Collective Molecular Activities of the Plant: Garcinia Lucida
Country/Region:
CameroonTraditional Medicine System:
Gabon; Congo; Cameroon
Overview of Ingredients
22 All known Ingredients in Total
Unique ingredients have been isolated from this plant.Plant-Ingredients Associations were manually curated from publications or collected from other databases.
10 Ingredients with Acceptable Bioavailablity
Unique ingredients exhibit acceptable human oral bioavailablity, according to the criteria of SwissADME [PMID: 28256516] and HobPre [PMID: 34991690]. The criteria details:SwissADME: six descriptors are used by SwissADME to evaluate the oral bioavailability of a natural product:
☑ LIPO(Lipophility): -0.7 < XLOGP3 < +5.0
☑ SIZE: 150g/mol < MW < 500g/mol
☑ POLAR(Polarity): 20Ų < TPSA < 130Ų
☑ INSOLU(Insolubility): -6 < Log S (ESOL) < 0
☑ INSATU(Insaturation): 0.25 < Fraction Csp3 < 1
☑ FLEX(Flexibility): 0 < Num. rotatable bonds < 9
If 6 descriptors of a natural plant satisfy the above rules, it will be labeled high HOB.
HobPre: A natural plant ingredient with HobPre score >0.5 is labeled high human oral availability (HOB)
7 Ingredients with experimental-derived Activity
Unique ingredients have activity data available.Ingredient Structrual Cards
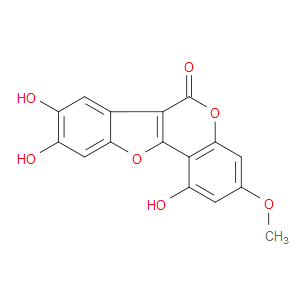
Ingredient ID: NPC86477
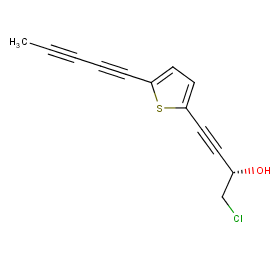
Ingredient ID: NPC67485
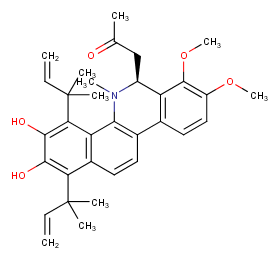
Ingredient ID: NPC471489
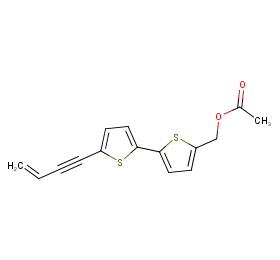
Ingredient ID: NPC302731
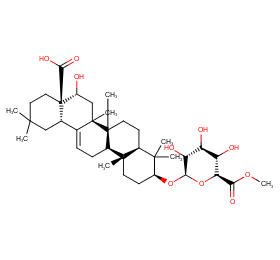
Ingredient ID: NPC281090
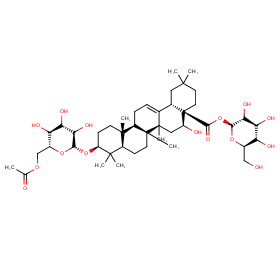
Ingredient ID: NPC248874
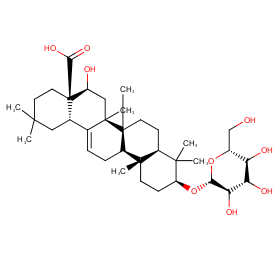
Ingredient ID: NPC238247
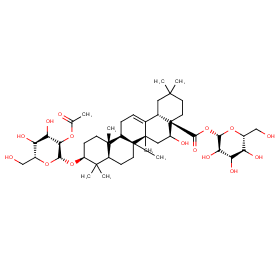
Ingredient ID: NPC23706

Ingredient ID: NPC235589

Ingredient ID: NPC223576
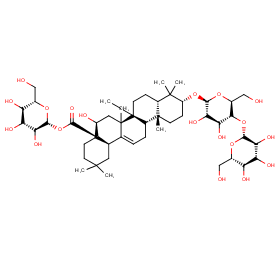
Ingredient ID: NPC222505

Ingredient ID: NPC213063
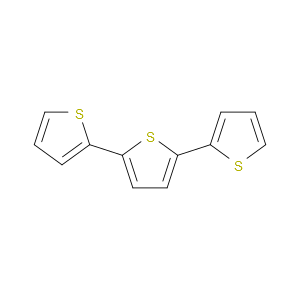
Ingredient ID: NPC195713
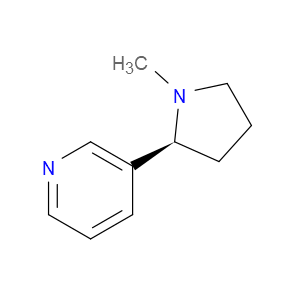
Ingredient ID: NPC182570
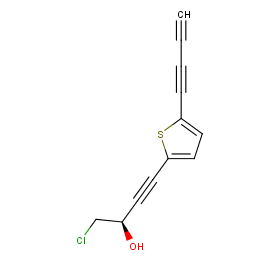
Ingredient ID: NPC182566
Ingredient ID: NPC179704

Ingredient ID: NPC17677

Ingredient ID: NPC173019
Ingredient ID: NPC171437
Ingredient ID: NPC149172

Ingredient ID: NPC143274
Ingredient ID: NPC121126
Classification of Human Proteins Collectively Targeted by the Plant
Detailed Information of Target Proteins
| Target Type | Protein Class | Gene ID | Protein Name | Uniprot ID | Target ChEMBL ID |
|---|---|---|---|---|---|
| Therapeutic Target | Nicotinic acetylcholine receptor | CHRNA3 | Neuronal acetylcholine receptor subunit alpha-3 | P32297 | CHEMBL3068 |
| Therapeutic Target | Phosphodiesterase | TDP1 | Tyrosyl-DNA phosphodiesterase 1 | Q9NUW8 | CHEMBL1075138 |
Clinical trials associated with plant from natural product (NP) & plant level:
| Clinical trials type | Number of clinical trials | |
|---|---|---|
| 252 | ||
| NCT ID | Title | Condition | Form in clinical use | Associated by plant or compound |
|---|---|---|---|---|
| NCT00469079 | Health Effects of SLT, Cigarette Smoking, and New Tobacco Products | nicotine dependence | Nicotine (NPC182570) | |
| NCT00622752 | Effects of EVT 302 With or Without NRT on Craving and Withdrawal in Healthy Male Smokers Deprived of Cigarettes | smoking cessation | Nicotine (NPC182570) | |
| NCT02720445 | Memory Improvement Through Nicotine Dosing (MIND) Study | Cognitive impairment | Nicotine (NPC182570) | |
| NCT01539434 | Behavioral Intervention to Maintain Physical Capacity and Activity in Patients With Chronic Obstructive Pulmonary Disease (COPD) | chronic obstructive pulmonary disease | Nicotine (NPC182570) | |
| NCT00851357 | Telephone Counseling and the Distribution of Nicotine Patches to Smokers | smoking cessation | Nicotine (NPC182570) | |
| NCT03000387 | Personalized Dosing of Nicotine Replacement (NRT to Effect) | nicotine dependence | Nicotine (NPC182570) | |
| NCT04608201 | Evaluation of the Efficacy of Nicotine Patches in SARS-CoV2 (COVID-19) Infection in Hospitalized Patients | COVID-19 | Nicotine (NPC182570) | |
| NCT01058876 | Comparison of Low Yield Cigarettes in African Americans vs. Whites | nicotine dependence | Nicotine (NPC182570) | |
| NCT03442413 | Nicotinic Hepatic Metabolism on Neuroreceptor Substrates of Nicotine Addiction | nicotine dependence | Nicotine (NPC182570) | |
| NCT00091468 | Nicotine Treatment of Mild Cognitive Impairment (MCI) | memory impairment | Nicotine (NPC182570) | |
| NCT01664741 | Nicotine Dependence, Withdrawal and Replacement Therapy Assessed by PET Imaging | nicotine dependence | Nicotine (NPC182570) | |
| NCT01208935 | Safety, Tolerability and Efficacy of the Nicotine Patch and Gum for the Treatment of Adolescent Tobacco Dependence | nicotine dependence | Nicotine (NPC182570) | |
| NCT05206435 | Methadone-Maintained Smokers Switching to E-Cigarettes | nicotine dependence | Nicotine (NPC182570) | |
| NCT02955329 | Vaping THC From Electronic Cigarettes | cannabis dependence | Nicotine (NPC182570) | |
| NCT03272685 | Impact of Very Low Nicotine Content Cigarettes in a Complex Marketplace | nicotine dependence | Nicotine (NPC182570) | |
| NCT02905734 | Nicotine Replacement Therapy in Nicotine-dependent Arrestees | nicotine dependence | Nicotine (NPC182570) | |
| NCT04124432 | Behavioral Pharmacology of Cannabis and Nicotine | cannabis dependence | Nicotine (NPC182570) | |
| NCT00108537 | Non-Nicotine Agents for Smoking Cessation | nicotine dependence | Nicotine (NPC182570) | |
| NCT00527020 | First Time in Human Study With GSK1018921 | schizophrenia | Nicotine (NPC182570) | |
| NCT03262233 | Stress Neuroadaptation in Tobacco Dependence | nicotine dependence | Nicotine (NPC182570) | |
| NCT02587312 | Evaluation of Very Low Nicotine Content Cigarettes in Adolescent Smokers | nicotine dependence | Nicotine (NPC182570) | |
| NCT01091363 | Tobacco Dependence Treatment for Asian Americans | nicotine dependence | Nicotine (NPC182570) | |
| NCT00392379 | Nicotine Lozenges for Treatment of Smokeless Tobacco Addiction | nicotine dependence | Nicotine (NPC182570) | |
| NCT01065506 | Smoking Termination Enhancement Project (STEP) | nicotine dependence | Nicotine (NPC182570) | |
| NCT01406223 | Mechanistic Evaluations of Pre-Cessation Therapies for Smoking Cessation | nicotine dependence | Nicotine (NPC182570) | |
| NCT02371850 | Transdermal Patch CVD 1000: The Effect of Heat on Nicotine Release From Nicotine Patches in Adult Smokers | nicotine dependence | Nicotine (NPC182570) | |
| NCT03809897 | Varenicline for Smoking Cessation in Hospitalized Patients With Psychiatric Disorders | nicotine dependence | Nicotine (NPC182570) | |
| NCT03634839 | Effects of E-cigarette Flavors on Youth TCORS 2.0 | nicotine dependence | Nicotine (NPC182570) | |
| NCT04590404 | Metabolism Informed Smoking Treatment: The MIST RCT | nicotine dependence | Nicotine (NPC182570) | |
| NCT01048944 | Nicotine Replacement Therapy (NRT) and Bupropion Mechanisms of Effectiveness in Smokers | nicotine dependence | Nicotine (NPC182570) | |
| NCT01075659 | Early Effects of a New Oral Nicotine Replacement Product and NiQuitin™ Lozenge | smoking cessation | Nicotine (NPC182570) | |
| NCT02179034 | Flavors and E-cigarette Effects in Adolescent Smokers- STUDY 1 | nicotine dependence | Nicotine (NPC182570) | |
| NCT04535362 | Nicotine's Potential Abuse With Menthol | nicotine dependence | Nicotine (NPC182570) | |
| NCT01838460 | Sublingual Nicotine Tablets Compared With Swedish Snus | smoking cessation | Nicotine (NPC182570) | |
| NCT02721082 | Changing the Default for Tobacco Treatment | nicotine dependence | Nicotine (NPC182570) | |
| NCT03404596 | Applying mHealth to Tobacco-related Health Disparities | smoking cessation | Nicotine (NPC182570) | |
| NCT01443858 | Meclizine as a Potential Smoking Cessation Treatment | smoking cessation | Nicotine (NPC182570) | |
| NCT00985985 | Efficacy and Safety Study of Nicotine Mint Lozenge (2mg and 4mg) in Smoking Cessation | nicotine dependence | Nicotine (NPC182570) | |
| NCT00064844 | Combination Nicotine Replacement for Alcoholic Smokers | nicotine dependence | Nicotine (NPC182570) | |
| NCT05159934 | Nicotine Differences in Smokers | nicotine dependence | Nicotine (NPC182570) | |
| NCT04946825 | Quit Smoking Study for People Who Use E-Cigarettes | nicotine dependence | Nicotine (NPC182570) | |
| NCT00586482 | Pilot Study: Preoperative Nicotine Lozenges | nicotine dependence | Nicotine (NPC182570) | |
| NCT02245308 | Abstinence Reinforcement Therapy (ART) for Homeless Veteran Smokers | nicotine dependence | Nicotine (NPC182570) | |
| NCT00105482 | Targeted Interventions for Weight-Concerned Smokers | nicotine dependence | Nicotine (NPC182570) | |
| NCT03856515 | Gender Differences in Switching From Smoking Regular Cigarettes to E-Cigarettes | carcinoma | Nicotine (NPC182570) | |
| NCT00939029 | Highdose Nicotine Patch Therapy for Smokeless Tobacco Use | nicotine dependence | Nicotine (NPC182570) | |
| NCT03521141 | PRecision Interventions for SMoking in the SCCS | nicotine dependence | Nicotine (NPC182570) | |
| NCT01314001 | Pharmacogenetics of Nicotine Addiction Treatment | nicotine dependence | Nicotine (NPC182570) | |
| NCT04606797 | Targeting Mailed Nicotine Patch Distribution Interventions to Rural Regions of Canada | nicotine dependence | Nicotine (NPC182570) | |
| NCT01867411 | Multimodal Neuroimaging Genetic Biomarkers of Nicotine AddictionSeverity | nicotine dependence | Nicotine (NPC182570) | |
| NCT03176784 | UW Quitting Using Intensive Treatment Study (QUITS) | nicotine dependence | Nicotine (NPC182570) | |
| NCT03865121 | Pilot Trial of Transnasal Nicotine in Parkinson Disease | Parkinson disease | Nicotine (NPC182570) | |
| NCT01536704 | Nicotine Lozenge Bioequivalence Study | smoking cessation | Nicotine (NPC182570) | |
| NCT01702519 | A Pharmacokinetic Evaluation of Two Nicotine Patches in Smokers | nicotine dependence | Nicotine (NPC182570) | |
| NCT00967174 | Cumulative Skin Irritation Potential of a New 25 mg Nicotine Patch | smoking cessation | Nicotine (NPC182570) | |
| NCT02250534 | Project 1, Study 2: Extended Exposure to Low Nicotine Content Cigarettes in Childbearing Age Women | nicotine dependence | Nicotine (NPC182570) | |
| NCT01980550 | Association of Functional COMT Val108/Met Polymorphism With Smoking Cessation in Nicotine Replacement Therapy | nicotine dependence | Nicotine (NPC182570) | |
| NCT01047527 | An Effectiveness Trial of Maintenance Therapy for Nicotine Dependence | nicotine dependence | Nicotine (NPC182570) | |
| NCT00755716 | Topiramate Alone and in Combination With the Nicotine Patch for Smoking Cessation: A Pilot Study | nicotine dependence | Nicotine (NPC182570) | |
| NCT03243630 | Effects of Flavors on Nicotine Reinforcement in Smokers | nicotine dependence | Nicotine (NPC182570) | |
| NCT01287377 | Smoking Cessation With the Nicotine Patch: A Pilot Study of Patch Messaging | smoking cessation | Nicotine (NPC182570) | |
| NCT02271919 | Varenicline and Combined NRT for Smoking Cessation | nicotine dependence | Nicotine (NPC182570) | |
| NCT02927847 | Neuroimaging Reward, Behavioral Treatment, and Smoking Cessation | nicotine dependence | Nicotine (NPC182570) | |
| NCT00132236 | Nurse Smoking Cessation of Patients With Chronic Obstructive Pulmonary Disease (COPD) With Nicotine Replacement Therapy (NRT) and Behavioral Support | chronic obstructive pulmonary disease;nicotine dependence | Nicotine (NPC182570) | |
| NCT02942212 | Health Literacy and Smoking Cessation in Low-SES Diverse Smokers - Project HALT II | smoking cessation | Nicotine (NPC182570) | |
| NCT05233228 | Mobile Health Technology for Personalized Tobacco Cessation Support in Laos | nicotine dependence | Nicotine (NPC182570) | |
| NCT03635333 | Effects of E-Cigarette Flavors on Adults TCORS 2.0 | nicotine dependence | Nicotine (NPC182570) | |
| NCT01438944 | Evaluation of Transdermal Nicotine Replacement Therapy (NRT) Activity Through Metabolic Induction | nicotine dependence | Nicotine (NPC182570) | |
| NCT05060965 | Inpatient Smoking Cessation Pilot Program | nicotine dependence | Nicotine (NPC182570) | |
| NCT01560754 | Disease-modifying Potential of Transdermal NICotine in Early Parkinson's Disease | Parkinson disease | Nicotine (NPC182570) | |
| NCT00888459 | A Pilot Study Evaluating Nicotine Lozenges and Self Help | nicotine dependence | Nicotine (NPC182570) | |
| NCT01502306 | A Randomized Trial of Medi-Cal Beneficiaries Calling the California Smokers' Helpline | smoking cessation | Nicotine (NPC182570) | |
| NCT00738595 | Phase II Study to Evaluate the Effectiveness of EVT 302 in Smoking Cessation. | smoking cessation | Nicotine (NPC182570) | |
| NCT04033237 | Project 4, Study 2: Extended Exposure to Low Nicotine Content Cigarettes in Pregnant Women | nicotine dependence | Nicotine (NPC182570) | |
| NCT00543881 | Telemedical Interventional Monitoring in Heart Failure | heart failure | Nicotine (NPC182570) | |
| NCT01847443 | Comparison of Pharmacokinetic Profiles of Two Nicotine Gum Formulations | smoking cessation | Nicotine (NPC182570) | |
| NCT00833404 | Smoking Cessation and Sexual Health in Men | smoking cessation | Nicotine (NPC182570) | |
| NCT04969198 | UW Withdraw From Tobacco Study | nicotine dependence | Nicotine (NPC182570) | |
| NCT04126135 | Comparative Effectiveness of Cystine Versus Nicotine Replacement Therapy | nicotine dependence | Nicotine (NPC182570) | |
| NCT04521647 | Effects of Menthol in E-cigarettes on Smoking Behaviors | nicotine dependence | Nicotine (NPC182570) | |
| NCT00332644 | Smoking Cessation Medications: Efficacy, Mechanisms and Algorithms | nicotine dependence | Nicotine (NPC182570) | |
| NCT00701207 | Study of Nicotine Patches in Patients With Sarcoidosis | pulmonary sarcoidosis | Nicotine (NPC182570) | |
| NCT01806779 | Combination Bupropion / Varenicline for Smoking Cessation in Male Smokers | nicotine dependence | Nicotine (NPC182570) | |
| NCT02964182 | Very Low Nicotine Content Cigarettes and E-Cigarettes in Promoting Smoking Cessation in Daily and Intermittent Cigarette Smokers | nicotine dependence | Nicotine (NPC182570) | |
| NCT01506908 | Provoked Craving Assessment | nicotine dependence | Nicotine (NPC182570) | |
| NCT00018148 | Combined Nortriptyline and Transdermal Nicotine for Smoking Cessation | nicotine dependence | Nicotine (NPC182570) | |
| NCT01085032 | Nicotine Patch Trial in Syrian Primary Care Settings | smoking cessation | Nicotine (NPC182570) | |
| NCT02081144 | Texting to Promote Tobacco Abstinence in Emergency Department Smokers: A Pilot Study | nicotine dependence | Nicotine (NPC182570) | |
| NCT04433767 | Depressed Mood Improvement Through Nicotine Dosing 2 | depressive disorder | Nicotine (NPC182570) | |
| NCT00734617 | Concurrent Nicotine Patch / Denicotinized Cigarette Therapy for Smoking Cessation II | nicotine dependence | Nicotine (NPC182570) | |
| NCT01417429 | Galantamine Effects on Nicotine Responses in Smokers | nicotine dependence | Nicotine (NPC182570) | |
| NCT00957918 | Study of NP002 in Subjects With Idiopathic Parkinson's Disease to Treat Dyskinesias Due to Levodopa Therapy | Parkinson disease | Nicotine (NPC182570) | |
| NCT01476202 | Provoked Craving Relief Study by NRT | smoking cessation | Nicotine (NPC182570) | |
| NCT01303861 | Concurrent Bupropion / Varenicline for Smoking Cessation | nicotine dependence | Nicotine (NPC182570) | |
| NCT01372254 | Behavioral Intervention for Low Income Depressed Smokers in Drug Treatment | nicotine dependence | Nicotine (NPC182570) | |
| NCT00616746 | Interactive Psychopharmacologic Effects of Alcohol and Nicotine in Humans | alcohol drinking | Nicotine (NPC182570) | |
| NCT00931021 | Smoking Cessation Treatment for Head and Neck Cancer Patients | smoking cessation | Nicotine (NPC182570) | |
| NCT03421210 | Effects of Smoking Environments on Brain Reactivity | nicotine dependence | Nicotine (NPC182570) | |
| NCT01907594 | Effects of Nicotine Replacement Therapy and D-cycloserine on Nicotine Treatment Seekers | nicotine dependence | Nicotine (NPC182570) | |
| NCT01478230 | An Exploratory Study Comparing Two Nicotine Inhalers | nicotine dependence | Nicotine (NPC182570) | |
| NCT00297492 | Gradual vs. Abrupt Cessation Treatment for Smoking | smoking cessation | Nicotine (NPC182570) | |
| NCT02018263 | Validation of a Remote Wireless Sensor Network (WSN) Approach to the Individualized Detection of Cocaine Use in Humans | cocaine dependence | Nicotine (NPC182570) | |
| NCT00744913 | Study of Nicotine Replacement Therapy in Pregnancy | smoking cessation | Nicotine (NPC182570) | |
| NCT05351606 | Integrating Tobacco Use Cessation Into HIV Care and Treatment in Kisumu County, Kenya | nicotine dependence | Nicotine (NPC182570) | |
| NCT01444131 | Varenicline in Combination With Nicotine Replacement Therapy for Smoking Cessation | nicotine dependence | Nicotine (NPC182570) | |
| NCT00307203 | Safety and Effectiveness of Sustained Release Bupropion in Treating Individuals With Schizophrenia Who Smoke | nicotine dependence | Nicotine (NPC182570) | |
| NCT00553709 | Study of Nicotine for the Prevention of PONV | post operative nausea and vomiting | Nicotine (NPC182570) | |
| NCT00967070 | Skin Sensitization Potential of a New Nicotine Patch | smoking cessation | Nicotine (NPC182570) | |
| NCT02147132 | Pilot Study of Nicotine Nasal Spray and Varenicline on Smoking in Methadone-Maintained Patients | nicotine dependence | Nicotine (NPC182570) | |
| NCT03808818 | Implementing a Virtual Tobacco Treatment for Cancer Patients in Community Oncology Practices | in situ carcinoma | Nicotine (NPC182570) | |
| NCT01592695 | Tailored Tobacco Quitline for Rural Veterans | nicotine dependence | Nicotine (NPC182570) | |
| NCT03178422 | Safety and Efficacy Study of the NRT CQSS2 System for Smoking Cessation in Treatment-Seeking Subjects | smoking cessation | Nicotine (NPC182570) | |
| NCT03492463 | The Role of Nicotine Dose and Route of Delivery in Affecting Adoption of E-cigarettes and Reducing Exposure to Toxic Combustion Products | nicotine dependence | Nicotine (NPC182570) | |
| NCT01926626 | Evaluation of Moclobemide, a Reversible MAO-A Inhibitor, as an Adjunct to Nicotine Replacement Therapy in Female Smokers | nicotine dependence | Nicotine (NPC182570) | |
| NCT00969137 | Sensitivity to Intravenous Nicotine: Genetic Moderators | nicotine dependence | Nicotine (NPC182570) | |
| NCT01989507 | Very Low-Nicotine Cigarettes in Smokers With SUD | nicotine dependence | Nicotine (NPC182570) | |
| NCT00086411 | Comparing Smoking Treatment Programs for Lighter Smokers - 1 | nicotine dependence | Nicotine (NPC182570) | |
| NCT03163303 | Smoking Tobacco and Drinking Study | nicotine dependence | Nicotine (NPC182570) | |
| NCT02487953 | Electronic Nicotine Delivery Systems as a Smoking Cessation Treatment | nicotine dependence | Nicotine (NPC182570) | |
| NCT01621022 | Bupropion Alone or Combined With Nicotine Gum | nicotine dependence | Nicotine (NPC182570) | |
| NCT03408574 | The Nicotinic Cholinergic System and Cognitive Aging | aging | Nicotine (NPC182570) | |
| NCT03432273 | Single-Dose Bioequivalence Study Comparing Two 2 mg Nicotine Lozenges | smoking cessation | Nicotine (NPC182570) | |
| NCT01560507 | Tailored Smoking Cessation Treatment for LIVE FOR LIFE® Participants | nicotine dependence | Nicotine (NPC182570) | |
| NCT03538938 | Improving Quitline Support Study | nicotine dependence | Nicotine (NPC182570) | |
| NCT02893345 | Safe@Home: A Self-Management Program for Individuals With TBI and Their Families | injury | Nicotine (NPC182570) | |
| NCT04262817 | The Interaction of Flavor With Nicotine Form in Adult Smokers | nicotine dependence | Nicotine (NPC182570) | |
| NCT01669122 | Pharmacokinetic Study of 4 mg Nicotine Lozenge. | smoking cessation | Nicotine (NPC182570) | |
| NCT03432312 | Single-Dose Bioequivalence Study Comparing Two 4 mg Nicotine Lozenges | smoking cessation | Nicotine (NPC182570) | |
| NCT03580525 | Nicotine Delivery Rate and Its Abuse Potential: Impact of Menthol | nicotine dependence | Nicotine (NPC182570) | |
| NCT01018394 | Nicotine Lozenge or Tobacco-Free Snuff for Smokeless Tobacco Reduction | nicotine dependence | Nicotine (NPC182570) | |
| NCT01034020 | Effects of Nicotine on Elements of Attentions in Smokers and Nonsmokers | nicotine dependence | Nicotine (NPC182570) | |
| NCT03069482 | Feasibility Trial of a Tailored Smoking Cessation App for People With Serious Mental Illness | nicotine dependence | Nicotine (NPC182570) | |
| NCT03168191 | Flavors and E-cigarette Effects in Adolescent Smokers- STUDY 2 | nicotine dependence | Nicotine (NPC182570) | |
| NCT00326781 | Bio-Behavioral Predictors of the Efficacy of Nicotine Replacement Therapy (NRT) | nicotine dependence | Nicotine (NPC182570) | |
| NCT05487807 | Adapting and Evaluating a Tobacco Use Cessation Program for People Living With HIV in Uganda and Zambia | nicotine dependence | Nicotine (NPC182570) | |
| NCT01892813 | Dissemination of a Tailored Tobacco Quitline for Rural Veteran Smokers | nicotine dependence | Nicotine (NPC182570) | |
| NCT02162849 | Reward Sensitivity and Pharmacotherapy for Smoking Cessation | nicotine dependence | Nicotine (NPC182570) | |
| NCT01932996 | Enhancing Smoking Cessation in the Homeless Population | nicotine dependence | Nicotine (NPC182570) | |
| NCT00258479 | Safety of Combining Modafinil and Nicotine Replacement Therapy in Treating Nicotine Dependent Adolescents | nicotine dependence | Nicotine (NPC182570) | |
| NCT03059940 | Smoking Cessation Intervention During Low Dose CT (LDCT) Screening for Lung Cancer | smoking cessation | Nicotine (NPC182570) | |
| NCT00657020 | Functional Magnetic Resonance Imaging (fMRI) Investigation of Nicotine Withdrawal Symptoms | nicotine dependence | Nicotine (NPC182570) | |
| NCT00879918 | Pharmacogenetics of Nicotine Metabolism in African-Americans | nicotine dependence | Nicotine (NPC182570) | |
| NCT02439944 | Efficacy and Neural Correlates of Personalized Treatment With Transdermal Nicotine Replacement | nicotine dependence | Nicotine (NPC182570) | |
| NCT04999644 | Reduced Nicotine Cigarette Purchasing Decisions | nicotine dependence | Nicotine (NPC182570) | |
| NCT04709471 | E-cigarette Nicotine Study | nicotine dependence | Nicotine (NPC182570) | |
| NCT00218283 | Nicotine Lozenge to Reduce Smokeless Tobacco Use | nicotine dependence | Nicotine (NPC182570) | |
| NCT02997657 | Positive Psychotherapy for Smoking Cessation Enhanced With Text Messaging: A Randomized Controlled Trial | smoking cessation | Nicotine (NPC182570) | |
| NCT00000447 | Behavioral/Drug Therapy for Alcohol-Nicotine Dependence (Naltrexone/Nicotine Patch) | nicotine dependence | Nicotine (NPC182570) | |
| NCT00046813 | Nicotine Patch for Nicotine Dependence in Individuals With Schizophrenia or Schizoaffective Disorder - 1 | nicotine dependence | Nicotine (NPC182570) | |
| NCT01351766 | Behavioral Activation Intervention, Reward Processing, and Youth Smoking Cessation | nicotine dependence | Nicotine (NPC182570) | |
| NCT00777569 | Comparisons of Nicotine-free Cigarettes, Extra Low Nicotine Cigarettes vs. Medicinal Nicotine | nicotine dependence | Nicotine (NPC182570) | |
| NCT04598594 | Evaluation of the Efficacy of Nicotine Patches in SARS-CoV2 (COVID-19) Infection in Intensive Care Unit Patients | COVID-19;severe acute respiratory syndrome | Nicotine (NPC182570) | |
| NCT05018117 | Effects of Nicotine and Attention on Frequency Tuning in Auditory Cortex | aging | Nicotine (NPC182570) | |
| NCT04063267 | Electronic Cigarettes as a Harm Reduction Strategy in Individuals With Substance Use Disorder | nicotine dependence | Nicotine (NPC182570) | |
| NCT00960778 | Gender Differences in Response to Nicotine Replacement Therapy and De-Nicotinized Cigarettes | nicotine dependence | Nicotine (NPC182570) | |
| NCT01457469 | Enhanced Quitline Intervention in Smoking Cessation for Patients With Non-Metastatic Lung Cancer | nicotine dependence | Nicotine (NPC182570) | |
| NCT04020718 | Engagement With an Adaptive Mobile Health Smoking Cessation Intervention | nicotine dependence | Nicotine (NPC182570) | |
| NCT00894166 | Evaluation of a Tailored Smoking Cessation Treatment Algorithm Based on Initial Treatment Response and Genotype | smoking cessation | Nicotine (NPC182570) | |
| NCT03543137 | To Assess the Bioequivalence of the 4mg Prototype Mini Nicotine Lozenge to the Reference Product (Nicorette) in Healthy Smokers | nicotine dependence | Nicotine (NPC182570) | |
| NCT00296647 | Smoking Cessation Intervention: Effectiveness in Primary Care | nicotine dependence | Nicotine (NPC182570) | |
| NCT00218296 | Comparison Across Treatment Conditions to Reduce Smokeless Tobacco Use | nicotine dependence | Nicotine (NPC182570) | |
| NCT05301660 | Effect of Nicotine Transdermal Patch on Cognitive Function and Glycolipid Metabolism in Non-smoking Schizophrenia | schizophrenia | Nicotine (NPC182570) | |
| NCT00032084 | S0002 - A Program to Quit Smoking With or Without Bupropion in Treating Patients With Stage I or II Non-Small Cell Lung Cancer Who Have Undergone Surgery | lung cancer | Nicotine (NPC182570) | |
| NCT02501265 | Adaptive Pharmacotherapy for Smoking Cessation | nicotine dependence | Nicotine (NPC182570) | |
| NCT02511704 | Pharmacokinetics and Acute Effects of Multiple Dose of Nicotine: Electronic Cigarette and Cigarette | nicotine dependence | Nicotine (NPC182570) | |
| NCT00775944 | Trial Comparing Different Methods of Support With Stopping Smoking (PORTSSS/Stop Together Trial) | nicotine dependence | Nicotine (NPC182570) | |
| NCT03972137 | Heart Rate Variability Biofeedback for Smoking Cessation | nicotine dependence | Nicotine (NPC182570) | |
| NCT02355665 | Study to Determine the Efficacy and Safety of a Novel Nicotine Replacement Therapy | nicotine dependence | Nicotine (NPC182570) | |
| NCT04488744 | Smoking by IV Pulsed Nicotine | smoking cessation | Nicotine (NPC182570) | |
| NCT00000437 | Tobacco Dependence in Alcoholism Treatment (Nicotine Patch/Naltrexone) | nicotine dependence | Nicotine (NPC182570) | |
| NCT03541044 | A Bioequivalence Study of Two Nicotine Lozenges in Fasting Conditions in Healthy Smokers | nicotine dependence | Nicotine (NPC182570) | |
| NCT01104896 | Effect of Daily Nicotine Patch Application on Mechanical Ventilation Weaning in Smoking Patients | nicotine dependence | Nicotine (NPC182570) | |
| NCT01050569 | Innovative Interventions for Smoking Cessation | nicotine dependence | Nicotine (NPC182570) | |
| NCT04490057 | A Smart Approach to Treating Tobacco Use Disorder in Persons Living With HIV | nicotine dependence | Nicotine (NPC182570) | |
| NCT02482233 | The END Perioperative Smoking Pilot Study | nicotine dependence | Nicotine (NPC182570) | |
| NCT03612960 | Measuring Neuroadaptations in Response to Very Low Nicotine Content Cigarettes | nicotine dependence | Nicotine (NPC182570) | |
| NCT03860077 | Impact of Nicotine Reduction on Adolescent Cigarette Use, Alternative Tobacco Use, and Harm From Tobacco | nicotine dependence | Nicotine (NPC182570) | |
| NCT00067158 | Smoking Cessation Treatment With Transdermal Nicotine Replacement Therapy - 1 | nicotine dependence | Nicotine (NPC182570) | |
| NCT01783912 | Helping Those With Mental Illness Quit Smoking | nicotine dependence | Nicotine (NPC182570) | |
| NCT02906644 | Combination Nicotine Patch / Lorcaserin for Smoking Cessation | nicotine dependence | Nicotine (NPC182570) | |
| NCT04740008 | Impact of Low Nicotine Cigarette Messaging on Perceptions and Cigarette Choices | nicotine dependence | Nicotine (NPC182570) | |
| NCT00061074 | Tobacco Cessation in Postmenopausal Women (Part II) - 2 | nicotine dependence | Nicotine (NPC182570) | |
| NCT01654107 | Persistence-Targeted Smoking Cessation | nicotine dependence | Nicotine (NPC182570) | |
| NCT00664755 | Menstrual Cycle Effects on Smoking Cessation and Cue Reactivity | nicotine dependence | Nicotine (NPC182570) | |
| NCT01136642 | Assessing Top Down and Bottom Up Attention Mechanisms in Smokers Using Nicotine Nasal Spray | nicotine dependence | Nicotine (NPC182570) | |
| NCT01296698 | Study to Determine if a New Nicotine Replacement Therapy Can Safely Help Smokers to Quit Smoking | nicotine dependence | Nicotine (NPC182570) | |
| NCT00965913 | Cumulative Skin Irritation Potential of a New 15 mg Nicotine Patch | smoking cessation | Nicotine (NPC182570) | |
| NCT00699556 | Do Treatments for Smoking Cessation Affect Alcohol Drinking? Study 1: Nicotine Replacement Therapy | alcohol drinking | Nicotine (NPC182570) | |
| NCT02265874 | Nicotine Treatment for Pulmonary Sarcoidosis: A Clinical Trial Pilot Study | sarcoidosis | Nicotine (NPC182570) | |
| NCT05335915 | Acute Pharmacokinetic and Pharmacodynamic Effects of "Tobacco-Free" Oral Nicotine Pouches in Smokers | nicotine dependence | Nicotine (NPC182570) | |
| NCT00364156 | Comparison of Standard Versus Extended Nicotine Patch Therapy for Smoking Cessation | nicotine dependence | Nicotine (NPC182570) | |
| NCT01857531 | Proof-Of-Concept Investigation With a Neurosteroid Analog (Ganaxolone) as a Smoking Cessation Candidate | smoking cessation | Nicotine (NPC182570) | |
| NCT02896400 | Optimizing Tobacco Dependence Treatment in the Emergency Department | nicotine dependence | Nicotine (NPC182570) | |
| NCT00749463 | Evaluation of the Safety Profile, Quit and Reduction Rate After Nicorette® Gum and Patch Treatment | nicotine dependence | Nicotine (NPC182570) | |
| NCT02356913 | Comparison of Pharmacokinetic Profiles of 3 New Nicotine Gum Formulations With a Reference Gum | smoking cessation | Nicotine (NPC182570) | |
| NCT04696380 | Do Flavors Increase the Addiction Potential of Nicotine? | nicotine dependence | Nicotine (NPC182570) | |
| NCT04003805 | Biomarkers of Exposure and Effect in Standardized Research E-cigarette (SREC) Users | nicotine dependence | Nicotine (NPC182570) | |
| NCT01184664 | Combination of Nicotine Replacement Therapy (NRT) and Varenicline to Increase Cessation of Tobacco (CONVICT Study) | smoking cessation;nicotine dependence | Nicotine (NPC182570) | |
| NCT05176418 | IV Pulsed-Nicotine as a Model of Smoking: The Effects of Dose and Delivery Rate | nicotine dependence | Nicotine (NPC182570) | |
| NCT00873392 | Efficacy of Transdermal Nicotine, on Motor Symptoms in Advanced Parkinson's Disease | Parkinson disease | Nicotine (NPC182570) | |
| NCT05024747 | A Bioequivalence Study of 21 Milligram (mg) Nicotine Transdermal Patches (NicoDerm CQ, GlaxoSmithKline [GSK] Dungarvan) Compared to the Current Marketed 21 mg Nicotine Transdermal Patches (NicoDerm CQ, Alza) in Healthy Adult Smokers | nicotine dependence | Nicotine (NPC182570) | |
| NCT02521662 | The Use of Nicotine Patches Together With E-cigarettes (With and Without Nicotine) for Smoking Cessation | smoking cessation | Nicotine (NPC182570) | |
| NCT01172561 | Community Awareness Resources and Education - Project 1 | cervical cancer | Nicotine (NPC182570) | |
| NCT00507975 | Study of Nicotine Patch in Pregnancy (SNIPP) | nicotine dependence | Nicotine (NPC182570) | |
| NCT00018187 | Combinations of Pharmacologic Smoking Cessation Treatments | nicotine dependence | Nicotine (NPC182570) | |
| NCT02796391 | Facilitating Smoking Cessation With Reduced Nicotine Cigarettes | smoking cessation | Nicotine (NPC182570) | |
| NCT05311085 | Cytisine and E-cigarettes With Supportive Text-messaging for Smoking Cessation (Cess@Tion) | smoking cessation | Nicotine (NPC182570) | |
| NCT05130788 | WeChat Quit Coach Pilot Study | smoking cessation | Nicotine (NPC182570) | |
| NCT00000454 | Smoking Cessation in Alcoholism Treatment | nicotine dependence | Nicotine (NPC182570) | |
| NCT04974580 | Research and Innovation to Stop E-cigarette/Vaping in Young Adults | nicotine dependence | Nicotine (NPC182570) | |
| NCT01329614 | Effects of Nicotine Replacement and Repeated Cue Exposure on Cigarette Craving | nicotine dependence | Nicotine (NPC182570) | |
| NCT01517022 | Intensive Smoking-cessation Intervention Versus Smoking-cessation Advice in Smear-positive Patients With Pulmonary Tuberculosis | tuberculosis | Nicotine (NPC182570) | |
| NCT03290430 | End Nicotine Dependence Clinic | smoking cessation | Nicotine (NPC182570) | |
| NCT00000444 | Timing of Smoking Intervention in Alcohol Treatment (Nicotine Patch) | nicotine dependence | Nicotine (NPC182570) | |
| NCT05253573 | Mobile Health Technology for Personalized Tobacco Cessation Support Among Cancer Survivors in Laos | nicotine dependence | Nicotine (NPC182570) | |
| NCT03091699 | Effects of Exercise and Nicotine on Cognition in Smokers | smoking cessation | Nicotine (NPC182570) | |
| NCT04583410 | Efficacy of Nicotine in Preventing COVID-19 Infection | COVID-19;severe acute respiratory syndrome | Nicotine (NPC182570) | |
| NCT00764439 | Pilot Study of a New Nicotine Replacement Therapy | smoking cessation | Nicotine (NPC182570) | |
| NCT01736085 | Providing Free Nicotine Patches to Quitline Smokers | smoking cessation | Nicotine (NPC182570) | |
| NCT01292642 | Cognitive Behavioral Therapy and the Nicotine Transdermal Patch for Cannabis Dependence and Nicotine Dependence | cannabis dependence;nicotine dependence | Nicotine (NPC182570) | |
| NCT00956943 | Assessment of High Dose Transdermal Nicotine for Fast Metabolizers of Nicotine | nicotine dependence | Nicotine (NPC182570) | |
| NCT01495819 | Nicotine Reinforcement and Aversion in Young Adult Light Smokers | nicotine dependence | Nicotine (NPC182570) | |
| NCT00004551 | Behavioral Counseling for Alcohol Dependent Smokers (Nicotine Patch) | nicotine dependence | Nicotine (NPC182570) | |
| NCT02681510 | UW-CTRI Triple Medication Smoking Cessation Study | nicotine dependence | Nicotine (NPC182570) | |
| NCT00018161 | Treatment to Quit Smoking | nicotine dependence | Nicotine (NPC182570) | |
| NCT01056926 | Neurobiology of Nicotine and Non-nicotine Components of Tobacco Addiction | nicotine dependence | Nicotine (NPC182570) | |
| NCT02232737 | Project 3, Study 2: Extended Exposure to Low Nicotine Content Cigarettes in People With Current Affective Disorders | nicotine dependence | Nicotine (NPC182570) | |
| NCT04994444 | Preloading With Nicotine Replacement Therapy in HIV-positive Smokers to Improve Self-Efficacy and Quit Attempts | nicotine dependence | Nicotine (NPC182570) | |
| NCT00840242 | Smoking Reduction or Cessation With Nicotine Replacement Therapy | smoking cessation | Nicotine (NPC182570) | |
| NCT01702532 | Nicotine Mouth Film for Craving Relief. | smoking cessation | Nicotine (NPC182570) | |
| NCT05154669 | Integrating Financial Coaching and Smoking Cessation Coaching | nicotine dependence | Nicotine (NPC182570) | |
| NCT01321931 | Multiple-dose Nicotine Pharmacokinetics With Three Oral Nicotine Replacement Products | nicotine dependence | Nicotine (NPC182570) | |
| NCT05236894 | Appeal of Nicotine Pouches Versus Cigarettes in the Ohio Appalachia Population | carcinoma | Nicotine (NPC182570) | |
| NCT00831155 | Extinction Based Treatment for Nicotine Dependence | nicotine dependence | Nicotine (NPC182570) | |
| NCT01199380 | Behavioral Activation Intervention for Smoking Cessation in Smokers With Depressive Symptoms | nicotine dependence | Nicotine (NPC182570) | |
| NCT01819259 | Nicotine, Non-Smokers With and Without ADHD, and Genetics Study | attention deficit hyperactivity disorder | Nicotine (NPC182570) | |
| NCT03897439 | Individualizing Pharmacotherapy for African American Smokers | smoking cessation | Nicotine (NPC182570) | |
| NCT00966901 | Photoirritation and Photoallergic Potential of a New Nicotine Patch | smoking cessation | Nicotine (NPC182570) | |
| NCT00715871 | Clinical Study of Smoke-Break Liquid Cigarettes | nicotine dependence | Nicotine (NPC182570) | |
| NCT00710034 | Efficacy of Oral Tobacco Products Compared to a Medicinal Nicotine | nicotine dependence | Nicotine (NPC182570) | |
| NCT01484717 | Interactive Voice Response Technology to Mobilize Contingency Management for Smoking Cessation | nicotine dependence | Nicotine (NPC182570) | |
| NCT05224050 | Heart Rate Variability Biofeedback for Smoking Cessation Treatment | nicotine dependence | Nicotine (NPC182570) | |
| NCT03482583 | Tobacco Use Treatment in Cancer Patients | nicotine dependence | Nicotine (NPC182570) | |
| NCT02840435 | Study on Sit to Quit Phone Intervention | nicotine dependence | Nicotine (NPC182570) | |
| NCT03879889 | Smoking Reduction Intervention for Smoking Parents of Paediatric Patients in Hong Kong | nicotine dependence | Nicotine (NPC182570) | |
| NCT00295984 | Nicotine Skin Patch in Treating Patients With Kaposi's Sarcoma | sarcoma | Nicotine (NPC182570) | |
| NCT04084210 | Impact of Alternative Nicotine-Delivery Products on Combustible Cigarette Use | nicotine dependence | Nicotine (NPC182570) | |
| NCT02250664 | Project 2, Study 2: Extended Exposure to Low Nicotine Content Cigarettes in Opioid Abusers | nicotine dependence | Nicotine (NPC182570) | |
| NCT00793468 | Relapse Prevention Study in Newly Abstinent Smokers | substance dependence | Nicotine (NPC182570) | |
| NCT01429129 | Randomized Controlled Trial of Mailed Nicotine Replacement Therapy to Canadian Smokers | nicotine dependence | Nicotine (NPC182570) | |
| NCT03611881 | Assessing the Integration of Tobacco Cessation Treatment Into Lung Cancer Screening (LCS) | nicotine dependence | Nicotine (NPC182570) | |
| NCT02019459 | Very Low Nicotine Cigarettes in Smokers With Schizophrenia | nicotine dependence | Nicotine (NPC182570) |
❱❱❱ Associated Human Diseases and Detailed Association Evidence
How do we define the Plant-Targeted Human Disease Association?
Associated human diseases of an individual plant are summurized based on FOUR types of association evidence, these include:
❶ Association by Therapeutic Target: Bioactive protein targets of the plant were defined in "Molecular Targets" section, target-disease associations collected from TTD database were subsequently used to build the associations between the plant and its targeted human diseases.
❷ Association by Disease Gene Reversion: Plant and a specific disease will be associated when >= 1 plant target gene overlaped with disease's DEGs.
❸ Association by Clinical Trials of Plant: Plant and a specific disease will be associated when >= 1 clinical trial (the plant is the intervetion) can be matched in ClinicalTrials.gov database.
❹ Association by Clinical Trials of Plant Ingredients: Plant and a specific disease will be associated when >= 1 clinical trial (the plant ingredient is the intervetion) can be matched in ClinicalTrials.gov database.
Associated Disease of the Plant |
Association Type & Detailed Evidence |
|---|---|
ParkinsonismDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8A00 |
CHRFAM7A,CHRNA7
NCT00957918,NCT01560754,NCT00873392,NCT03865121 |
SchizophreniaDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6A20 |
CHRNA7,CHRFAM7A
NCT00527020,NCT05301660 |
DepressionDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6A70-6A7Z |
CHRFAM7A,CHRNA2,CHRNA7,CHRNA4
|
Corneal diseaseDisease Category: 09.Diseases of the visual systemDisease ICD-11 Code: 9A76-9A78 |
CHRNA7,CHRNA4,CHRFAM7A,CHRNA2
|
AsthmaDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CA23 |
CBR1,CHRNA7,CHRFAM7A
|
Alzheimer diseaseDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8A20 |
CHRFAM7A,CHRNA7,CHRNA4
|
Cannabis use disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6C41 |
NCT04124432,NCT01292642,NCT02955329
|
COVID-19Disease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D6Y |
NCT04583410,NCT04598594,NCT04608201
|
Carcinosarcoma of uterusDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C76.43 |
CHRNA4,CHRNA3,TDP1
|
Diffuse large B-cell lymphomasDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A81 |
CBR1,CHRNA3,TDP1
|
Cytomegaloviral diseaseDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D82 |
CHRNA7,CHRNA4,CHRNA3
|
Malignant neoplasms of adrenal glandDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D11 |
CHRNA7,CHRNA4,CBR1
|
Sex development malformative disorderDisease Category: 20.Developmental anomaliesDisease ICD-11 Code: LD2A |
CHRNA7,CHRFAM7A
|
Mental/behavioural/neurodevelopmental disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6E20-6E8Z |
CHRFAM7A,CHRNA7
|
Sexual dysfunctionDisease Category: 17.Conditions related to sexual healthDisease ICD-11 Code: HA00-HA01 |
CHRNA7,CHRFAM7A
|
General pain disorderDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8E43 |
CHRFAM7A,CHRNA7
|
Breast cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C60-2C6Y |
CHRFAM7A,CHRNA7
|
Attention deficit hyperactivity disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6A05 |
CHRNA4
NCT01819259 |
Dissociative neurological symptom disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6B60 |
CHRFAM7A,CHRNA7
|
Adaptive immunity immunodeficiencyDisease Category: 04.Diseases of the immune systemDisease ICD-11 Code: 4A01 |
CHRFAM7A,CHRNA7
|
Mild neurocognitive disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6D71 |
CHRFAM7A,CHRNA7
|
Severe acute respiratory syndromeDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D65 |
NCT04598594,NCT04583410
|
Alcohol beverageDisease Category: X.Extension CodesDisease ICD-11 Code: XM1A61 |
NCT00616746,NCT00699556
|
SarcoidosisDisease Category: 04.Diseases of the immune systemDisease ICD-11 Code: 4B20 |
NCT02265874,NCT00701207
|
Chronic obstructive pulmonary diseaseDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CA22 |
NCT00132236,NCT01539434
|
Ageing associated decline in intrinsic capacityDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: MG2A |
NCT05018117,NCT03408574
|
Neuroendocrine carcinomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C34 |
NCT03856515,NCT05236894
|
Serous cystadenoma,borderline malignancy of ovaryDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C73.4 |
CHRNA3,TDP1
|
PheochromocytomaDisease Category: X.Extension CodesDisease ICD-11 Code: XH3854 |
CHRNA7,CHRNA3
|
Malignant neoplasms of biliary tract, distal bile ductDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C15 |
CHRNA7,CHRNA3
|
HepatoblastomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C12.01 |
CHRNA7,CHRNA3
|
Squamous cell carcinoma of bronchus or lungDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25.2 |
CHRNA7,CBR1
|
West Nile virus infectionDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D46 |
CHRNA2,CBR1
|
Adenocarcinoma of pancreasDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C10.0 |
CHRNA3,CBR1
|
Mesothelioma of pleuraDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C26.0 |
CBR1,TDP1
|
Nicotine use disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6C4A |
CHRNA2,CHRNA4,CHRNA7,CHRFAM7A
NCT03972137,NCT04709471,NCT00715871,NCT00392379,NCT01314001,NCT00046813,NCT04003805,NCT05233228,NCT03538938,NCT00749463,NCT00364156,NCT03272685,NCT01048944,NCT01199380,NCT05487807,NCT00586482,NCT01892813,NCT01926626,NCT03879889,NCT00831155,NCT01296698,NCT04020718,NCT01136642,NCT00664755,NCT04969198,NCT00108537,NCT03809897,NCT01429129,NCT05024747,NCT01329614,NCT03611881,NCT00879918,NCT02271919,NCT02501265,NCT00507975,NCT01056926,NCT00960778,NCT04126135,NCT05335915,NCT00775944,NCT01867411,NCT01664741,NCT02019459,NCT01621022,NCT04740008,NCT04606797,NCT02927847,NCT00004551,NCT00469079,NCT01184664,NCT04262817,NCT00939029,NCT02162849,NCT01417429,NCT00969137,NCT00777569,NCT01104896,NCT00000447,NCT04974580,NCT00657020,NCT02964182,NCT01592695,NCT03634839,NCT01091363,NCT01907594,NCT02179034,NCT03000387,NCT00218296,NCT04590404,NCT01980550,NCT03243630,NCT04521647,NCT03069482,NCT00105482,NCT03635333,NCT02245308,NCT05224050,NCT03442413,NCT03541044,NCT00296647,NCT01783912,NCT02371850,NCT03543137,NCT02487953,NCT01208935,NCT03492463,NCT01047527,NCT01372254,NCT01292642,NCT02840435,NCT04696380,NCT04490057,NCT03168191,NCT04084210,NCT00218283,NCT02250534,NCT04999644,NCT00307203,NCT03580525,NCT01303861,NCT00086411,NCT03482583,NCT00061074,NCT00064844,NCT05159934,NCT01457469,NCT02147132,NCT02511704,NCT02232737,NCT00332644,NCT00326781,NCT05206435,NCT01351766,NCT00000454,NCT02439944,NCT01806779,NCT02081144,NCT01478230,NCT00000444,NCT00985985,NCT02896400,NCT00888459,NCT01702519,NCT05154669,NCT05060965,NCT00067158,NCT00710034,NCT01484717,NCT00000437,NCT02482233,NCT01654107,NCT02250664,NCT02681510,NCT00956943,NCT04033237,NCT01058876,NCT00258479,NCT02355665,NCT04946825,NCT02906644,NCT03421210,NCT01321931,NCT01065506,NCT05176418,NCT01444131,NCT03612960,NCT01438944,NCT04994444,NCT01018394,NCT03262233,NCT01506908,NCT01495819,NCT03176784,NCT03521141,NCT00734617,NCT01932996,NCT05253573,NCT03860077,NCT00018148,NCT00018187,NCT02721082,NCT02587312,NCT01406223,NCT03163303,NCT00755716,NCT00018161,NCT01050569,NCT04063267,NCT01034020,NCT01989507,NCT01560507,NCT04535362,NCT00132236,NCT02905734,NCT05351606 |
GlaucomaDisease Category: 09.Diseases of the visual systemDisease ICD-11 Code: 9C61 |
CHRNA2
|
Digestive system diseaseDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DE2Z |
CHRNA4
|
Tonus and reflex abnormalityDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: MB47 |
CHRNA2
|
Hypertensive crisisDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA03 |
CHRNA4
|
Aneurysm/dissectionDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BD50 |
CHRNA4
|
Mood disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6A60-6E23 |
CHRNA4
|
HypotensionDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA20-BA21 |
CHRNA4
|
Irritable bowel syndromeDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DD91 |
CHRNA4
|
Cervical cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C77 |
NCT01172561
|
Unspecific body region injuryDisease Category: 22.Injury, poisoning or certain other consequences of external causesDisease ICD-11 Code: ND56 |
NCT02893345
|
HIV-infected patients with tuberculosisDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1B10-1B14 |
NCT01517022
|
Cocaine use disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6C45 |
NCT02018263
|
Breast in situ carcinomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2E65 |
NCT03808818
|
Substance abuseDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6C40 |
NCT00793468
|
Depressive disorders, unspecifiedDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6A7Z |
NCT04433767
|
Nausea/vomitingDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: MD90 |
NCT00553709
|
Heart failureDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BD10-BD1Z |
NCT00543881
|
Pleomorphic sarcomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B54 |
NCT00295984
|
Age-associated cognitive declineDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: MB21.0 |
NCT00091468
|
Symptoms and signs involving cognition, unspecifiedDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: MB21.Z |
NCT02720445
|
Lung cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25 |
NCT00032084
|
Idiopathic pulmonary fibrosisDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CB03.4 |
CHRNA4
|
Melanoma of skinDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C30 |
CHRNA7
|
Hepatocellular carcinoma of liverDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C12.02 |
CHRNA3
|
Adenocarcinoma of bronchus or lungDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25.0 |
CBR1
|
Chikungunya virus diseaseDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D40 |
CHRNA4
|
Malignant neoplasms of thymusDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C27 |
CHRNA2
|
Invasive carcinoma of breastDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C61 |
CHRNA4
|
Heart failure, unspecifiedDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BD1Z |
CHRNA3
|
Germ cell tumour of testisDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C80.2 |
CHRNA3
|

