Malignant Tumors | upper aerodigestive tract neoplasm | Paclitaxel (NPC208553) | |
| NCT01649336 | TRINOVA-3: A Study of AMG 386 or AMG 386 Placebo in Combination With Paclitaxel and Carboplatin to Treat Ovarian Cancer | peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT00570531 | Phase II Trial for Patients With Loco-Regional Esophageal Carcinoma | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT05367206 | Neoadjuvant Chemotherapy Followed by Chemoradiation Versus Chemoradiation for Stage IIIC Cervical Cancer Patients: A Randomized Phase III Trial | cervical cancer | Paclitaxel (NPC208553) | |
| NCT04632992 | A Study to Determine the Safety, Tolerability, Pharmacokinetics, and Preliminary Efficacy of ABBV-368 Plus Tilsotolimod and Other Therapy Combinations in Participants With Recurrent/Metastatic Head and Neck Squamous Cell Carcinoma | cancer | Paclitaxel (NPC208553) | |
| NCT00112294 | Study of Taxane/Carboplatin +/- Cetuximab as First-Line Treatment for Patients With Advanced/Metastatic Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT04089150 | MFOLFIRINOX And Stereotactic Radiotherapy (SBRT) for Pancreatic Cancer With High Risk and Locally Advanced Disease | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00002917 | Paclitaxel in Treating Patients With Early-Stage Bladder Cancer | urinary bladder cancer | Paclitaxel (NPC208553) | |
| NCT03705429 | Study of Boserolimab (MK-5890) as Monotherapy and in Combination With Pembrolizumab (MK-3475) in Adults With Advanced Solid Tumors (MK-5890-001) | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT01491204 | Clinical Trial to Determine the Maximum Tolerated Dose and to Assess the Safety and Pharmacokinetic Profile of Oral Paclitaxel in Patients With Advanced Solid Cancer | neoplasm | Paclitaxel (NPC208553) | |
| NCT00270790 | EVALUATION OF AMIFOSTINE FOR MUCOSAL AND HEMOPOETIC PROTECTION AND CARBOPLATIN, TAXOL, RADIOTHERAPY IN THE MANAGEMENT OF PATIENTS WITH HEAD AND NECK CANCER.(GCC 0202) | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT00027937 | Combination Chemotherapy, Peripheral Stem Cell Transplantation, and Biological Therapy in Treating Patients With Solid Tumors or Lymphoma | lymphoma | Paclitaxel (NPC208553) | |
| NCT03678883 | Lapatinib Plus Chemotherapy Versus Trastuzumab Plus Chemotherapy in HER2-positive Breast Cancer | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT02057380 | A Rollover Study for Subjects That Have Participated in an Astellas Sponsored Linsitinib Trial | neoplasm | Paclitaxel (NPC208553) | |
| NCT02595554 | Neoadjuvant Chemotherapy and Radical Surgery in Stage IIB Cervical Cancer | cervical carcinoma | Paclitaxel (NPC208553) | |
| NCT04176952 | PRIMUS002: Looking at 2 Neo-adjuvant Treatment Regimens for Resectable and Borderline Resectable Pancreatic Cancer | neoplasm | Paclitaxel (NPC208553) | |
| NCT00158379 | Taxol Carboplatin and Erythropoetin | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00084565 | Paclitaxel, Topotecan, and Estramustine in Treating Patients With Metastatic Hormone-Refractory Prostate Cancer | prostate cancer | Paclitaxel (NPC208553) | |
| NCT01487226 | Adjuvant Chemotherapy in Patients With Lymph Node Metastasis After Radical Surgery in Cervical Cancer | cervical cancer | Paclitaxel (NPC208553) | |
| NCT03503786 | Carboplatin, Paclitaxel With or Without Avelumab in Advanced or Recurrent Endometrial Cancer | endometrial cancer | Paclitaxel (NPC208553) | |
| NCT00005849 | Bryostatin 1 Plus Paclitaxel in Treating Patients With Stage IIIB, Stage IV, or Recurrent Non-small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00448591 | A Study of Avastin (Bevacizumab) Plus Taxane-Based Therapy in Patients With Locally Recurrent or Metastatic Breast Cancer. | breast cancer | Paclitaxel (NPC208553) | |
| NCT02766582 | Avelumab in Previously Untreated Patients With Epithelial Ovarian Cancer (JAVELIN OVARIAN 100) | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00426556 | Efficacy and Safety of Everolimus in Combination Therapy, in Patients With HER2-overexpressing Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT02593175 | Women's MoonShot: Neoadjuvant Treatment With PaCT for Patients With Locally Advanced TNBC | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT00003294 | Chemotherapy Given With Amifostine and Filgrastim in Treating Patients With Recurrent or Metastatic Cancer | leukemia;lymphoma | Paclitaxel (NPC208553) | |
| NCT01966003 | Efficacy and Safety Study of ABP 215 Compared With Bevacizumab in Patients With Advanced Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT04937673 | The Neoadjuvant Treatment of Locally Advanced Thoracic Esophageal Squamous Cell Carcinoma | squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT04526470 | Alpelisib and Paclitaxel in PIK3CA-altered Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT03167164 | QUILT-3.045: NANT Merkel Cell Carcinoma (MCC) Vaccine: Combination Immunotherapy in Subjects With MCC Who Have Progressed on or After PD-L1 Therapy | Merkel cell skin cancer | Paclitaxel (NPC208553) | |
| NCT00316199 | Efficacy Study of Gemcitabine-Paclitaxel to Treat Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00009932 | Combination Chemotherapy in Treating Patients With Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT04541108 | Phase 0 Master Protocol for CIVO Intratumoral Microdosing of Anti-Cancer Therapies | neoplasm | Paclitaxel (NPC208553) | |
| NCT00781612 | A Safety Extension Study of Trastuzumab Emtansine in Participants Previously Treated With Trastuzumab Emtansine Alone or in Combination With Other Anti-Cancer Therapy in One of the Parent Studies | metastasis | Paclitaxel (NPC208553) | |
| NCT00625573 | Phase II Trial of Abraxane and Capecitabine in Metastatic Colon Cancer (COL 01) | colorectal carcinoma | Paclitaxel (NPC208553) | |
| NCT00393068 | Chemotherapy, Radiation Therapy and Immunotherapy Prior to Surgery in Operable Esophageal Cancer | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00209612 | Phase I/II Study of Paclitaxel Plus CPT-11 in Pts. With 2nd Line Chemotherapy of Inoperable or Recurrent GC. | gastric cancer | Paclitaxel (NPC208553) | |
| NCT01911598 | A Study of MEHD7945A in Combination With Cisplatin and 5-Fluorouracil (5-FU) or Paclitaxel and Carboplatin in Participants With Recurrent/Metastatic Squamous Cell Carcinoma of the Head and Neck (R/M SCCHN) | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT03125902 | A Study of Atezolizumab and Paclitaxel Versus Placebo and Paclitaxel in Participants With Previously Untreated Locally Advanced or Metastatic Triple Negative Breast Cancer (TNBC) | triple-negative breast cancer | Paclitaxel (NPC208553) | |
| NCT00096343 | Paclitaxel and Carboplatin in Treating Women Who Are Undergoing Surgery for Newly Diagnosed, Locally Advanced Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00055601 | Combination Chemotherapy and Radiation Therapy With/Without Surgery In Patients With Stage II/III Bladder Cancer | urinary bladder cancer | Paclitaxel (NPC208553) | |
| NCT04935359 | Study of Efficacy and Safety of NIS793 in Combination With Standard of Care (SOC) Chemotherapy in First-line Metastatic Pancreatic Ductal Adenocarcinoma (mPDAC) - daNIS-2 | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00796991 | Drug-Drug Interaction - 3 Arm - Carboplatin/Paclitaxel, Dacarbazine | melanoma | Paclitaxel (NPC208553) | |
| NCT01515306 | A Study of Ramucirumab (IMC-1121B) and Paclitaxel in Participants With Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT00513786 | Evaluation of Carboplatin/Paclitaxel/Bevacizumab in the Treatment of Advanced Stage Endometrial Carcinoma | endometrial cancer | Paclitaxel (NPC208553) | |
| NCT00054548 | Combination Chemotherapy Plus Oblimersen in Treating Patients With Advanced Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT04888663 | Study of Ramucirumab, Trastuzumab and Paclitaxel in Patients With HER2-positive Recurrent/Metastatic Gastric Cancer (HER-RAM Study) | gastric cancer | Paclitaxel (NPC208553) | |
| NCT01893801 | Nab-Pac+Cis+Gem in Pts w Previously Untreated Metastatic PDA | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT01170663 | A Study of Paclitaxel With or Without Ramucirumab (IMC-1211B) in Metastatic Gastric Adenocarcinoma | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00231582 | High-dose Chemotherapy With Autologous Stem Cell Transplantation in Poor Prognosis Germ-cell Tumors: TAXIF II | testicular neoplasm | Paclitaxel (NPC208553) | |
| NCT01107626 | Bevacizumab or Pemetrexed Disodium Alone or In Combination After Induction Therapy in Treating Patients With Advanced Non-Squamous Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT02483104 | Open-label Phase 1b Study of ARQ 092 in Combination With Anastrozole | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00399126 | Phase I Trial of Paclitaxel With Perifosine | neoplasm | Paclitaxel (NPC208553) | |
| NCT05251038 | Study of Sotorasib Combined With Chemotherapy for Second Line Treatment of Pancreas Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT03520790 | Paricalcitol Plus Gemcitabine and Nab-paclitaxel in Metastatic Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT05294900 | Trial of Neoadjuvant Therapy With Paclitaxel and Carboplatin in Operable Locally Advanced Head and Neck Cancer Patients | head and neck squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT03600090 | Phase I Study of EOC202 Plus Paclitaxel in Chinese Patients With Metastatic Breast Cancer | neoplasm | Paclitaxel (NPC208553) | |
| NCT05044871 | Oregovomab in Combination With Bevacizumab Plus Chemo in BRCA Wild Type Platinum Sensitive Recurrent Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT01620190 | Paclitaxel Albumin-Stabilized Nanoparticle Formulation in Treating Patients With Previously Treated Advanced Non-small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT04705519 | Nab-paclitaxel Combined With Bevacizumab in the Treatment of Metastatic Neuroendocrine Carcinoma | neuroendocrine carcinoma | Paclitaxel (NPC208553) | |
| NCT00479856 | Lapatinib In Combination With Chemotherapy In Subjects With Relapsed Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00436566 | Doxorubicin and Cyclophosphamide Followed By Trastuzumab, Paclitaxel, and Lapatinib in Treating Patients With Early-Stage HER2-Positive Breast Cancer That Has Been Removed By Surgery | breast cancer | Paclitaxel (NPC208553) | |
| NCT04001829 | Docetaxel or Paclitaxel in Reducing Chemotherapy-Induced Peripheral Neuropathy in African American Patients With Stage I-III Breast Cancer | breast carcinoma | Paclitaxel (NPC208553) | |
| NCT03517449 | Lenvatinib in Combination With Pembrolizumab Versus Treatment of Physician's Choice in Participants With Advanced Endometrial Cancer (MK-3475-775/E7080-G000-309 Per Merck Standard Convention [KEYNOTE-775]) | endometrial neoplasm | Paclitaxel (NPC208553) | |
| NCT00003193 | Paclitaxel and Radiation Therapy Plus Chemoprotection With Amifostine in Treating Patients With Stage III or Stage IV Head and Neck Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT00723125 | Carboplatin, Abraxane, Avastin as Neoadjuvant Therapy for Her2-Negative Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT02470585 | Dose Dense Paclitaxel With Pembrolizumab (MK-3475) in Platinum Resistant Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT02399137 | A Phase 2 Study of MM-141 Plus Nab-paclitaxel and Gemcitabine in Front-line Metastatic Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT01858883 | Safety Study of Itacitinib (INCB039110) in Combination With Gemcitabine and Nab-Paclitaxel in Subjects With Advanced Solid Tumors | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT02212015 | Evaluation of Votrient in Angiosarcoma | angiosarcoma | Paclitaxel (NPC208553) | |
| NCT00456846 | First Line Therapy for Patients With Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00473889 | A Clinical Trial of Vorinostat (MK0683, SAHA) in Combination With FDA Approved Cancer Drugs in Patients With Advanced Non-Small Cell Lung Cancer (NSCLC)(0683-056) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00005065 | Chemotherapy, Radiation Therapy, and Surgery in Treating Patients With Stage IIIA Non-small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT03393507 | ROCKIF Trial: Re-sensitization of Carboplatin-resistant Ovarian Cancer With Kinase Inhibition of FAK | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT05491694 | To Evaluate the Efficacy of Toripalimab Combined With Chemotherapy After HIFU Induction in the Treatment of Early Triple-negative Breast Cancer | triple-negative breast cancer | Paclitaxel (NPC208553) | |
| NCT01616303 | A Controlled Study of the Effectiveness of Oregovomab (Antibody) Plus Chemotherapy in Advanced Ovarian Cancer | ovarian neoplasm | Paclitaxel (NPC208553) | |
| NCT03690739 | Evaluation of Symptom Benefit Rate of Trabectedin/PLD in Patients With Recurrent Ovarian Cancer | ovarian carcinoma | Paclitaxel (NPC208553) | |
| NCT04282070 | SHR-1701 in Patients With Recurrent/Metastatic Nasopharyngeal Carcinoma | nasopharyngeal neoplasm | Paclitaxel (NPC208553) | |
| NCT04594005 | CDK4/6 Tumor, Abemaciclib, Paclitaxel | neoplasm | Paclitaxel (NPC208553) | |
| NCT03126812 | Olaparib +/- Cediranib or Chemotherapy in Patients With Platinum-resistant Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT01730586 | Abraxane in CIMP-High Colorectal and Small Bowel Adenocarcinomas | colorectal carcinoma | Paclitaxel (NPC208553) | |
| NCT05481645 | Efficacy and Safety of TQB2450 Combined With Chemotherapy ± Anlotinib for Advanced Endometrial Cancer | endometrial cancer | Paclitaxel (NPC208553) | |
| NCT05171530 | Lenvatinib With Taxane Drugs Treatment for Advanced Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00096668 | Evaluation of Safety and Efficacy of TOCOSOL(R) Paclitaxel as Initial Treatment for Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT00004265 | Paclitaxel in Treating Patients With Recurrent or Refractory Non-small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT02158520 | Nab-Paclitaxel and Bevacizumab or Ipilimumab as First-Line Therapy in Treating Patients With Stage IV Melanoma That Cannot Be Removed by Surgery | cutaneous melanoma;metastatic melanoma | Paclitaxel (NPC208553) | |
| NCT02993731 | A Study of Napabucasin Plus Nab-Paclitaxel With Gemcitabine in Adult Patients With Metastatic Pancreatic Adenocarcinoma | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT02413320 | Neoadjuvant Study of Two Platinum Regimens in Triple Negative Breast Cancer | triple-negative breast cancer | Paclitaxel (NPC208553) | |
| NCT05168527 | The First Line Treatment of Fruquintinib Combined With Albumin Paclitaxel and Gemcitabine in Pancreatic Cancer Patients | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00833261 | Nab-Paclitaxel, Cetuximab, Cisplatin, and Radiation Therapy in Treating Patients With Recurrent Head and Neck Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT01236716 | Nab-Paclitaxel Treatment in Advanced Squamous Cell Carcinoma of Lung | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00954174 | Paclitaxel and Carboplatin or Ifosfamide in Treating Patients With Newly Diagnosed, Persistent or Recurrent Uterine, Ovarian, Fallopian Tube, or Peritoneal Cavity Cancer | Fallopian Tube Carcinoma;Uterine Carcinosarcoma | Paclitaxel (NPC208553) | |
| NCT01647828 | A Phase 1b/2 Study of OMP-59R5 (Tarextumab) in Combination With Nab-Paclitaxel and Gemcitabine in Subjects With Previously Untreated Stage IV Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT01485848 | A Study of MM-121 With Paclitaxel in Platinum Resistant/ Refractory Advanced Ovarian Cancers | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT02382263 | Nab-paclitaxel in Combination With Gemcitabine in Fragile Patients With Advanced Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT02937272 | A Study of LY3200882 in Participants With Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT02520154 | Dose-Escalation Study of Intraperitoneal (IP) Cisplatin, IV/IP Paclitaxel, IV Bevacizumab, and Oral Olaparib for Newly Diagnosed Ovarian, Primary Peritoneal, and Fallopian Tube Cancer | fallopian tube cancer | Paclitaxel (NPC208553) | |
| NCT00929162 | A Study Evaluating Intermittent and Continuous OSI-906 and Weekly Paclitaxel in Patients With Recurrent Epithelial Ovarian Cancer (and Other Solid Tumors) | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00005032 | Bcl-2 Antisense Oligodeoxynucleotide G3139 and Paclitaxel in Treating Patients With Recurrent Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT01088815 | Hedgehog Inhibitors for Metastatic Adenocarcinoma of the Pancreas | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT01649336 | Weekly Paclitaxel With or Without Pazopanib in Platinum Resistant or Refractory Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00006229 | Paclitaxel + Carboplatin With/ut BMS-275291 in Advanced or Metastatic Non-small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT02827201 | FIrst Line Treatment of Metastatic Pancreatic Cancer: Sequential Nab-paclitaxel + Gemcitabine/FOLFIRI.3 VS Nab-paclitaxel + Gemcitabine | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00520013 | Avastin +/- Erlotinib Consolidation Chemotherapy After Carboplatin, Paclitaxel, and Avastin (CTA) Induction Therapy for Advanced Ovarian, Fallopian Tube, Primary Peritoneal Cancer & Papillary Serous or Clear Cell Mullerian Tumors | fallopian tube cancer;ovarian cancer | Paclitaxel (NPC208553) | |
| NCT03287271 | Clinical Trial of Combined Fostamatinib and Paclitaxel in Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT04148885 | A Trial of Paclitaxel (Albumin-binding) for Castration-resistant Prostate Cancer | prostate cancer | Paclitaxel (NPC208553) | |
| NCT00527735 | Phase II Study for Previously Untreated Subjects With Non Small Cell Lung Cancer (NSCLC) or Small Cell Lung Cancer (SCLC) | small cell lung carcinoma;non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT03085056 | Trametinib in Combination With Paclitaxel in the Treatment of Anaplastic Thyroid Cancer | thyroid cancer | Paclitaxel (NPC208553) | |
| NCT04943445 | Study of a Pembrolizumab-based Organ Preservation Strategy for Locally Advanced Larynx Cancers | laryngeal squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT01827111 | Phase II Study of Abraxane Plus Ipilimumab in Patients With Metastatic Melanoma | melanoma | Paclitaxel (NPC208553) | |
| NCT04683315 | PurIST Classification-Guided Adaptive Neoadjuvant Chemotherapy by RNA Expression Profiling of EUS Aspiration Samples | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT03086369 | A Study of Nab-Paclitaxel and Gemcitabine With or Without Olaratumab (LY3012207) in Participants With Metastatic Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT03574324 | TPF Induction Chemotherapy vs PF Adjuvant Chemotherapy Combined With Concurrent Chemoradiotherapy in the Treatment of Locally Advanced NPC | nasopharyngeal neoplasm | Paclitaxel (NPC208553) | |
| NCT01015339 | Paclitaxel Plus Capecitabine With Capecitabine Maintenance Treatment in Metastatic Adenocarcinoma of the Stomach or Esophagogastric Junction | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00014118 | Chemotherapy and Radiation Therapy in Treating Patients With Stage III or Stage IV Larynx or Oropharynx Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT02020707 | Abraxane/Bevacizumab | peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT00033410 | Chemotherapy, Tirapazamine, and Radiation Therapy in Treating Patients With Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT02229058 | Albumin Bound Paclitaxel Plus S-1 as the First Line Chemotherapy in Advanced or Recurrent Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00041470 | Navelbine, Taxol, Herceptin and Neupogen in Stage IV Breast Cancer: A Phase I - II Trial | breast cancer | Paclitaxel (NPC208553) | |
| NCT01862328 | Dose Escalation, Multi-arm Study of MLN4924 Plus Docetaxel, Gemcitabine, or Combination of Carboplatin and Paclitaxel in Participants With Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT03428425 | A Study of HIPEC Plus Apatinib and S-1 Conversion Therapy for Gastric Cancer With Positive Exfoliative Cancer Cells | gastric cancer | Paclitaxel (NPC208553) | |
| NCT04647344 | A Study of AK104 in Patients With Locally Advanced or Metastatic Non-small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT02713386 | Pembrolizumab, Carboplatin, and Paclitaxel in Treating Patients With Stage III-IV Ovarian, Primary Peritoneal, or Fallopian Tube Cancer | fallopian tube cancer | Paclitaxel (NPC208553) | |
| NCT00581971 | Radiosensitization With Celecoxib and Chemoradiation for Head and Neck Cancer | cancer | Paclitaxel (NPC208553) | |
| NCT03639246 | A Study of the Efficacy and Safety of Bevacizumab in Chinese Women With Newly Diagnosed, Previously Untreated Stage III or Stage IV Epithelial Ovarian, Fallopian Tube, or Primary Peritoneal Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT04213898 | SHR-1210 Combined With Albumin-bound Paclitaxel and Epirubicin Neoadjuvant for Triple Negative Breast Cancer | triple-negative breast cancer | Paclitaxel (NPC208553) | |
| NCT00556088 | LBH589, Paclitaxel, Carboplatin +/- Bevacizumab for Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT01644825 | A Phase IIclinical Trial of Carboplatin and Paclitaxel or Carboplatin and Gemcitabine in Platinum-sensitive, Recurrent Ovarian, Fallopian Tube, and Primary Peritoneal Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00004934 | Paclitaxel and Carboplatin With or Without Epirubicin in Treating Patients With Stage IIB, Stage III, or Stage IV Invasive Ovarian Epithelial, Fallopian Tube, or Peritoneal Cancer | fallopian tube cancer;ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT02798536 | Mesothelin-Targeted Immunotoxin LMB-100 in People With Malignant Mesothelioma | mesothelioma | Paclitaxel (NPC208553) | |
| NCT00964626 | Combination Chemotherapy With CS-1008 to Treat Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT02545010 | Pembrolizumab, Carboplatin, and Paclitaxel in Treating Patients With Stage III-IV Ovarian, Primary Peritoneal, or Fallopian Tube Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00004057 | Paclitaxel Plus L-778,123 in Treating Patients With Recurrent or Refractory Solid Tumors or Lymphomas | lymphoma | Paclitaxel (NPC208553) | |
| NCT03252808 | Phase I Study of TBI-1401(HF10) Plus Chemotherapy in Patients With Unresectable Pancreatic Cancer. | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT02448329 | Study of AZD1775 in Combination With Paclitaxel, in Advanced Gastric Adenocarcinoma Patients Harboring TP53 Mutation as a Second-line Chemotherapy | gastric adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00079287 | Vinorelbine, Gemcitabine, and Docetaxel Compared With Paclitaxel and Carboplatin in Treating Patients With Advanced Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT04714190 | A Study of RC48-ADC in Local Advanced or Metastatic Gastric Cancer With the HER2-Overexpression | gastric carcinoma | Paclitaxel (NPC208553) | |
| NCT02931890 | Multicentric Randomised Trial for Resectable Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00070629 | CPG 7909 Injection in Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00251095 | Study Of SU011248 Versus Chemotherapy For Patients With Previously Treated Triple Receptor Negative Breast Cancer | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT00003733 | Sequential Chemotherapy in Treating Patients With Residual Disease Following Surgery for Stage IIB, Stage III, or Stage IV Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00596830 | Carboplatin And Paclitaxel With Or Without CP-751, 871 (An IGF-1R Inhibitor) For Advanced NSCLC Of Squamous, Large Cell And Adenosquamous Carcinoma Histology | large cell lung carcinoma;squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT03029611 | Pembrolizumab, Paclitaxel, and Carboplatin in Patients With Advanced Stage Epithelial Ovarian Cancer (EOC). | fallopian tube cancer | Paclitaxel (NPC208553) | |
| NCT00850577 | Ph II of a Novel Anti-angiogenic Agent in Combination With Chemotherapy for the Treatment of Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01220128 | Lapatinib or Trastuzumab Given Prior to Surgery With Chemotherapy in Patients With Early Breast Cancer | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT00003350 | Paclitaxel Compared With Doxorubicin in Treating Patients With Advanced AIDS-Related Kaposi's Sarcoma | Kaposi's sarcoma | Paclitaxel (NPC208553) | |
| NCT00654758 | A Phase 1b Study With Volociximab in Combination With Carboplatin and Paclitaxel in First-line, Advanced Non-Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01566240 | Induction Chemotherapy Plus Chemoradiation as First Line Treatment for Locally Advanced Cervical Cancer | cervical cancer | Paclitaxel (NPC208553) | |
| NCT00005612 | Combination Chemotherapy Followed by Peripheral Stem Cell Transplantation in Treating Patients With Ovarian Epithelial Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00454324 | Study of Abraxane and Carboplatin to Treat Small Cell Lung Cancer | small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00462423 | Abraxane and Avastin as Therapy for Patients With Malignant Melanoma, a Phase II Study | melanoma | Paclitaxel (NPC208553) | |
| NCT05255471 | Antitumor Activity of Neoadjuvant Chemotherapy With or Without BINTRAFUSP ALFA in Patients With Metastatic Advanced Stage Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT04483076 | Oxaliplatin Combined With S-1(SOX) Neoadjuvant Chemotherapy for Different Cycles in Patients With Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT01779050 | Afatinib in HER2 (Human Epidermal Growth Factor Receptor)-Treatment Failures | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT00033553 | Combination Chemotherapy and Computer-Planned Radiation Therapy in Treating Patients With Unresectable Stage III Non-Small Cell Lung Cancer | lung cancer | Paclitaxel (NPC208553) | |
| NCT00075270 | Paclitaxel With / Without GW572016 (Lapatinib) As First Line Therapy For Women With Advanced Or Metastatic Breast Cancer | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT02152137 | Inolitazone Dihydrochloride and Paclitaxel in Treating Patients With Advanced Anaplastic Thyroid Cancer | thyroid cancer | Paclitaxel (NPC208553) | |
| NCT05346146 | A Prospective, Single-arm, Exploratory Clinical Study of Sintilimab Injection in Combination With Paclitaxel (Albumin-bound) and Gemcitabine in Translational Therapy in Patients With Unresectable Locally Advanced Pancreatic Cancer. | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT05145218 | Biomarker-driven Targeted Therapy in Patients With Recurrent Platinum-resistant Epithelial Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00724386 | Concomitant Chemoradiotherapy With Weekly Paclitaxel and Vinorelbine and Granulocyte Colony Stimulating Factor (GCSF) Support in Patients With Advanced Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT03912402 | Efficacy and Safety of BCD-100 (Anti-PD-1) in Combination With Platinum-Based Chemotherapy and Bevacizumab in Patients With Recurrent, Persistent or Metastatic Cervical Cancer (CAESURA) | cervical cancer | Paclitaxel (NPC208553) | |
| NCT03018080 | Pilot Study of Paclitaxel Plus Pembrolizumab in Metastatic HER2-Negative Breast Cancer | male breast carcinoma | Paclitaxel (NPC208553) | |
| NCT03801668 | Albumin-bound Paclitaxel Plus S-1 Versus SOX as First-line Treatment in Advanced or Recurrent Gastric Adenocarcinoma | gastric cancer | Paclitaxel (NPC208553) | |
| NCT01042379 | I-SPY TRIAL: Neoadjuvant and Personalized Adaptive Novel Agents to Treat Breast Cancer | angiosarcoma;triple-negative breast cancer | Paclitaxel (NPC208553) | |
| NCT01992341 | AMG 386 Drug-Drug Interaction Study With Paclitaxel | neoplasm | Paclitaxel (NPC208553) | |
| NCT00454649 | Investigational Agent AG-013736 In Combinations With Standard Of Care Treatments For Patient's With Advanced Solid Tumor | neoplasm | Paclitaxel (NPC208553) | |
| NCT03416153 | Individualized Adaptive De-escalated Radiotherapy for HPV-related Oropharynx Cancer | oropharynx cancer | Paclitaxel (NPC208553) | |
| NCT00028743 | Combination Chemotherapy Regimens in Ovarian Epithelial Cancer, Primary Peritoneal Cancer, or Fallopian Tube Cancer | fallopian tube cancer;ovarian cancer;peritoneum cancer | Paclitaxel (NPC208553) | |
| NCT00356811 | Combined Treatment of Cetuximab and Paclitaxel in Basal Like Breast Carcinoma | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT00723957 | A Randomized Phase 2 Study of Ixabepilone Plus Carboplatin and Paclitaxel Plus Carboplatin in Advanced Nonsmall-Cell Lung Cancer | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01617928 | A Study of Veliparib in Combination With Carboplatin and Paclitaxel in Japanese Subjects With Solid Tumors | neoplasm | Paclitaxel (NPC208553) | |
| NCT03003546 | Nab-paclitaxel/Rituximab-coated Nanoparticle AR160 in Treating Patients With Relapsed or Refractory B-Cell Non-Hodgkin Lymphoma, LS1681 Trial | chronic lymphocytic leukemia | Paclitaxel (NPC208553) | |
| NCT00475670 | A Study of Herceptin (Trastuzumab) in Women With Metastatic Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT01283204 | Trial of 4-regimen (SP, FL/Tax, FL/Doc, FOLFOX) in Patients With Recurrent or Metastatic Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00844649 | Phase III Study of ABI-007(Albumin-bound Paclitaxel) Plus Gemcitabine Versus Gemcitabine in Metastatic Adenocarcinoma of the Pancreas | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT05241249 | Stimulation With Bethanechol in Combination With Gemcitabine and Nab-paclitaxel in Pancreatic Adenocarcinoma | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00170664 | Taxol/ Carboplatin as First Line Therapy in Patients With Epithelial Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT00191672 | A Trial Of Gemcitabine Plus Paclitaxel And Gemcitabine Plus Docetaxel In Metastatic Breast Cancer | breast neoplasm | Paclitaxel (NPC208553) | |
| NCT00054028 | Suramin and Paclitaxel in Treating Women With Stage IIIB-IV Breast Cancer | breast cancer | Paclitaxel (NPC208553) | |
| NCT04188860 | Immunotherapy for Recurrent Cervical Cancer Refractory to Platinum-based Chemotherapy | cervical carcinoma | Paclitaxel (NPC208553) | |
| NCT01084083 | Induction Chemotherapy Followed By Cetuximab and Radiation in HPV-Associated Resectable Stage III/IV Oropharynx Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT00066716 | Celecoxib, Paclitaxel, and Carboplatin in Treating Patients With Cancer of the Esophagus | esophageal cancer | Paclitaxel (NPC208553) | |
| NCT00001384 | A Pilot Trial of AC (Adriamycin, Cyclophosphamide) Chemotherapy With G-CSF (Granulocyte Colony-Stimulating Factor) Followed by Infusional Taxol (Paclitaxel) as Adjuvant Treatment for High Risk Stage II and Stage III Breast Cancer Patients | breast cancer | Paclitaxel (NPC208553) | |
| NCT00016341 | Combination Chemotherapy Compared With Hormone Therapy in Treating Patients With Recurrent, Stage III, or Stage IV Endometrial Cancer | endometrial cancer | Paclitaxel (NPC208553) | |
| NCT01024062 | Phase II Study of Weekly Paclitaxel (BMS-181339) in Patient With Non-Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT00828009 | BLP25 Liposome Vaccine and Bevacizumab After Chemotherapy and Radiation Therapy in Treating Patients With Newly Diagnosed Stage IIIA or Stage IIIB Non-Small Cell Lung Cancer That Cannot Be Removed by Surgery | lung cancer | Paclitaxel (NPC208553) | |
| NCT03496662 | BMS-813160 With Nivolumab and Gemcitabine and Nab-paclitaxel in Borderline Resectable and Locally Advanced Pancreatic Ductal Adenocarcinoma (PDAC) | pancreatic ductal adenocarcinoma | Paclitaxel (NPC208553) | |
| NCT00176241 | Phase I Trial of Biweekly Gemcitabine & Paclitaxel & Low-Dose Radiation for Metastatic or Recurrent Head & Neck Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT03563144 | QUILT-3.088: NANT Pancreatic Cancer Vaccine | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT00933374 | Trial to Evaluate Paclitaxel Plus RAD001 in Urothelial Carcinoma | urinary bladder cancer | Paclitaxel (NPC208553) | |
| NCT04428151 | Lenvatinib (E7080/MK-7902) in Combination With Pembrolizumab (MK-3475) vs. Standard Chemotherapy and Lenvatinib Monotherapy in Participants With Recurrent/Metastatic Head and Neck Squamous Cell Carcinoma That Progressed After Platinum Therapy and Immunotherapy (MK-7902-009/E7080-G000-228/LEAP-009) | head and neck squamous cell carcinoma | Paclitaxel (NPC208553) | |
| NCT00089297 | Cetuximab, Chemotherapy, and Radiation Therapy for Operable Stage III or IV Head and Neck Cancer | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT01491217 | A Study of Oraxol® in Gastric Cancer Patients | gastric cancer | Paclitaxel (NPC208553) | |
| NCT02207465 | A Phase I Dual Dose Escalation Study of Radiation and Nab-Paclitaxel in Patients With Unresectable and Borderline Resectable Pancreatic Cancer | pancreatic carcinoma | Paclitaxel (NPC208553) | |
| NCT01929941 | An Open-Label Study of a Novel JAK-inhibitor, INCB047986, Given in Patients With Advanced Malignancies | Hodgkins lymphoma | Paclitaxel (NPC208553) | |
| NCT02319889 | Feasibility Study of SBRT Plus Chemotherapy for Non-Small Cell Lung Carcinoma | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01167712 | Paclitaxel and Carboplatin With or Without Bevacizumab in Treating Patients With Stage II, Stage III, or Stage IV Ovarian Epithelial Cancer, Primary Peritoneal Cancer, or Fallopian Tube Cancer | endometrioid carcinoma;Fallopian Tube Carcinoma | Paclitaxel (NPC208553) | |
| NCT03398655 | Atezolizumab With Neoadjuvant Chemotherapy for Patients With Newly-Diagnosed Advanced-Stage Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT01980472 | Chemotherapy Plus Bevacizumab in Elderly Non-small Cell Lung Cancer Patients | non-small cell lung carcinoma | Paclitaxel (NPC208553) | |
| NCT01412229 | Induction Chemotherapy for Locally Advanced Squamous Cell Carcinoma of the Head and Neck | head and neck malignant neoplasia | Paclitaxel (NPC208553) | |
| NCT01694576 | NPC Staged N2-3M0:Adjuvant Chemotherapy or Just Observation After Concurrent Chemoradiation | nasopharyngeal neoplasm | Paclitaxel (NPC208553) | |
| NCT01136031 | Paclitaxel and Irinotecan in Advanced Gastric Cancer | gastric cancer | Paclitaxel (NPC208553) | |
| NCT00503750 | Phase II Neoadjuvant Trial of Trastuzumab in Combination With Dose-Dense ABI-007 (Abraxane™) | breast cancer | Paclitaxel (NPC208553) | |
| NCT02012192 | A Study to Evaluate the Effect of the Combination of Pertuzumab With Carboplatin-Based Standard Chemotherapy in Patients With Recurrent Ovarian Cancer | ovarian cancer | Paclitaxel (NPC208553) | |
| NCT02739529 | Study to Evaluate the Efficacy and Safety of Genexol-PM Once a Week for Gynecologic Cancer | female reproductive system neoplasm | Paclitaxel (NPC208553) |
❱❱❱ Associated Human Diseases and Detailed Association Evidence
How do we define the Plant-Targeted Human Disease Association?
Associated human diseases of an individual plant are summurized based on FOUR types of association evidence, these include:
❶ Association by Therapeutic Target: Bioactive protein targets of the plant were defined in "Molecular Targets" section, target-disease associations collected from TTD database were subsequently used to build the associations between the plant and its targeted human diseases.
❷ Association by Disease Gene Reversion: Plant and a specific disease will be associated when >= 1 plant target gene overlaped with disease's DEGs.
❸ Association by Clinical Trials of Plant: Plant and a specific disease will be associated when >= 1 clinical trial (the plant is the intervetion) can be matched in ClinicalTrials.gov database.
❹ Association by Clinical Trials of Plant Ingredients: Plant and a specific disease will be associated when >= 1 clinical trial (the plant ingredient is the intervetion) can be matched in ClinicalTrials.gov database.
Associated Disease of the Plant |
Association Type & Detailed Evidence |
|---|---|
Pleomorphic sarcomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B54 |
MTOR
NCT00020449,NCT03678883,NCT00003128,NCT00112489,NCT00003624,NCT00003008,NCT00217607,NCT00003054 |
Endometrioid adenocarcinoma, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH0SD2 |
NCT00466960,NCT00262847,NCT01167712,NCT02713386,NCT00085358,NCT00079430,NCT00002913,NCT00989651,NCT00108745
|
Malignant neoplasms of stomachDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B72 |
NCT04267549,NCT03322969,NCT04155671,NCT04483076,NCT04683939,NCT03579784,NCT04149015,NCT05441254,NCT01183559,NCT02934464,NCT05052931,NCT02697838,NCT01457846,NCT03751761,NCT04694183,NCT05319639,NCT05002686,NCT04888663,NCT03026881,NCT05190445,NCT04195828,NCT05070598,NCT04182724,NCT04835896,NCT04308837,NCT04781413,NCT01248403,NCT01170663,NCT05025033,NCT04258657,NCT05427383,NCT05171530,NCT01924533,NCT01283204,NCT01491217,NCT01471132,NCT04602117,NCT05095467,NCT02038621,NCT04286711,NCT02451956,NCT02486601,NCT02855788,NCT02229058,NCT02072317,NCT02442362,NCT05410847,NCT04001569,NCT02574663,NCT01739894,NCT00003298,NCT02845908,NCT05204173,NCT04342910,NCT03977220,NCT03286244,NCT00154726,NCT01980810,NCT04943653,NCT01063517,NCT01839773,NCT02931890,NCT03428425,NCT01249443,NCT04592211,NCT03718624,NCT02890511,NCT05400915,NCT04563975,NCT01746771,NCT01224652,NCT01015339,NCT00003862,NCT00209612,NCT03704077,NCT00661167,NCT03475615,NCT04526470,NCT04258644,NCT01136031,NCT03801668,NCT04135781,NCT03766607,NCT01641939,NCT03263741
|
Squamous cell carcinomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B60-2D01 |
NCT04764227,NCT00753038,NCT05322499,NCT03866993,NCT04063683,NCT05319730,NCT01969955,NCT03387111,NCT00998192,NCT03940001,NCT05189730,NCT02027428,NCT01591135,NCT05312372,NCT04625543,NCT04472429,NCT03946969,NCT02059967,NCT04767295,NCT04354961,NCT01166542,NCT02916511,NCT02459457,NCT03762564,NCT00176254,NCT03975270,NCT02350517,NCT01258192,NCT00001442,NCT04804696,NCT00816634,NCT03748134,NCT04138212,NCT05007145,NCT02976909,NCT02272699,NCT05394415,NCT03957590,NCT04718415,NCT04929041,NCT03278626,NCT04888403,NCT00596830,NCT01822613,NCT03964753,NCT04844385,NCT05213312,NCT05281003,NCT03292822,NCT04568200,NCT01386385,NCT02784288,NCT01225523,NCT04949256,NCT04548440,NCT02611700,NCT04943445,NCT03603756,NCT01249443,NCT04225364,NCT00042835,NCT04644250,NCT02033538,NCT05342636,NCT01688700,NCT04807673,NCT01752205,NCT04084158,NCT04506138,NCT04460352,NCT03430843,NCT03719924,NCT05449483,NCT05125055,NCT00176267,NCT04937673,NCT02677597,NCT03116152,NCT04390958,NCT03783442
|
Esophageal cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B70 |
NCT03308552,NCT00493025,NCT00008047,NCT02279134,NCT00154700,NCT00686114,NCT02644863,NCT00154804,NCT00344552,NCT04046575,NCT00139633,NCT03328234,NCT00107341,NCT01704690,NCT00655876,NCT00006245,NCT02598687,NCT00815308,NCT02461043,NCT00971841,NCT00193141,NCT00453323,NCT02317991,NCT02607982,NCT02429622,NCT02762474,NCT00230451,NCT01372202,NCT05013697,NCT00281788,NCT02389751,NCT05357846,NCT03490292,NCT02861690,NCT03126708,NCT00002711,NCT03279237,NCT00730353,NCT02924909,NCT03691090,NCT01998347,NCT03281369,NCT01183559,NCT04821765,NCT00016900,NCT00021320,NCT00002984,NCT04006041,NCT03165994,NCT04460066,NCT02446574,NCT02297217,NCT05174156,NCT02570893,NCT04879368,NCT00066716,NCT02569242,NCT02273713,NCT00069953,NCT00570531,NCT01444547,NCT02133612,NCT04005170,NCT01843829,NCT00393068,NCT02741856,NCT02891083,NCT00006472,NCT00003326,NCT04278287,NCT00003087,NCT02858206,NCT03087864,NCT01249443,NCT02735239,NCT02574663,NCT01034189,NCT03731442,NCT04426955,NCT04540211
|
AngiosarcomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B56 |
NCT04339738,NCT01303497,NCT04859465,NCT02212015,NCT03512834,NCT03544567,NCT01055028,NCT01042379
|
Multiple myelomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A83 |
MTOR
NCT01889420,NCT02985333,NCT00003399,NCT01646762,NCT00003401,NCT02075021 |
Colorectal cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B91 |
ITGAV
NCT02103062,NCT00024401,NCT03169777,NCT02574663,NCT01730586,NCT00625573 |
Urethral cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C93 |
NCT00022633,NCT00045630,NCT00003376,NCT04579224,NCT00478361,NCT00022191,NCT00005644
|
Biliary tract cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C17 |
NCT03830606,NCT02632305,NCT04492033,NCT04027764,NCT04692051,NCT05170438,NCT04004234
|
Malignant neoplasms of cervix uteri, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C77.Z |
NCT02312804,NCT03556839,NCT02595554,NCT02492503,NCT05290935,NCT04652076,NCT04188860
|
Fallopian tube cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C74 |
NCT00226915,NCT00091377,NCT00993655,NCT01402271,NCT01083537,NCT00003896,NCT01649336,NCT00003160,NCT00838656,NCT03740165,NCT00466960,NCT03635489,NCT00079430,NCT02520154,NCT03462212,NCT01219777,NCT00003624,NCT02713386,NCT00008138,NCT00550784,NCT00373217,NCT00189566,NCT01091428,NCT00408070,NCT00520013,NCT00004934,NCT03056833,NCT01570582,NCT02834975,NCT00031954,NCT00085358,NCT02012192,NCT01493505,NCT00672295,NCT03038100,NCT00652119,NCT00052468,NCT01447706,NCT03393884,NCT00702299,NCT00028743,NCT00003385,NCT00006454,NCT00189553,NCT00517621,NCT01220154,NCT01204749,NCT00003120,NCT03029611,NCT02050009,NCT00331422,NCT01253681,NCT00003064,NCT02121990,NCT01196741,NCT00075712,NCT00483782,NCT01146795,NCT00003944,NCT00466986,NCT03126812,NCT00479817
|
Thyroid cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D10 |
NCT01701349,NCT00507429,NCT02152137,NCT03085056,NCT00786552,NCT00603941
|
Neoplasms of unknown behaviour of urinary organsDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2F98 |
NCT00478361,NCT04876313,NCT00005831,NCT05024773,NCT05203913,NCT02560038
|
Urogenital cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C95-2C9Z |
NCT00151034,NCT00034177,NCT02033993,NCT02256436,NCT04527991,NCT02756013
|
Transitional cell carcinoma, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH8EH1 |
NCT03871036,NCT03464734,NCT04603846,NCT03240016,NCT04211012,NCT03197571
|
GerminomaDisease Category: X.Extension CodesDisease ICD-11 Code: XH1E13 |
NCT00864318,NCT02414685,NCT00611962,NCT04581265,NCT01873326,NCT05455918
|
Adenocarcinoma, endocervical typeDisease Category: X.Extension CodesDisease ICD-11 Code: XH0GS9 |
NCT00064077,NCT02020707,NCT00295789,NCT04723875,NCT04516616,NCT01755897
|
Uterine ligament/parametrium/uterine adnexa neoplasmDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C72 |
NCT00584909,NCT01650376,NCT00506779,NCT00147680,NCT00584857,NCT00231868
|
Diffuse large B-cell lymphomasDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A81 |
TUBB6,PMP22,GMNN,ITGAV,THRB,LMNA
|
Adenocarcinoma of pancreasDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C10.0 |
TUBB6,SLCO1B3,NPC1,PMP22,ITGAV,LMNA
|
AdenocarcinomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D40 |
NCT02303977,NCT02047513,NCT02059967,NCT00384826,NCT00955305,NCT01506973,NCT04046887,NCT02426281,NCT00226746,NCT00369551,NCT00005838,NCT02723331,NCT03908333,NCT04329949,NCT02926183,NCT02186847,NCT00368992,NCT02468557,NCT01578551,NCT05257993,NCT01253525,NCT03361319,NCT01676259,NCT02754726,NCT04821284,NCT02101021,NCT03697239,NCT01218516,NCT01431794,NCT03410030,NCT04827953,NCT02583477,NCT02501902,NCT03517176,NCT00042835,NCT05042128,NCT02243007,NCT04481204,NCT02715804,NCT03496662,NCT02487277,NCT04134468,NCT04469556,NCT00040794,NCT03915444,NCT02993731,NCT04581343,NCT02732938,NCT00021060,NCT04390763,NCT04634539,NCT00201734,NCT02048943,NCT00002852,NCT04257448,NCT04929041,NCT03345810,NCT04935359,NCT05254171
|
Neoplasms of unknown behaviour of breastDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2F95 |
NCT00046514,NCT00174434,NCT00356811,NCT01220128,NCT02455141,NCT03742102,NCT01572038,NCT00191672,NCT01271725,NCT02622074,NCT00849472,NCT00553358,NCT01953536,NCT04895358,NCT03036488,NCT00075270,NCT00373256,NCT00388076,NCT00194753,NCT02162719,NCT00046527,NCT00110695,NCT02051751,NCT00048893,NCT00272987,NCT01779050,NCT00201435,NCT03396445,NCT00448305,NCT03678883,NCT00709761,NCT03997123,NCT03272477,NCT05244993,NCT01953445,NCT04251533,NCT00251095,NCT00353717,NCT00246571,NCT03273595,NCT02776917,NCT01137994,NCT00524303,NCT00281658,NCT00429299,NCT05067530,NCT02307227,NCT00550771,NCT00367471,NCT01138046,NCT01205217,NCT01026116,NCT03705429,NCT00111787
|
Unspecified malignant neoplasms of unspecified sitesDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D4Z |
NCT01653470,NCT02366949,NCT02193633,NCT02383212,NCT04632992,NCT03328494,NCT02761694,NCT01497470,NCT01204996,NCT01376310,NCT02483247,NCT01636622,NCT00002854,NCT02892123,NCT01604863,NCT00113438,NCT01633970,NCT01351350,NCT04196283,NCT03507491,NCT02138812,NCT00358163,NCT01325441,NCT02412722,NCT02630199,NCT00473720,NCT00881166,NCT00337376,NCT01187199,NCT01301716,NCT01705483,NCT01113476,NCT01015222,NCT03498521,NCT02251951,NCT01075464,NCT00798252,NCT05081609,NCT00508326,NCT01688791,NCT02327169,NCT01579578,NCT01110603,NCT00581971,NCT02640755,NCT01192165,NCT05235542,NCT00954642,NCT01235897,NCT01057264,NCT01374620,NCT02240212,NCT02317419
|
Malignant neoplasms of retroperitoneumDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C50 |
NCT00702299,NCT01493505,NCT00091377,NCT00466960,NCT00003944,NCT00031954,NCT02050009,NCT00652119,NCT01402271,NCT00993655,NCT02020707,NCT00002717,NCT02272790,NCT00084448,NCT01649336,NCT00491855,NCT00008138,NCT00003385,NCT00002734,NCT00003160,NCT01447706,NCT00373217,NCT00331422,NCT00028743,NCT00004934,NCT00002894,NCT00003624,NCT00483782,NCT00003322,NCT01091428,NCT00550784,NCT00003896,NCT00003998,NCT00003064,NCT00003880,NCT00408070,NCT03126812,NCT00838656,NCT00023907,NCT00079430,NCT01083537,NCT00825201,NCT01821859,NCT00005046,NCT02834975,NCT00005026,NCT00075712,NCT00017303,NCT00003378,NCT00672295,NCT00003120,NCT00085358
|
Cervical cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C77 |
NCT01487226,NCT04341883,NCT03367871,NCT02039791,NCT01229930,NCT03534713,NCT04551950,NCT04806945,NCT05367206,NCT02432365,NCT01566240,NCT01405235,NCT00340184,NCT00003379,NCT00997009,NCT00980954,NCT05247619,NCT01594099,NCT02446652,NCT02036164,NCT04799639,NCT00003624,NCT04731038,NCT02422563,NCT04974944,NCT04150575,NCT04982237,NCT01756170,NCT01755845,NCT00462397,NCT00276796,NCT03912402,NCT03852979,NCT00138151,NCT04864782,NCT04868708,NCT04016142,NCT05179239,NCT00003377,NCT04973904,NCT03635567,NCT04884906,NCT00057928,NCT00823186,NCT02467907,NCT00626561,NCT00002949,NCT04238988,NCT03912415,NCT01667211,NCT00003065
|
Head and neck cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D42 |
MTOR
NCT00004865,NCT01911598,NCT02013453,NCT00004089,NCT01333085,NCT00319839,NCT01024101,NCT00002632,NCT00851877,NCT00392704,NCT00971867,NCT00270790,NCT00089297,NCT00959387,NCT03887442,NCT00003592,NCT04338399,NCT00566540,NCT00833261,NCT00003327,NCT00005647,NCT00176241,NCT00002888,NCT00997906,NCT01084083,NCT00003251,NCT00011999,NCT00003206,NCT00005087,NCT01412229,NCT00014118,NCT04883281,NCT00003193,NCT00113399,NCT00731380,NCT00337532,NCT01264328,NCT00004094,NCT00022217,NCT00006248,NCT00343083,NCT00301028,NCT00100789,NCT00083057,NCT00025298,NCT00002922,NCT01126216,NCT00004097,NCT00855764 |
Malignant neoplasms of stomach, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B72.Z |
NCT02163291,NCT00639522,NCT04714190,NCT00881816,NCT03081143
|
Testicular cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C80 |
NCT00072215,NCT00104676,NCT00004077,NCT00531687,NCT00231582
|
CholangiocarcinomaDisease Category: X.Extension CodesDisease ICD-11 Code: XH7M15 |
NCT03943043,NCT02181634,NCT04077983,NCT01963325,NCT04683939
|
Malignant neoplasms of corpus uteriDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C76 |
NCT01970722,NCT00505492,NCT00063999,NCT02684227,NCT02112552
|
ovarian cystadenocarcinomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C73.Y |
NCT00085358,NCT02050009,NCT00079430,NCT00002913,NCT00466960
|
Malignant lymphoma, not elsewhere classifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B33.5 |
NCT00088530,NCT01555853,NCT00038545,NCT03013218,NCT03169790
|
Malignant neoplasms of biliary tract, distal bile ductDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C15 |
TUBB6,PMP22,GMNN,ITGAV,LMNA
|
Superficial ovarian endometriosisDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GA10.B4 |
TUBB6,PMP22,TUBB3,LMNA,RAB9A
|
Glioblastoma of brainDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A00.00 |
TUBB6,GMNN,ITGAV,LMNA,RAB9A
|
Renal cell carcinomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C90 |
MTOR
NCT05092373,NCT02419495,NCT00001383 |
Pulmonary hypertensionDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BB01 |
MTOR
NCT01340131,NCT01246193,NCT01352689 |
Squamous cell carcinoma of oropharynxDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B6A.0 |
NCT03829722,NCT04572100,NCT05108870,NCT04718415
|
Hepatocellular carcinoma, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH4W48 |
NCT05092373,NCT03008512,NCT03563170,NCT04175912
|
Adenocarcinoma of prostateDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C82.0 |
SLCO1B1,SLCO1B3
NCT04221828,NCT00521781 |
Large cell carcinoma of bronchus or lungDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25.3 |
NCT00596830,NCT03345810,NCT02186847,NCT00955305
|
Serous cystadenoma,borderline malignancy of ovaryDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C73.4 |
THRB,GMNN,TUBB3,SLCO1B3
|
Germ cell tumour of testisDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C80.2 |
GMNN,THRB,RAB9A,SLCO1B1
|
Breast in situ carcinomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2E65 |
NCT04216472,NCT02603679,NCT03725436,NCT05177796,NCT02474173,NCT02632071,NCT05422794,NCT03554044,NCT01935492,NCT02672475,NCT02689427,NCT00520975,NCT01275677,NCT04266249,NCT03101748,NCT03179904,NCT03853707,NCT02593175,NCT01238133,NCT01463072,NCT04249167,NCT03201861,NCT00785291,NCT02436993,NCT01263145,NCT01091428,NCT02187991,NCT03606967,NCT05233696,NCT01750073,NCT00542451,NCT02883062,NCT04081389,NCT02654119,NCT05092373,NCT02419495,NCT04862585,NCT04001829
|
Endometrial cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C76 |
MTOR
NCT01367002,NCT00883116,NCT00006377,NCT02730416,NCT01770171,NCT05201547,NCT02630823,NCT04527900,NCT00003691,NCT00003127,NCT00006027,NCT05481645,NCT03603184,NCT00003624,NCT02744898,NCT05489848,NCT00002949,NCT00373620,NCT03932409,NCT03503786,NCT03189446,NCT03935256,NCT00515073,NCT04159155,NCT00016341,NCT04652076,NCT01244789,NCT00879359,NCT00022620,NCT00513786,NCT05092373,NCT00052312,NCT00334321,NCT02476955,NCT00411138 |
Mixed tumour, malignant, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH0V86 |
NCT01969578,NCT05000892,NCT04825938
|
LeukaemiaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A60-2B33 |
NCT00003294,NCT00003230,NCT00003402
|
Carcinosarcoma of uterusDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C76.43 |
GMNN
NCT00687687,NCT00954174 |
Malignant mixed epithelial mesenchymal tumour of ovaryDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B5D.0 |
NCT02822157,NCT01249443,NCT02107937
|
Hereditary breast and ovarian cancer syndromeDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C65 |
NCT02978495,NCT00535119,NCT05485766
|
Gastrointestinal stromal tumourDisease Category: X.Extension CodesDisease ICD-11 Code: XH9HQ1 |
NCT02574663,NCT03944304,NCT02607332
|
Malignant neoplasms of kidney, except renal pelvisDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C90 |
NCT00077129,NCT00024388,NCT03678883
|
Central nervous system diseaseDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8A04-8D87 |
NCT00002814,NCT00014573,NCT00031577
|
Malignant neoplasms of oesophagusDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B70 |
SLCO1B1,SLCO1B3
NCT02395705 |
Thymic carcinoma, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH6AK2 |
NCT03694002,NCT00818090,NCT00010257
|
Intrahepatic cholangiocarcinomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C12.10 |
NCT04175912,NCT02392637,NCT03579771
|
HepatoblastomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C12.01 |
PMP22,GMNN,TUBB3
|
Squamous cell carcinoma of bronchus or lungDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25.2 |
SLCO1B1,GMNN,SLCO1B3
|
Mesothelioma of pleuraDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C26.0 |
NPC1,GMNN,LMNA
|
Adenocarcinoma of bronchus or lungDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25.0 |
SLCO1B1,GMNN,SLCO1B3
|
MelanomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C30 |
NCT01200342,NCT02300935,NCT00329641,NCT01827111,NCT00111007,NCT00093119,NCT00864253,NCT00255762,NCT01174238,NCT02023710,NCT04979585,NCT00779714,NCT01006252,NCT01704287,NCT01039844,NCT01666418,NCT00483301,NCT00434252,NCT01676649,NCT00522834,NCT01009515,NCT03167177,NCT00796991,NCT00084214,NCT02617849,NCT01107665,NCT00288041,NCT00462423,NCT00970996
|
Malignant neoplasm metastasis in lymph nodes of head, face or neckDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D60.0 |
SLCO1B1,SLCO1B3
NCT05459129,NCT00736944,NCT05283226,NCT04282109,NCT01612351,NCT02573493,NCT03169764,NCT05272696,NCT04278092,NCT02270814,NCT03723967,NCT04882462,NCT04843098,NCT05054439,NCT00513383,NCT04831320,NCT04826679,NCT02124707,NCT01154920,NCT01732640,NCT04164238,NCT04428151,NCT05189184,NCT04807140,NCT03830385,NCT05294900,NCT01852292 |
Prostate cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C82 |
MTOR
NCT00024414,NCT00004054,NCT00003614,NCT00028769,NCT00038246,NCT01558492,NCT00933426,NCT04148885,NCT00038168,NCT00003394,NCT00193193,NCT02911350,NCT00009750,NCT00005028,NCT00030654,NCT00084565,NCT00005847,NCT00003084,NCT00005048,NCT00049257,NCT00016913,NCT00151060,NCT00003717,NCT00284752,NCT00003622 |
Malignant neoplasms of bladder, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C94.Z |
NCT00003930,NCT00003342,NCT00003105,NCT00022633,NCT04060459,NCT00006118,NCT00003701,NCT00268450,NCT00055601,NCT02302807,NCT00003133,NCT00585689,NCT05328336,NCT00022191,NCT00002917,NCT00583349,NCT00136175,NCT00005644,NCT00002949,NCT00933374,NCT04602117,NCT00310011,NCT00003376,NCT00028860,NCT00045630
|
Breast cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C60-2C6Y |
MTOR
NCT00146549,NCT00001384,NCT05227664,NCT04213898,NCT00533936,NCT00434356,NCT00546156,NCT00052351,NCT00050167,NCT00129922,NCT00320541,NCT00668616,NCT00251472,NCT00544232,NCT00096668,NCT00403130,NCT04129996,NCT00003086,NCT00900627,NCT00455533,NCT00616967,NCT00334802,NCT02413320,NCT00003042,NCT00025688,NCT00136539,NCT00009763,NCT00006256,NCT00003992,NCT00475670,NCT00003927,NCT00003035,NCT00407888,NCT00482391,NCT00054028,NCT00795899,NCT00618826,NCT04296175,NCT00542191,NCT00679029,NCT00068757,NCT00404404,NCT00629278,NCT00476827,NCT00394251,NCT00654836,NCT00622466,NCT00499083,NCT00532285,NCT00607438,NCT00254592,NCT00005635,NCT00041470,NCT00724386,NCT00394082,NCT04138719,NCT03197935,NCT00748553,NCT00004092,NCT00600340,NCT00070278,NCT00915018,NCT00723125,NCT00006459,NCT00629499,NCT00971945,NCT00479674,NCT00004013,NCT00194779,NCT00140140,NCT00574587,NCT00789581,NCT03057600,NCT00675259,NCT02315196,NCT00680758,NCT00567554,NCT00356681,NCT00256243,NCT00022230,NCT00448591,NCT00096343,NCT00733408,NCT00070564,NCT00915369,NCT00110084,NCT00004125,NCT00951665,NCT00944047,NCT00370552,NCT00003539,NCT00002628,NCT00930930,NCT00128856,NCT00580333,NCT00561119,NCT00191854,NCT00856492,NCT00038402,NCT00499525,NCT00031876,NCT00691912,NCT00004174,NCT00397761,NCT00426556,NCT00331630,NCT00803556,NCT00618657,NCT00960960,NCT00028990,NCT00511459,NCT05491694,NCT00028873,NCT00041119,NCT00919880,NCT01042379,NCT04085276,NCT00006108,NCT00431080,NCT00915603,NCT00093145,NCT00007904,NCT00148681,NCT00589238,NCT00445458,NCT00717340,NCT00107094,NCT00503750,NCT00093795,NCT00503906,NCT04958785,NCT00004067,NCT00807859,NCT00559845,NCT01881230,NCT04177108,NCT00691379,NCT00196872,NCT03002103,NCT00194792,NCT00002627,NCT00193206,NCT00887575,NCT00016406,NCT00001498,NCT00486668,NCT00154882,NCT00788931,NCT00433420,NCT00635050,NCT00014222,NCT00002837,NCT00039546,NCT00378313,NCT00003877,NCT00005581,NCT05192798,NCT03805399,NCT00768859,NCT00662129,NCT00146562,NCT00129389,NCT03872791,NCT00019812,NCT00316199,NCT04148911,NCT00002679,NCT00707707,NCT00591851,NCT00003440,NCT00820170,NCT00295893,NCT00846027,NCT00392392,NCT00003392,NCT00912639,NCT00336791,NCT00467012,NCT00442260,NCT00756470,NCT00236899,NCT00645177,NCT00002937,NCT00499603,NCT00532857,NCT04584112,NCT00934895,NCT00002772,NCT00001383,NCT00637897,NCT00274456,NCT00536939,NCT00311636,NCT00009997,NCT05390710,NCT00072319,NCT01009983,NCT01003158,NCT00289263,NCT00773695,NCT00301730,NCT00609791,NCT00876395,NCT00322400,NCT00479856,NCT00456846,NCT00983424,NCT00002662,NCT00667251,NCT00436566,NCT00777673,NCT00002953,NCT00006120,NCT00003972,NCT04770272,NCT00002826,NCT01818063,NCT00003013,NCT00003050,NCT01008150,NCT00003612,NCT03125902,NCT00003088,NCT00513292,NCT00005649,NCT00006110 |
Non-small cell lung cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25 |
NCT00093756,NCT02367794,NCT02252796,NCT00174356,NCT01225302,NCT02382406,NCT00112294,NCT00563784,NCT01980472,NCT00199758,NCT02083679,NCT00113516,NCT02041533,NCT02289456,NCT00268970,NCT01769391,NCT00294762,NCT00001499,NCT02419495,NCT01702844,NCT00986674,NCT00662597,NCT02276560,NCT00087802,NCT01822496,NCT00088556,NCT02366143,NCT02264990,NCT00686322,NCT00642759,NCT02259621,NCT00198432,NCT01454102,NCT00583830,NCT02453282,NCT02403895,NCT00481078,NCT00527735,NCT02405910,NCT02039674,NCT02319889,NCT00252798,NCT00049790,NCT01783197,NCT01993810,NCT01493843,NCT00603538,NCT00369070,NCT00034164,NCT00434226,NCT00666692,NCT02073968,NCT00602797,NCT00005065,NCT02182232,NCT02250326,NCT00198354,NCT02142738,NCT01757288,NCT00708812,NCT00654758,NCT00753909,NCT00280787,NCT00085839,NCT00073723,NCT00001450,NCT02420314,NCT00254891,NCT00006929,NCT00191256,NCT00318136,NCT00300885,NCT00070629,NCT00735696,NCT01069328,NCT00050960,NCT00343291,NCT00970580,NCT00976677,NCT01249443,NCT01179269,NCT00273507,NCT00655850,NCT00614484,NCT01507428,NCT00449657,NCT00042302,NCT00178256,NCT00298415,NCT00850577,NCT00716534,NCT01763671,NCT02016209,NCT00540514,NCT00539331,NCT00434135,NCT00097227,NCT02477826,NCT00508625,NCT01620190,NCT01011075,NCT00313768,NCT02106546,NCT01158144,NCT01236716,NCT00806286,NCT01820325,NCT00578149,NCT02226757,NCT02392507,NCT01199055,NCT00861627,NCT00088088,NCT02172846,NCT01702714,NCT01100931,NCT00832494,NCT01872403,NCT01171170,NCT01165216,NCT01763645,NCT00680940,NCT01560104,NCT00034541,NCT00168883,NCT02364999,NCT00259675,NCT00874848,NCT01912625,NCT00874107,NCT02220894,NCT00558636,NCT00161278,NCT01966003,NCT00960297,NCT01836679,NCT00687817,NCT00702975,NCT00322452,NCT00094835,NCT00460317,NCT00933803,NCT00991796,NCT01303926,NCT00974584,NCT01820754,NCT01364012,NCT00662311,NCT01234038,NCT00198367,NCT00035152,NCT02117024,NCT00061646,NCT00291850,NCT00807612,NCT00832819,NCT00480831,NCT02204345,NCT00347412,NCT01090830,NCT01527864,NCT00918203,NCT00576225,NCT00828841,NCT00686959,NCT02412371,NCT01310244,NCT02322281,NCT00866528,NCT00800202,NCT01496742,NCT00762034,NCT00723957,NCT00269828,NCT00787852,NCT00473889,NCT00437749,NCT00147537,NCT00903942,NCT00915005,NCT00152477,NCT01160601,NCT02064491,NCT01519804,NCT00752115,NCT00071188,NCT00702572,NCT01196234,NCT00730925,NCT01711697,NCT01024062,NCT01383148,NCT01326767,NCT01268059,NCT01366131,NCT01413750,NCT00948675,NCT01649947,NCT00966914,NCT00768131
|
Pleural mesotheliomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C26 |
MTOR
NCT02798536 |
Pain, unspecifiedDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: MG3Z |
MTOR
NCT02311907 |
Solid tumour/cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A00-2F9Z |
MTOR,ITGAV
|
Liver cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C12 |
MTOR
NCT00732836 |
Chronic arterial occlusive disease, unspecifiedDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BD4Z |
NCT03064126,NCT00518284
|
Soft tissue disorderDisease Category: 15.Diseases of the musculoskeletal system or connective tissueDisease ICD-11 Code: FB56 |
NCT05189483,NCT03524898
|
Carcinoma, undifferentiated, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH1YY4 |
NCT00989651,NCT00108745
|
Hodgkin lymphomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B30 |
NCT01929941,NCT01555853
|
strictureDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BB80.Z-LB15.1 |
NCT01868984,NCT02040454
|
Polyneuropathy, unspecifiedDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8C0Z |
NCT00979082,NCT03492047
|
Skin cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C30-2C37 |
NCT02054884,NCT03167164
|
ThymomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C27 |
NCT00010257,NCT00818090
|
Penile cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C81 |
NCT04475016,NCT00512096
|
Other specified malignant neoplasms ofcolonDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B90.Y |
SLCO1B1,SLCO1B3
|
Hepatocellular carcinoma of liverDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C12.02 |
GMNN,GMNN
|
Other specified malignant neoplasms of bronchus or lungDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25.Y |
TUBB3,SLCO1B3
|
Urothelial carcinoma of bladderDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C94.2 |
SLCO1B1,SLCO1B3
|
Malignant neoplasms of adrenal glandDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D11 |
GMNN,TUBB3
|
Ovarian cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C73 |
NCT00391118,NCT00006454,NCT00005026,NCT00003413,NCT00652119,NCT01493505,NCT00002819,NCT00052468,NCT00063401,NCT00993655,NCT00003160,NCT00331422,NCT01802749,NCT00003896,NCT00226915,NCT01253681,NCT01650376,NCT00003449,NCT00517621,NCT04679064,NCT00084448,NCT00002734,NCT00401674,NCT01570582,NCT05145569,NCT05316376,NCT00408070,NCT03989336,NCT03737643,NCT00750386,NCT00838656,NCT00003644,NCT03806049,NCT00005046,NCT00023907,NCT01447706,NCT00520013,NCT01220154,NCT00660842,NCT00001383,NCT00550784,NCT00189553,NCT00657878,NCT01654146,NCT00003998,NCT00002568,NCT00772798,NCT02483104,NCT02012192,NCT00326456,NCT00976911,NCT01649336,NCT00490711,NCT00228319,NCT02718417,NCT03197584,NCT00390611,NCT00003732,NCT03287271,NCT01239732,NCT00886717,NCT00170664,NCT00003733,NCT01146795,NCT02766582,NCT01739218,NCT01357161,NCT00003385,NCT03818282,NCT01081951,NCT03394885,NCT03740165,NCT02312661,NCT00189371,NCT00373217,NCT00003378,NCT01083537,NCT00002928,NCT00158379,NCT00075712,NCT01684878,NCT01653912,NCT00889382,NCT00031954,NCT00937560,NCT04931342,NCT00002717,NCT00005051,NCT00028743,NCT00003880,NCT00189566,NCT00840450,NCT00091377,NCT03398655,NCT03410784,NCT01821859,NCT02470585,NCT00738699,NCT03699449,NCT03393884,NCT01485848,NCT03776812,NCT00314678,NCT00929162,NCT01402271,NCT00825201,NCT00002894,NCT01711970,NCT03025477,NCT03763123,NCT00003322,NCT00479817,NCT02520154,NCT00003624,NCT03901118,NCT00102622,NCT01644825,NCT00585052,NCT00003214,NCT00008138,NCT04374630,NCT01778803,NCT04000295,NCT02121990,NCT02159820,NCT00011986,NCT00702299,NCT04815408,NCT03942068,NCT00129727,NCT02476955,NCT05145218,NCT02004093,NCT01219777,NCT03117933,NCT00003064,NCT00672295,NCT04261465,NCT04729387,NCT04590625,NCT00017303,NCT00483782,NCT03393507,NCT04921527,NCT02092363,NCT01838538,NCT00964626,NCT01196741,NCT04072263,NCT02157870,NCT04938583,NCT01954355,NCT00610714,NCT00004921,NCT01332656,NCT00003386,NCT02440425,NCT03639246,NCT00193297,NCT03126812,NCT00034151,NCT01204749,NCT02834975,NCT04661696,NCT03539328,NCT00003944,NCT00945191,NCT03635489,NCT04729608,NCT03038100,NCT00466986,NCT03940196,NCT03246074,NCT02125513,NCT00231075,NCT00004934,NCT02001272,NCT03029611,NCT00003120,NCT02389985,NCT02383251,NCT05255471,NCT02948075,NCT04337632,NCT00005612,NCT04908787,NCT03056833,NCT01696032,NCT04608409,NCT05044871,NCT02545010,NCT03467178,NCT05371301
|
Small cell carcinoma of bronchus or lungDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25.1 |
NCT01441349,NCT00454324,NCT04314895,NCT02551432,NCT00617409,NCT03219762,NCT03810872,NCT02262897,NCT04213937,NCT00317200,NCT04210037,NCT04247126,NCT04672928,NCT00527735,NCT05420636,NCT02038647,NCT04698941,NCT02769832,NCT04056949
|
Carcinomas of ovaryDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C73.0 |
NCT03756818,NCT02272790,NCT03400306,NCT02107378,NCT05446870,NCT04602117,NCT03598270,NCT01847677,NCT02419495,NCT05158062,NCT01970722,NCT05092373,NCT01091428,NCT03030287,NCT03690739,NCT02437812,NCT00181701,NCT02244502
|
Adenocarcinoma of stomachDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B72.0 |
SLCO1B1,SLCO1B3
NCT03281369,NCT04209686,NCT04604132,NCT04077255,NCT02615730,NCT02449655,NCT04510064,NCT01336062,NCT04572542,NCT03990103,NCT02448329,NCT03193918,NCT01641783,NCT04190745,NCT02625623 |
Malignant neoplasms of nasopharynxDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B6B |
NCT03020329,NCT04766359,NCT03668730,NCT04850235,NCT03574324,NCT01534585,NCT03047265,NCT03604965,NCT01797900,NCT04004871,NCT04223024,NCT03306121,NCT01712919,NCT02250599,NCT01694576,NCT04282070,NCT04015661
|
Mature T-cell lymphomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A90 |
MTOR
NCT00003397,NCT00003402,NCT04856137,NCT03435250,NCT00003957,NCT00021372,NCT00005021,NCT00008021,NCT00003294,NCT00027937,NCT00004916,NCT00001383,NCT00009776,NCT00004057,NCT03884556 |
NeoplasmsDisease Category: 02.NeoplasmsDisease ICD-11 Code: 02 |
NCT05322720,NCT03834948,NCT00004979,NCT05344742,NCT04047862,NCT00003092,NCT00003242,NCT05214976,NCT00454649,NCT00003555,NCT03918278,NCT02283489,NCT03600090,NCT04180384,NCT00005819,NCT02979392,NCT03577704,NCT03588039,NCT00003742,NCT00001387,NCT04541108,NCT04594005,NCT02051751,NCT00003899,NCT05017012,NCT00287937,NCT04143711,NCT00496028,NCT00399126,NCT00087217,NCT04481009,NCT01297452,NCT02430311,NCT01362374,NCT05381909,NCT00003004,NCT01930292,NCT03154294,NCT00350025,NCT00009932,NCT02057380,NCT03421353,NCT04999969,NCT00042809,NCT02122770,NCT04176952,NCT00793897,NCT05262335,NCT03894540,NCT02597036,NCT04675528,NCT02379416,NCT03503604,NCT04742036,NCT04794699,NCT04761601,NCT01293630,NCT00046423,NCT00360360,NCT00054548,NCT01593228,NCT02630264,NCT03693612,NCT00768469,NCT00881101,NCT01967043,NCT00001442,NCT00080418,NCT00607048,NCT00520000,NCT00458315,NCT04802980,NCT02593708,NCT00556088,NCT00009828,NCT04778839,NCT03892018,NCT00307255,NCT04046016,NCT00486954,NCT02715531,NCT01240655,NCT04151277,NCT00088114,NCT03601897,NCT02711137,NCT03486314,NCT01285466,NCT02341456,NCT00811993,NCT01862328,NCT00809133,NCT00028587,NCT00193596,NCT00002939,NCT01992341,NCT01515306,NCT03907475,NCT00009802,NCT01994031,NCT00891605,NCT01494688,NCT01938846,NCT02317874,NCT00030368,NCT01617928,NCT01455532,NCT02134067,NCT00095914,NCT02607202,NCT00004173,NCT01566435,NCT01525602,NCT03981796,NCT03537690,NCT00848718,NCT00024310,NCT00100139,NCT00422682,NCT04449549,NCT02937272,NCT01411410,NCT00016874,NCT01868022,NCT02762981,NCT01248949,NCT01031212,NCT00005078,NCT01159418,NCT01419548,NCT03330106,NCT02723955,NCT00002587,NCT00954512,NCT00004863,NCT00006018,NCT00003130,NCT01281176,NCT01548924,NCT00006257,NCT02730481,NCT03595059,NCT03309150,NCT03928314,NCT03057366,NCT00511849,NCT04336098,NCT01491204,NCT00653939,NCT01946074,NCT03430882,NCT03085914,NCT02794571,NCT03564691,NCT01968915,NCT04035473
|
Malignant neoplasms of ovaryDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C73 |
NCT01121406,NCT00001426,NCT03314740,NCT02903004,NCT01616303,NCT00919984,NCT01608009,NCT00191646,NCT04561817,NCT00653952,NCT00069901,NCT04711161,NCT01279291,NCT03574779,NCT00001272
|
Neuroendocrine carcinomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C34 |
NCT00001569,NCT00193531,NCT00873119,NCT01593306,NCT00894569,NCT00936702,NCT01253681,NCT04705519,NCT03136055,NCT00731861,NCT02215447,NCT00003943,NCT00737243,NCT00003558
|
Peritoneal cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C51 |
NCT03056833,NCT02125513,NCT00191646,NCT00517621,NCT00063401,NCT00391118,NCT03030287,NCT00189371,NCT00001569,NCT00666991,NCT01279291,NCT03038100,NCT03740165,NCT00189566
|
Malignant neoplasm of pancreas, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C10.Z |
NCT02241551,NCT02106884,NCT00111904,NCT02135822,NCT01632306,NCT00003591,NCT01088815,NCT04814485,NCT00691054,NCT04707118,NCT02050178,NCT02155088,NCT01730222,NCT03885219,NCT01851174,NCT03585062,NCT05193604,NCT02506803,NCT04643405,NCT05497778,NCT00398086,NCT03649321,NCT01470417,NCT01893801,NCT00882973,NCT02047500,NCT05047991,NCT04723030,NCT05298020,NCT03693677,NCT03825328,NCT03721744,NCT01693276,NCT05168527,NCT05218889,NCT00844649,NCT02391662,NCT00323583,NCT03989310,NCT02109445,NCT02717091,NCT03316599,NCT03815461,NCT01836432,NCT03136406,NCT03563144,NCT01964534,NCT02340117,NCT01647828,NCT04858009,NCT04683315,NCT03344172,NCT02124317,NCT04902261,NCT01488552,NCT02109341,NCT02588443,NCT04054362,NCT02481635,NCT04674956,NCT02581501,NCT03600623,NCT02651727,NCT02514031,NCT03487016,NCT02767557,NCT02358161,NCT04581876,NCT02207465,NCT01461915,NCT02975141,NCT02283372,NCT03777462,NCT03797443,NCT04589234,NCT03086369,NCT03502343,NCT03278015,NCT02272738,NCT02399137,NCT01822756,NCT02043730,NCT03252808,NCT02873598,NCT02210559,NCT01010945,NCT04089150,NCT01839487,NCT01834235,NCT05185869,NCT03336216,NCT03417921,NCT03714555,NCT04247126,NCT03941093,NCT01621243,NCT01858883,NCT02559674,NCT04617067,NCT04592861,NCT03519308,NCT02138383,NCT02574663,NCT03329248,NCT04104672,NCT00024375,NCT03586869,NCT02930902,NCT03257033,NCT04390399,NCT02570711,NCT03850769,NCT03641183,NCT02005315,NCT02506842,NCT03387098,NCT01934634,NCT01978184,NCT03520790,NCT05251038,NCT04158635,NCT02705196,NCT02827201,NCT02608229,NCT03412799,NCT02382263,NCT05346146,NCT03435289,NCT03415802,NCT04672005,NCT04683939,NCT04498689,NCT05241249,NCT04447092,NCT04753879,NCT03636308,NCT04241276
|
Inflammatory carcinoma of breastDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C62 |
NCT03515798,NCT01730833,NCT02199418,NCT00513695,NCT01938833,NCT01583426,NCT02876302,NCT02879513,NCT00016276,NCT02682693,NCT05383196,NCT02125344,NCT02221999
|
Malignant neoplasm metastasis, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2E2Z |
NCT02419495,NCT01721746,NCT00048893,NCT02158520,NCT01933061,NCT00046423,NCT00046514,NCT00731861,NCT01014351,NCT00046527,NCT00781612,NCT03678883,NCT00984464
|
Carcinoma of male breastDisease Category: X.Extension CodesDisease ICD-11 Code: XH5KW8 |
NCT00470301,NCT00861705,NCT01493310,NCT00513695,NCT03018080,NCT00770809,NCT02060253,NCT01276496,NCT01938833,NCT00368875,NCT01281150,NCT01288261,NCT00119262
|
Malignant neoplasms of fallopian tube, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C74.Z |
NCT00191646,NCT00954174,NCT01167712,NCT00063401,NCT02419495,NCT00391118,NCT03030287,NCT02125513,NCT01279291,NCT01970722,NCT05092373,NCT02272790,NCT03776812
|
Lung cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25 |
MTOR
NCT00003943,NCT00005059,NCT00028938,NCT00003089,NCT00006378,NCT00002972,NCT00055887,NCT00004126,NCT00006469,NCT00005646,NCT00062179,NCT00025389,NCT00005849,NCT00003881,NCT00004253,NCT00003587,NCT00004265,NCT02525653,NCT00004887,NCT00006049,NCT00004093,NCT00004137,NCT00278148,NCT00453167,NCT02279732,NCT00054392,NCT00081302,NCT00005806,NCT00054210,NCT00533585,NCT00226590,NCT00006012,NCT03348904,NCT00245154,NCT00079287,NCT00003696,NCT00003284,NCT00006004,NCT00003111,NCT02328105,NCT00003589,NCT00003317,NCT00004160,NCT00004859,NCT00003281,NCT00005032,NCT00748163,NCT00004055,NCT00003158,NCT00193310,NCT00077220,NCT01862081,NCT00085501,NCT00004924,NCT00077246,NCT00729612,NCT00144053,NCT00011921,NCT00003235,NCT00033410,NCT00006484,NCT00062010,NCT00023673,NCT00003812,NCT00006229,NCT00020007,NCT00016315,NCT00096226,NCT00062062,NCT00533949,NCT02513563,NCT00979212,NCT00287989,NCT00004202,NCT00039039,NCT00004011,NCT01486602,NCT04647344,NCT00661193,NCT00801736,NCT00276588,NCT00033553,NCT00003117,NCT00280150,NCT00002519,NCT00003313,NCT00452803,NCT00334763,NCT01285609,NCT00459121,NCT00002969,NCT00003159,NCT00547443,NCT00795340,NCT00003299,NCT00193362,NCT02044601,NCT00551733,NCT00025480,NCT00043108,NCT01057342,NCT00290537,NCT00017407,NCT00553462,NCT00006374,NCT00063258,NCT00807573,NCT00032032,NCT00550537,NCT03322566,NCT00544648,NCT00495170,NCT01107626,NCT00828009,NCT00033696,NCT02733250,NCT03432598,NCT00616031,NCT00728845,NCT00003387,NCT01024712,NCT00465907,NCT00004159,NCT00526890 |
Melanoma of skin, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C30.Z |
NCT02419495,NCT00278122,NCT02020707,NCT00976573,NCT00110019,NCT00568451,NCT00081042,NCT02308553,NCT00404235,NCT00626405,NCT02158520
|
Malignant neoplasms of corpus uteri, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C76.Z |
SLCO1B3
NCT04634877,NCT01820858,NCT04865289,NCT05173987,NCT03517449,NCT04602117,NCT02725268,NCT04269200,NCT03884101 |
Malignant neoplasm of pancreasDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C10 |
NCT03807999,NCT05083247,NCT02301143,NCT04289792,NCT05222204,NCT04808687,NCT05493995,NCT02810418,NCT02017015,NCT02024009
|
Metastatic lymph node neoplasmDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D60 |
MTOR
|
Bladder cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C94 |
MTOR
|
Multiple sclerosisDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8A40 |
ITGAV
|
Inborn lipid metabolism errorDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5C52 |
THRB
|
Chronic myelomonocytic leukaemiaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A40 |
MTOR
|
HydrocephalusDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8D64 |
MTOR
|
Malignant mesenchymal neoplasmDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B5D-2B5Y |
MTOR
|
LymphangioleiomyomatosisDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CB07 |
MTOR
|
Arteries/arterioles disorderDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BD52 |
MTOR
|
Malignant haematopoietic neoplasmDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B33 |
MTOR
|
Pancreatic cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C10 |
MTOR
|
Fungal infectionDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1F29-1F2F |
RAB9A
|
Hyper-lipoproteinaemiaDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5C80 |
THRB
|
Transplant rejectionDisease Category: 22.Injury, poisoning or certain other consequences of external causesDisease ICD-11 Code: NE84 |
MTOR
|
Hypo-thyroidismDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A00 |
THRB
|
Primary neoplasms of brainDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A00 |
NCT00479765
|
Malignant neoplasms of oropharynxDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B6A |
NCT03416153
|
GliosarcomaDisease Category: X.Extension CodesDisease ICD-11 Code: XH9RC8 |
NCT04528680
|
Small cell carcinoma, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH0YB0 |
NCT02881125
|
Neoplasms of unknown behaviour of female genital organsDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2F96 |
NCT02182245
|
Chronic lymphocytic leukaemia or small lymphocytic lymphomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A82.0 |
NCT03003546
|
Other specified malignant neoplasms of colonDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B90.Y |
NCT00034190
|
PsoriasisDisease Category: 14.Diseases of the skinDisease ICD-11 Code: EA90 |
NCT00006276
|
Malignant neoplasms of vulvaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C70 |
NCT00014599
|
Other specified malignant neoplasms of oropharynxDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B6A.Y |
NCT01249443
|
Malignant neoplasm metastasis in bone or bone marrowDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2E03 |
NCT03678883
|
AnemiaDisease Category: 03.Diseases of the blood or blood-forming organsDisease ICD-11 Code: 3A00-3A9Z |
NCT00189371
|
Sex cord-gonadal stromal tumour, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH9G57 |
NCT01770301
|
Verrucous carcinoma, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH5PM0 |
NCT01249443
|
Human immunodeficiency virus disease without mention of associated disease or condition, clinical stage unspecifiedDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1C62.Z |
NCT01249443
|
Malignant neoplasm metastasis in female reproductive systemDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2E05 |
NCT02739529
|
Parathyroid carcinomaDisease Category: X.Extension CodesDisease ICD-11 Code: XH7JQ0 |
NCT03181100
|
IschemiaDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8B10-8B11 |
NCT02835586
|
Metastatic lung neoplasmDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D70 |
NCT03678883
|
Allergic/hypersensitivity disorderDisease Category: 04.Diseases of the immune systemDisease ICD-11 Code: 4A80-4A8Z |
NCT00277043
|
Adrenal cortical carcinomaDisease Category: X.Extension CodesDisease ICD-11 Code: XH4KH2 |
NCT00786110
|
Glioblastoma, primary, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH0MB1 |
NCT00479765
|
Diseases of coronary artery, unspecifiedDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA8Z |
NCT00124943
|
Other specified malignant neoplasms of the ovaryDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C73.Y |
NCT02429700
|
Human immunodeficiency virus type 1Disease Category: X.Extension CodesDisease ICD-11 Code: XN8LD |
NCT01435018
|
Mesotheliomas of peritoneumDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C51.2 |
NCT05449366
|
Hypopharyngeal cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B6D |
NCT01249443
|
Prostate hyperplasiaDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GA90 |
NCT03423979
|
Ductal carcinoma in situ of breastDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2E65.2 |
NCT02779855
|
Teratoma, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH83G5 |
NCT00104676
|
Malignant neoplasms of anus or anal canalDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C00 |
NCT01249443
|
Other specified carcinoma in situ of breastDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2E65.Y |
NCT01891357
|
Actinic keratosisDisease Category: X.Extension CodesDisease ICD-11 Code: XH36H6 |
NCT03083470
|
NeuropathyDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8C0Z |
NCT02311907
|
Glioma, malignantDisease Category: X.Extension CodesDisease ICD-11 Code: XH4RQ3 |
NCT03678883
|
Malignant neoplasms of vaginaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C71 |
NCT00002949
|
Cervical intraepithelial neoplasiaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2E66 |
NCT03143491
|
Malignant neoplasms of other or unspecified parts of mouthDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B66 |
NCT00933387
|
Rectum cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B92 |
NCT02574663
|
Type1diabetesmellitusDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A10 |
NCT01581476
|
Pleomorphic adenomaDisease Category: X.Extension CodesDisease ICD-11 Code: XH2KC1 |
NCT00502203
|
Diseases of the digestive system, unspecifiedDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DE2Z |
NCT00208936
|
Kaposi sarcomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B57 |
NCT00003350
|
Neuroendocrine tumour, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH8DS0 |
NCT04602117
|
Gallbladder/bile ducts/liver structural developmental anomalyDisease Category: 20.Developmental anomaliesDisease ICD-11 Code: LB20 |
NCT02392637
|
Malignant trophoblastic neoplasms of placentaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C75.0 |
NCT02639650
|
Rheumatoid arthritisDisease Category: 15.Diseases of the musculoskeletal system or connective tissueDisease ICD-11 Code: FA20 |
NCT00055133
|
Crohn diseaseDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DD70 |
SLCO1B3
|
Inflammatory bowel diseases, unspecifiedDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DD7Z |
SLCO1B3
|
Idiopathic pulmonary fibrosisDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CB03.4 |
SLCO1B3
|
Cytomegaloviral diseaseDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D82 |
THRB
|
Systemic lupus erythematosusDisease Category: 04.Diseases of the immune systemDisease ICD-11 Code: 4A40.0 |
TUBB3
|
Chikungunya virus diseaseDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D40 |
LMNA
|
Polycystic ovary syndromeDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A80.1 |
ITGAV
|
Malignant neoplasms of thymusDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C27 |
GMNN
|
Renal cell carcinoma, chromophobe typeDisease Category: X.Extension CodesDisease ICD-11 Code: XH6153 |
NPC1
|
Carpal tunnel syndromeDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8C10.0 |
TUBB6
|

