Collective Molecular Activities of the Plant: Tinospora Crispa
Plant ID: NPO26700
Plant Latin Name: Tinospora Crispa
Taxonomy Genus: Tinospora
Taxonomy Family: Menispermaceae
Plant External Links:
NCBI TaxonomyDB:
285591
Plant-of-the-World-Online:
n.a.
Country/Region:
Philippines; Indonesia; India; Malaysia; Vietnam; Laos; ThailandTraditional Medicine System:
Indian FolkPhilippines; Indonesia; India; Malaysia; Vietnam; Laos; Thailand
Overview of Ingredients
30 All known Ingredients in Total
Unique ingredients have been isolated from this plant.Plant-Ingredients Associations were manually curated from publications or collected from other databases.
8 Ingredients with Acceptable Bioavailablity
Unique ingredients exhibit acceptable human oral bioavailablity, according to the criteria of SwissADME [PMID: 28256516] and HobPre [PMID: 34991690]. The criteria details:SwissADME: six descriptors are used by SwissADME to evaluate the oral bioavailability of a natural product:
☑ LIPO(Lipophility): -0.7 < XLOGP3 < +5.0
☑ SIZE: 150g/mol < MW < 500g/mol
☑ POLAR(Polarity): 20Ų < TPSA < 130Ų
☑ INSOLU(Insolubility): -6 < Log S (ESOL) < 0
☑ INSATU(Insaturation): 0.25 < Fraction Csp3 < 1
☑ FLEX(Flexibility): 0 < Num. rotatable bonds < 9
If 6 descriptors of a natural plant satisfy the above rules, it will be labeled high HOB.
HobPre: A natural plant ingredient with HobPre score >0.5 is labeled high human oral availability (HOB)
25 Ingredients with experimental-derived Activity
Unique ingredients have activity data available.Ingredient Structrual Cards
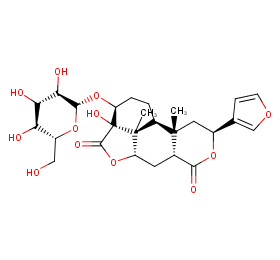
Ingredient ID: NPC82602
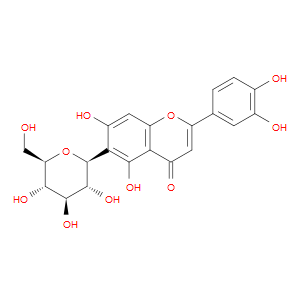
Ingredient ID: NPC53545
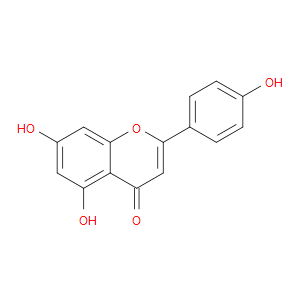
Ingredient ID: NPC50898
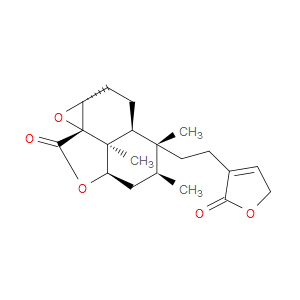
Ingredient ID: NPC486158
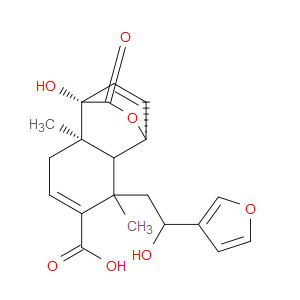
Ingredient ID: NPC486157
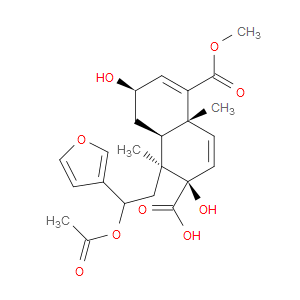
Ingredient ID: NPC486156
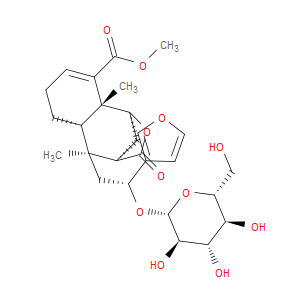
Ingredient ID: NPC486155
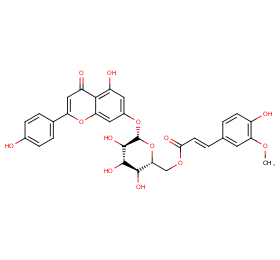
Ingredient ID: NPC472993

Ingredient ID: NPC472992
Ingredient ID: NPC472991
Ingredient ID: NPC472990
Ingredient ID: NPC289774
Ingredient ID: NPC279121

Ingredient ID: NPC27283
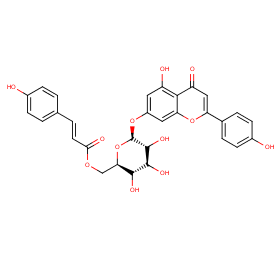
Ingredient ID: NPC270675
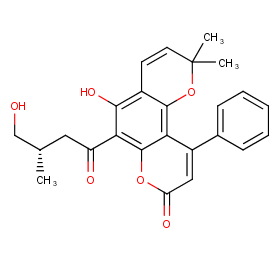
Ingredient ID: NPC252250
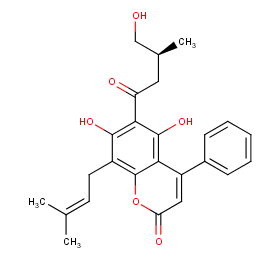
Ingredient ID: NPC246023

Ingredient ID: NPC230775
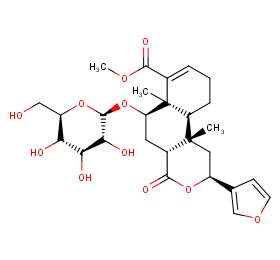
Ingredient ID: NPC211777
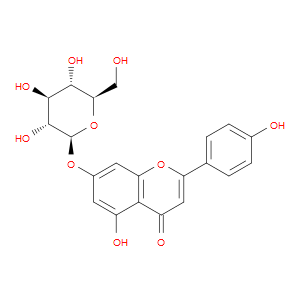
Ingredient ID: NPC210003
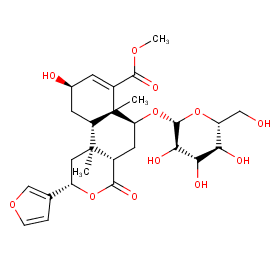
Ingredient ID: NPC198047
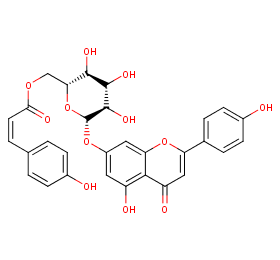
Ingredient ID: NPC195685
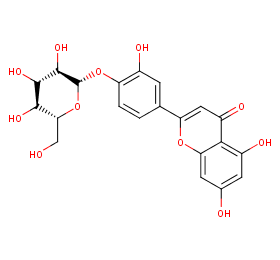
Ingredient ID: NPC191306
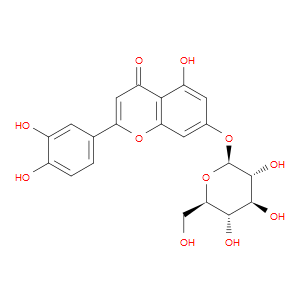
Ingredient ID: NPC189142
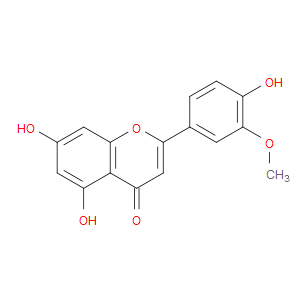
Ingredient ID: NPC183950
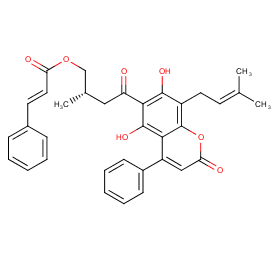
Ingredient ID: NPC151243
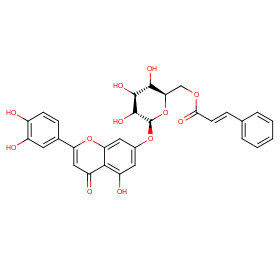
Ingredient ID: NPC12647
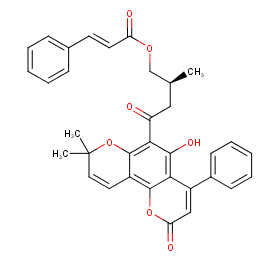
Ingredient ID: NPC12215
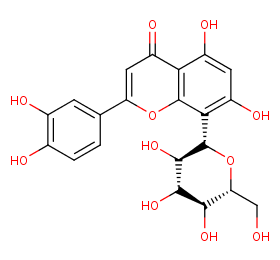
Ingredient ID: NPC117418
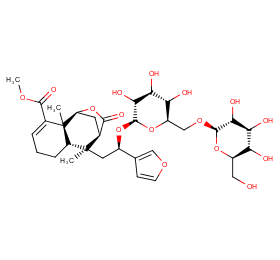
Ingredient ID: NPC100333
Classification of Human Proteins Collectively Targeted by the Plant
Detailed Information of Target Proteins
| Target Type | Protein Class | Gene ID | Protein Name | Uniprot ID | Target ChEMBL ID |
|---|---|---|---|---|---|
| Cytochrome P450 | Cytochrome P450 family 1 | CYP1A1 | Cytochrome P450 1A1 | P04798 | CHEMBL2231 |
| Cytochrome P450 | Cytochrome P450 family 1 | CYP1A2 | Cytochrome P450 1A2 | P05177 | CHEMBL3356 |
| Cytochrome P450 | Cytochrome P450 family 1 | CYP1B1 | Cytochrome P450 1B1 | Q16678 | CHEMBL4878 |
| Therapeutic Target | Enzyme | NOX4 | NADPH oxidase 4 | Q9NPH5 | CHEMBL1250375 |
| Therapeutic Target | Hydrolase | ACHE | Acetylcholinesterase | P22303 | CHEMBL220 |
| Therapeutic Target | Lyase | CA7 | Carbonic anhydrase VII | P43166 | CHEMBL2326 |
| Therapeutic Target | Lyase | CA4 | Carbonic anhydrase IV | P22748 | CHEMBL3729 |
| Therapeutic Target | Lyase | CA12 | Carbonic anhydrase XII | O43570 | CHEMBL3242 |
| Therapeutic Target | Metallo protease | MMP12 | Matrix metalloproteinase 12 | P39900 | CHEMBL4393 |
| Therapeutic Target | Nuclear hormone receptor subfamily 3 | ESR2 | Estrogen receptor beta | Q92731 | CHEMBL242 |
| Therapeutic Target | Oxidoreductase | HSD17B1 | Estradiol 17-beta-dehydrogenase 1 | P14061 | CHEMBL3181 |
| Therapeutic Target | Protein Kinase | FLT3 | Tyrosine-protein kinase receptor FLT3 | P36888 | CHEMBL1974 |
| Therapeutic Target | Protein Kinase | MKNK1 | MAP kinase-interacting serine/threonine-protein kinase MNK1 | Q9BUB5 | CHEMBL4718 |
| Therapeutic Target | Protein Kinase | CSNK2A1 | Casein kinase II alpha | P68400 | CHEMBL3629 |
| Therapeutic Target | Protein Kinase | MKNK2 | MAP kinase signal-integrating kinase 2 | Q9HBH9 | CHEMBL4204 |
| Therapeutic Target | Secreted protein | THPO | Thrombopoietin | P40225 | CHEMBL1293256 |
| Therapeutic Target | Secreted protein | TTR | Transthyretin | P02766 | CHEMBL3194 |
| Therapeutic Target | Serine protease | DPP4 | Dipeptidyl peptidase IV | P27487 | CHEMBL284 |
| Therapeutic Target | Structural protein | LMNA | Prelamin-A/C | P02545 | CHEMBL1293235 |
| Therapeutic Target | Transcription factor | HIF1A | Hypoxia-inducible factor 1 alpha | Q16665 | CHEMBL4261 |
Clinical trials associated with plant from natural product (NP) & plant level:
| Clinical trials type | Number of clinical trials | |
|---|---|---|
| 565 | ||
| NCT ID | Title | Condition | Form in clinical use | Associated by plant or compound |
|---|---|---|---|---|
| NCT01840007 | Pilot Study Evaluating the Efficacy and Safety of Metformin in Melanoma | metastatic melanoma | Metformin (NPC230775) | |
| NCT04925063 | The Effect of Metformin in Patients With Newly Diagnosed mHSPC | metastatic prostate cancer | Metformin (NPC230775) | |
| NCT01305551 | BE Study of the Fixed Dose Combination of 5 mg Saxagliptin and 500 mg Metformin HCl XR Tablet Relative to a 5 mg Saxagliptin (Onglyza™) Tablet and a 500 mg Metformin HCl XR (Glifage® XR Marketed in Brazil by Merck S.A.) Tablet Co-Administered to Healthy Subjects in the Fasted and Fed States | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01685762 | Metformin for the Treatment of Endometrial Hyperplasia | endometrial hyperplasia | Metformin (NPC230775) | |
| NCT01234649 | Combined Liraglutide and Metformin Therapy in Women With Previous Gestational Diabetes Mellitus (GDM) | abnormal glucose tolerance;metabolic syndrome | Metformin (NPC230775) | |
| NCT01814748 | A Study of the Safety and Efficacy of Omarigliptin (MK-3102) in ≥18 and <45 Year-Old Participants With Type 2 Diabetes Mellitus and Inadequate Glycemic Control (MK-3102-028) | diabetes mellitus | Metformin (NPC230775) | |
| NCT02086526 | Targeting Pathways in Polycystic Ovary Syndrome (PCOS) Using Metformin (MET) | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT01677897 | Impact of the Addition of Metformin to Abiraterone in Metastatic Prostate Cancer Patients | metastatic prostate cancer | Metformin (NPC230775) | |
| NCT03320694 | Comparison of Metformin and Insulin for Management of Gestational Diabetes Mellitus | gestational diabetes | Metformin (NPC230775) | |
| NCT02186847 | Chemotherapy and Radiation Therapy With or Without Metformin Hydrochloride in Treating Patients With Stage III Non-small Cell Lung Cancer | bronchoalveolar adenocarcinoma;large cell lung carcinoma | Metformin (NPC230775) | |
| NCT01877564 | A Randomized Pilot Study to Evaluate the Effects of a Short Course of Metformin Versus No Therapy in the Period Prior to Hysterectomy for Grade 1-2 Adenocarcinoma of the Endometrium in Obese Non-Diabetic Women | adenocarcinoma | Metformin (NPC230775) | |
| NCT04899349 | Study of Safety and Efficacy of Dapagliflozin + Metformin XR Versus Metformin XR in Participants With HR+, HER2-, Advanced Breast Cancer While on Treatment With Alpelisib and Fulvestrant | breast cancer | Metformin (NPC230775) | |
| NCT00859898 | Study of Dapagliflozin in Combination With Metformin XR to Initiate the Treatment of Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT03072485 | Phase 1 Study of the Effects of Combining Topical FDA-approved Drugs on Age-related Pathways on the Skin of Healthy Volunteers | aging | Metformin (NPC230775) | |
| NCT00772174 | Efficacy and Safety Study of Pioglitazone Combined With Metformin on Metabolic Syndrome in Subjects With Type 2 Diabetes | diabetes mellitus | Metformin (NPC230775) | |
| NCT00672386 | A Study of the Safety and Effectiveness of a R256918 in Patients With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02780024 | Metformin, Neo-adjuvant Temozolomide and Hypo- Accelerated Radiotherapy Followed by Adjuvant TMZ in Patients With GBM | glioblastoma multiforme | Metformin (NPC230775) | |
| NCT00868790 | A Study of the Safety and Efficacy of MK-3577 in Participants With Type 2 Diabetes Mellitus (MK-3577-009) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT04945148 | Oxidative Phosphorylation Targeting In Malignant Glioma Using Metformin Plus Radiotherapy Temozolomide | glioblastoma multiforme | Metformin (NPC230775) | |
| NCT01874080 | Bioequivalence Study | diabetes mellitus | Metformin (NPC230775) | |
| NCT05426044 | Metformin as a Neuroprotective Therapy for Glaucoma - A Randomized Controlled Trial | glaucoma | Metformin (NPC230775) | |
| NCT04121468 | A Phase I Double Blind Study of Metformin Acting on Endogenous Neural Progenitor Cells in Children With Multiple Sclerosis | multiple sclerosis | Metformin (NPC230775) | |
| NCT04780854 | Drug Repurposing for the Prevention of Chemotherapy-induced Peripheral Neuropathy (CIPN) | peripheral neuropathy | Metformin (NPC230775) | |
| NCT00924573 | Comparative Study of HOE490 O (Glimepiride and Metformin) Compared With Placebo on Top of Glimepiride | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT03039075 | Metformin SR Tablet (DuLeNing) and Glucophage in Patients With Type 2 Diabetes | diabetes mellitus | Metformin (NPC230775) | |
| NCT00614055 | Comparison of Two NN5401 Formulations Versus Insulin Glargine, All in Combination With Metformin in Subjects With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00087516 | Monotherapy Study in Patients With Type 2 Diabetes Mellitus (0431-021) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00263276 | A Trial of BMS-512148 in Patients With Type 2 Diabetes Mellitus | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01192152 | Bioequivalence Study of the Fixed Dose Combination of 5 mg Saxagliptin/1000 mg Metformin XR (Manufactured in Mt Vernon, IN) Relative to 5 mg of Onglyza and 2 × 500 mg Glucophage XR | diabetes mellitus | Metformin (NPC230775) | |
| NCT00930579 | Pre-Surgical Study: Effect of Metformin on Breast Cancer Proliferation | breast cancer | Metformin (NPC230775) | |
| NCT00770835 | Efficacy and Safety of Pioglitazone in Treating Subjects With Vascular Complications Associated With Type 2 Diabetes Mellitus. | diabetes mellitus | Metformin (NPC230775) | |
| NCT00960076 | An 18 Week Efficacy and Safety Study of Saxagliptin and Metformin XR Combination in Subjects With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00046462 | Determine Whether Glycemic Control is Different Between Lantus & a 3rd Oral Agent When Failure With Other Treatment | diabetes mellitus | Metformin (NPC230775) | |
| NCT00975286 | 24-week Treatment With Lixisenatide in Type 2 Diabetes Insufficiently Controlled With Metformin and Insulin Glargine | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01023581 | Efficacy and Safety of Alogliptin Plus Metformin in Patients With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT04607252 | Metformin Plus Megestrol Acetate as a Fertility-sparing Treatment in Patients With Atypical Endometrial Hyperplasia | endometrial hyperplasia | Metformin (NPC230775) | |
| NCT01204294 | Comprehensive Add on Study in Japan | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT03710343 | Metformin for Motor and Cognitive Improvement in Children With Cerebral Palsy: A Feasibility Study | spastic cerebral palsy | Metformin (NPC230775) | |
| NCT02018731 | L-citrulline and Metformin in Becker's Muscular Dystrophy | muscular dystrophy | Metformin (NPC230775) | |
| NCT01175473 | Effects of Lixisenatide Compared to Liraglutide on the Postprandial Plasma Glucose in Patients With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00961909 | A Study of RO5095932 in Patients With Type 2 Diabetes Mellitus | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01775176 | Effect of Weight & Insulin Sensitivity on Reproductive Function in PCOS: Studies in Skeletal Muscle & Adipose | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT01638676 | A Phase I/II Trial of Vemurafenib and Metformin to Melanoma Patients | melanoma | Metformin (NPC230775) | |
| NCT02614859 | Bicalutamide With or Without Metformin for Biochemical Recurrence in Overweight or Obese Prostate Cancer Patients | prostate carcinoma | Metformin (NPC230775) | |
| NCT02294006 | Activity and Safety of Everolimus+Octreotide LAR+Metformin in Advanced Pancreatic Well-differentiated NETs | pancreatic neuroendocrine tumor | Metformin (NPC230775) | |
| NCT03259919 | Metformin Treatment of Pregnant Women With Polycystic Ovary Syndrome: a Pilot Study | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT01059825 | Study Of Safety And Efficacy Of Ertugliflozin (PF-04971729, MK-8835) In Participants With Type 2 Diabetes (MK-8835-016) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT04068246 | The AMPK Modulator Metformin as a Novel Adjunct to Conventional Therapy in Rheumatoid Arthritis Patients | rheumatoid arthritis | Metformin (NPC230775) | |
| NCT03137186 | Repurposing Metformin as Anticancer Drug: in Advanced Prostate Cancer | prostate cancer | Metformin (NPC230775) | |
| NCT00519480 | A Study To Assess The Safety And Tolerability Of GSK189075 When Given With A Total Daily Dose Of >/ 2000mg of Metformin | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00714233 | Treatment of Adolescent Polycystic Ovary Syndrome (PCOS) | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT04826692 | TEsting METformin Against Cognitive Decline in HD | Huntington disease | Metformin (NPC230775) | |
| NCT02644954 | Assessing the Efficacy and Safety of Metformin in Treatment of Moderate Psoriasis | psoriasis | Metformin (NPC230775) | |
| NCT02659306 | Metformin Immunotherapy in HIV Infection | HIV infection | Metformin (NPC230775) | |
| NCT01217892 | Evaluation of Dapagliflozin Taken Twice-daily | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01613755 | Metformin-Dipyridamole Interaction Trial | diabetes mellitus | Metformin (NPC230775) | |
| NCT03349775 | Cardiometabolic Disease and Pulmonary Hypertension | obesity | Metformin (NPC230775) | |
| NCT00703755 | A Randomized, Double-Blind Trial Comparing the Efficacy and Safety of Fenofibrate, Metformin, Their Combination and Placebo in Patients With Metabolic Syndrome. | metabolic syndrome | Metformin (NPC230775) | |
| NCT03618654 | Durvalumab With or Without Metformin in Treating Participants With Head and Neck Squamous Cell Carcinoma | oral squamous cell carcinoma;oropharynx squamous cell carcinoma | Metformin (NPC230775) | |
| NCT01357876 | Effect of Metformin on Gut Peptides , Bile Acids and Lipid Profiles in Type 2 Diabetics | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02506790 | Neoadjuvant Toremifene With Melatonin or Metformin in Locally Advanced Breast Cancer | breast cancer | Metformin (NPC230775) | |
| NCT02629627 | Conjugated Linoleic Acid / Leucine Versus Metformin on Visceral Fat in Metabolic Syndrome | metabolic syndrome | Metformin (NPC230775) | |
| NCT00396357 | Efficacy/Safety of Vildagliptin and Metformin Combination Therapy in Patients With Type 2 Diabetes Not Well Controlled With Metformin Alone | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT03670043 | Tolerance & Responsiveness Improvement for Metformin (TRIM) | diabetes mellitus | Metformin (NPC230775) | |
| NCT00575588 | 52-week add-on to Metformin Comparison of Saxagliptin and Sulphonylurea, With a 52-week Extension Period | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00333151 | Effect of Liraglutide on Blood Glucose Control in Subjects With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02063802 | Metformin vs Conjugated Linoleic Acid and an Intervention Program With Healthy Habits in Obese Children | obesity | Metformin (NPC230775) | |
| NCT03076281 | Metformin Hydrochloride and Doxycycline in Treating Patients With Head and Neck Squamous Cell Carcinoma That Can Be Removed by Surgery | oral squamous cell carcinoma | Metformin (NPC230775) | |
| NCT02278965 | Metformin and Omega-3 Fatty Acids in Woman With a History of Early Stage Breast Cancer | breast carcinoma in situ | Metformin (NPC230775) | |
| NCT02488564 | A Study of Liposomal Doxorubicin + Docetaxel + Trastuzumab + Metformin in Operable and Locally Advanced HER2 Positive Breast Cancer | breast cancer | Metformin (NPC230775) | |
| NCT05243654 | Efficacy and Tolerability of Adjunct Metformin for Multibacillary Leprosy | leprosy | Metformin (NPC230775) | |
| NCT01215032 | Metformin in Castration-Resistant Prostate Cancer | prostate cancer | Metformin (NPC230775) | |
| NCT01340768 | Study to Compare Sitagliptin Versus Sulfonylurea Treatment During Ramadan Fasting in Patients With Type 2 Diabetes (MK-0431-262) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01296412 | Comparison of Two Treatment Regimens (Sitagliptin Versus Liraglutide) on Participants Who Failed to Achieve Good Glucose Control on Metformin Alone (MK-0431-403) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02931253 | Metformin as an Upstream Therapy in Atrial Fibrillation | atrial fibrillation | Metformin (NPC230775) | |
| NCT01797523 | A Phase II, Single-Arm Study of RAD001 (Everolimus), Letrozole, and Metformin in Patients With Advanced or Recurrent Endometrial Carcinoma | endometrial cancer | Metformin (NPC230775) | |
| NCT00303537 | Metformin in Non-Alcoholic Fatty Liver Disease | Hepatic steatosis | Metformin (NPC230775) | |
| NCT00427154 | Effect of Inhaled Pre-prandial Human Insulin on Blood Glucose Control in Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01766778 | Early add-on Vildagliptin in Patients With Type 2 Diabetes Inadequately Controlled by Metformin | diabetes mellitus | Metformin (NPC230775) | |
| NCT00968812 | CANagliflozin Treatment And Trial Analysis-Sulfonylurea (CANTATA-SU) SGLT2 Add-on to Metformin Versus Glimepiride | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00948402 | PED/PEA-15 Protein, PCOS, Obesity, Insulin Sensitivity Indexes, Metformin, Oral Contraceptives | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT01332071 | Avandamet Bioequivalence Study Brazil - Fed Administration | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02580331 | 1% Metformin Gel in the Treatment of Class II Furcation Defects | periodontitis | Metformin (NPC230775) | |
| NCT02064374 | Effect of Dolutegravir on Metformin Pharmacokinetics in Healthy Adult Subjects | HIV infection | Metformin (NPC230775) | |
| NCT03238495 | Randomized Trial of Neo-adjuvant Chemotherapy With or Without Metformin for HER2 Positive Operable Breast Cancer | breast cancer | Metformin (NPC230775) | |
| NCT04510194 | COVID-OUT: Early Outpatient Treatment for SARS-CoV-2 Infection (COVID-19) | COVID-19;severe acute respiratory syndrome | Metformin (NPC230775) | |
| NCT01804049 | Metformin and Muscle in Insulin-resistant Older Veterans | prediabetes syndrome | Metformin (NPC230775) | |
| NCT00038727 | Diabetes Prevention Program Outcomes Study | diabetes mellitus | Metformin (NPC230775) | |
| NCT00676338 | Safety and Efficacy of Exenatide Once Weekly Injection Versus Metformin, Dipeptidyl Peptidase-4 Inhibitor, or Thiazolidinedione as Monotherapy in Drug-Naive Patients With Type 2 Diabetes (DURATION-4) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01289119 | Efficacy and Safety of Alogliptin in Participants With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02244567 | Effect of Metformin Therapy on Serum Under-carboxylated Osteocalcin Levels in Hyperandrogenic Lean Polycystic Ovarian Syndrome Women | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT01725126 | To Investigate the Safety, Tolerability and Pharmacodynamics of GSK2890457 in Healthy Volunteers and Subjects With Type 2 Diabetes | obesity | Metformin (NPC230775) | |
| NCT03229057 | Comparing the Effects of Oral Contraceptive Pills Versus Metformin | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT00328172 | Efficacy and Safety of 3 Doses of BI1356 (Linagliptin) in Type 2 Diabetes Patients | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00449605 | A Glycemic Control Evaluation of Glimepiride Versus Rimonabant on Top of Metformin in Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00885378 | Efficacy and Safety Study of Saxagliptin + Metformin Immediate Release (IR) Versus Metformin IR Alone in Type 2 Diabetes Mellitus | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT04604678 | Pilot Study Into the Use of Metformin and LDN for Patients With COVID-19 | COVID-19 | Metformin (NPC230775) | |
| NCT02684578 | Metformin for the Minimization of Geographic Atrophy Progression in Patients With AMD | atrophic macular degeneration | Metformin (NPC230775) | |
| NCT01089179 | Bioequivalence Study of Metformin Hydrochloride Extended Release 500 mg Tablet Versus Glucophage XR® 500 mg Tablet in Healthy Volunteers Under Fed Conditions | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01087983 | Lapatinib With Sirolimus or Metformin | cancer | Metformin (NPC230775) | |
| NCT01324180 | Vincristine, Dexamethasone, Doxorubicin, and PEG-asparaginase (VPLD) and Metformin for Relapsed Childhood Acute Lymphoblastic Leukemia (ALL) | acute lymphoblastic leukemia | Metformin (NPC230775) | |
| NCT02967276 | Clinical Activity of Metformin With High-dose of Dexamethasone in Relapse Multiple Myeloma | multiple myeloma | Metformin (NPC230775) | |
| NCT03200015 | Rituximab, Cyclophosphamide, Vincristine and Prednisone (R-CHOP) Plus Metformin in Diffuse Large-B-cell Lymphoma | diffuse large B-cell lymphoma | Metformin (NPC230775) | |
| NCT05215990 | Efficacy of Metformin for Sputum Conversion in Patients With Active Pulmonary Tuberculosis | tuberculosis | Metformin (NPC230775) | |
| NCT01353391 | Metformin in Women With Type 2 Diabetes in Pregnancy Trial | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02275845 | Medico-GDM Trial - Metformine to Prevent Gestational Diabetes Mellitus | gestational diabetes | Metformin (NPC230775) | |
| NCT00851903 | Evaluation of Insulin Glargine in Combination With Sitagliptin in Type 2 Diabetes Patients: EASIE Extension Trial | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00855166 | Evaluation of the Effect of Dapagliflozin in Combination With Metformin on Body Weight in Subjects With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01037660 | Effects of Lipemia and Metformin on Endothelial Function | cardiovascular disease | Metformin (NPC230775) | |
| NCT03151772 | Bioavailability of Disulfiram and Metformin in Glioblastomas | glioblastoma multiforme | Metformin (NPC230775) | |
| NCT00272064 | ELEONOR STUDY: Insulin Glulisine in Type 2 Diabetes Mellitus. | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00727857 | Efficacy and Safety of Pioglitazone and Metformin Combination Therapy in Treating Type 2 Diabetes Mellitus. | diabetes mellitus | Metformin (NPC230775) | |
| NCT04055012 | Effects of Metformin on Low Back Pain | Low back pain | Metformin (NPC230775) | |
| NCT02360059 | Metformin for Reduction of Paclitaxel-Related Neuropathy in Patients With Breast Cancer | breast cancer | Metformin (NPC230775) | |
| NCT00110851 | Safety and Efficacy Study of PN2034 in Type 2 Diabetes Patients on Concomitant Rosiglitazone and Metformin (or Avandamet) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01106690 | The CANTATA-MP Trial (CANagliflozin Treatment and Trial Analysis - Metformin and Pioglitazone) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00754689 | Study of Rimonabant/Metformin Combinations to Investigate Diabetes (Blood Sugar) Control in Patients With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT03841591 | Maternal Weight Gain in Gestational Diabetes Controlled by Metformin Versus Insulin | gestational diabetes | Metformin (NPC230775) | |
| NCT00353691 | Glimepiride vs Metformin as Monotherapy in Pediatric Subjects With Type 2 Diabetes Mellitus | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT03135301 | Letrozole in Clomiphene Resistant Infertile Women With Polycystic Ovarian Syndrome | infertility | Metformin (NPC230775) | |
| NCT02325401 | Dose-finding Study of Metformin With Chemoradiation in Locally Advanced Head and Neck Squamous Cell Carcinoma | head and neck malignant neoplasia | Metformin (NPC230775) | |
| NCT05468554 | Evaluation of Metformin Effect on the Fertility of Women Treated With 131I for Thyroid Cancer | thyroid cancer | Metformin (NPC230775) | |
| NCT02689843 | Effects of Cyproterone Compound-spironolactone, Metformin and Pioglitazone on Inflammatory Markers in PCOS | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT00915772 | Treatment of Type 2 Diabetes With Linagliptin 2.5 mg Bid + Metformin 500 or 1000 mg Bid and Metformin 1000 mg Bid | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT05132439 | MetfOrmin BenefIts Lower Extremities With Intermittent Claudication | intermittent vascular claudication | Metformin (NPC230775) | |
| NCT02279758 | A Pilot Study of Metformin Treatment in Patients With Well-differentiated Neuroendocrine Tumors | neuroendocrine neoplasm | Metformin (NPC230775) | |
| NCT03617458 | Interventions Against Insulin Resistance in Pulmonary Arterial Hypertension | pulmonary arterial hypertension | Metformin (NPC230775) | |
| NCT01240759 | Safety and Efficacy of S-707106 in Subjects With Type 2 Diabetes Mellitus and Inadequate Glycemic Control With Metformin Therapy | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00901979 | A 12 Week Study in Patients With Type 2 Diabetes Mellitus (T2DM) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01926769 | A Phase II Study to Determine the Safety and Efficacy of Second-line Treatment With Metformin and Chemotherapy (FOLFOX6 or FOFIRI) in the Second Line Threatment of Advanced Colorectal Cancer | colorectal adenocarcinoma | Metformin (NPC230775) | |
| NCT00279240 | Life Style Modifications Prevents Type 2 Diabetes in Asian Indians | diabetes mellitus | Metformin (NPC230775) | |
| NCT01340664 | An Efficacy, Safety, and Tolerability Study of Canagliflozin in the Treatment of Patients With Type 2 Diabetes Mellitus With Inadequate Glycemic Control on Metformin Monotherapy | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01581814 | Effects of Drospirenone-ethinylestradiol and/or Metformin on Cardiovascular Risk in Hyperinsulinemic Women With Polycystic Ovary Syndrome | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT01779362 | RISE Adult Medication Study | prediabetes syndrome | Metformin (NPC230775) | |
| NCT04098666 | Metformin in Alzheimer's Dementia Prevention | Cognitive impairment | Metformin (NPC230775) | |
| NCT03000218 | Evaluation of PK and Biomarkers After UDCA Administrations to Subjects Who Are Overweight and Have Liver Problems | Elevated hepatic transaminase;obesity | Metformin (NPC230775) | |
| NCT00420511 | Beta-Cell Function and Sitagliptin Trial (BEST) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01733836 | Prostate Cancer Active Surveillance Metformin Trial | prostate cancer | Metformin (NPC230775) | |
| NCT04300790 | Study to Evaluate the Effect of Metformin in the Prevention of HG in HR[+]/HER2[-] PIK3CA-mut Advanced BC Patients | breast cancer | Metformin (NPC230775) | |
| NCT04414540 | Combining Pembrolizumab and Metformin in Metastatic Head and Neck Cancer Patients | head and neck squamous cell carcinoma | Metformin (NPC230775) | |
| NCT04649502 | Metformin for the Treatment of Hidradenitis Suppurativa (HS) | hidradenitis suppurativa | Metformin (NPC230775) | |
| NCT01002807 | Bioavailability Study of Fixed Dose Combination (FDC) Formulations of Dapagliflozin and Metformin XR Versus Individual Component Coadministered to Healthy Subjects in a Fasted State | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02460354 | Metformin and Congenital Nephrogenic Diabetes Insipidus | diabetes insipidus | Metformin (NPC230775) | |
| NCT01405118 | Effect Of CP-690,550 On The Pharmacokinetics Of Metformin In Healthy Volunteers | rheumatoid arthritis | Metformin (NPC230775) | |
| NCT01980823 | Pre-Surgical Trial of the Combination of Metformin and Atorvastatin in Newly Diagnosed Operable Breast Cancer | breast cancer | Metformin (NPC230775) | |
| NCT03311308 | A Trial of Pembrolizumab and Metformin Versus Pembrolizumab Alone in Advanced Melanoma | melanoma | Metformin (NPC230775) | |
| NCT00881530 | Empagliflozin (BI 10773) in Type Two Diabetes (T2D) Patients, Open Label Extension | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01028391 | 30-Week Extension to an Initial Combination Study (24 Weeks in Duration) of Sitagliptin With Pioglitazone (0431-064) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT04742751 | An Open-Label Pilot Intervention Trial to Prevent Diabetes in Prediabetic Adult Survivors of Childhood Cancer | prediabetes syndrome | Metformin (NPC230775) | |
| NCT00174642 | Opposing Step-by-step Insulin Reinforcement to Intensified Strategy | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT04839237 | The Effect and the Pharmacogenomics Study of Liraglutide in Obese Patients | obesity | Metformin (NPC230775) | |
| NCT03600363 | A Clinical Trial of Metformin in the Maintenance of Non-Hodgkin's Lymphoma Patients | diffuse large B-cell lymphoma;follicular lymphoma | Metformin (NPC230775) | |
| NCT00316758 | A Study to Evaluate the Long-Term Safety and Tolerability of GK Activator (2) in Patients With Type 2 Diabetes. | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01064687 | A Study in Participants With Type 2 Diabetes Mellitus | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02672488 | Metformin Plus Sorafenib for Advanced HCC | hepatocellular carcinoma | Metformin (NPC230775) | |
| NCT02365597 | An Efficacy and Safety Study of Erdafitinib (JNJ-42756493) in Participants With Urothelial Cancer | urogenital neoplasm | Metformin (NPC230775) | |
| NCT00976937 | 24-week Study Comparing Lixisenatide to Sitagliptin as add-on to Metformin in Obese Type 2 Diabetic Patients Younger Than 50 Years | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00909506 | Efficacy and Safety of Adjuvant Metformin for Operable Breast Cancer Patients | breast cancer | Metformin (NPC230775) | |
| NCT04428086 | A Pharmacokinetic Interaction Study Between Apatinib and Rosuvastatin or Metformin in Solid Tumor Subjects. | neoplasm | Metformin (NPC230775) | |
| NCT01046500 | Myo-inositol Versus Metformin Administration in Post-menopausal Women With Metabolic Syndrome | metabolic syndrome | Metformin (NPC230775) | |
| NCT03490019 | Treatment of Bardet-Biedl-Syndrome With Metformin for Evaluation of a Possible Visual Improvement | Bardet-Biedl syndrome;blindness (disorder) | Metformin (NPC230775) | |
| NCT04416269 | Oral Anti Diabetic Agents in the Hospital | diabetes mellitus | Metformin (NPC230775) | |
| NCT03477162 | Metformin Pharmacology in Human Cancers: A Proof of Principle Study | neoplasm of thorax | Metformin (NPC230775) | |
| NCT02990728 | Mirena® ± Metformin as Fertility-preserving Treatment for Young Asian Women With Early Endometrial Cancer | endometrial cancer | Metformin (NPC230775) | |
| NCT02640534 | Investigation of Metformin in Patients With Castration Resistant Prostate Cancer in Combination With Enzalutamide vs. Enzalutamide Alone | prostate carcinoma | Metformin (NPC230775) | |
| NCT00103857 | MK0431 (Sitagliptin) and Metformin Co-Administration Factorial Study in Patients With Type 2 Diabetes Mellitus (0431-036) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00707031 | GLP-1 Receptor Agonist Lixisenatide Versus Exenatide in Patients With Type 2 Diabetes for Glycemic Control and Safety Evaluation, on Top of Metformin | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00941382 | Sibutramine-metformin Combination Versus Sibutramine and Metformin Monotherapy in Obese Patients | obesity | Metformin (NPC230775) | |
| NCT00820573 | Mechanisms of Glucose Lowering Effects of Sitagliptin and Metformin Alone and in Combination in Patients With T2DM | diabetes mellitus | Metformin (NPC230775) | |
| NCT02402348 | Pilot Study of Metformin in HNSCC to Investigate the Effects of MF, Tumor Genotype and MF-genotype Interactions, on Tumor Metabolism and Anoikis | head and neck malignant neoplasia | Metformin (NPC230775) | |
| NCT01483560 | REducing With MetfOrmin Vascular Adverse Lesions in Type 1 Diabetes (REMOVAL) | type 1 diabetes mellitus | Metformin (NPC230775) | |
| NCT02587741 | Comparison of Diabetes Retinopathy Among Type 2 Diabetic Patients Treated With Different Regimens | diabetic retinopathy | Metformin (NPC230775) | |
| NCT01042379 | I-SPY TRIAL: Neoadjuvant and Personalized Adaptive Novel Agents to Treat Breast Cancer | angiosarcoma;triple-negative breast cancer | Metformin (NPC230775) | |
| NCT02028221 | Phase II Study of Metformin for Reduction of Obesity-Associated Breast Cancer Risk | breast cancer | Metformin (NPC230775) | |
| NCT00344851 | Exenatide and Metformin Therapy in Overweight Women With PCOS | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT01204775 | Study to Evaluate the Efficacy, Safety, Tolerability, and Pharmacokinetics of Saxagliptin as Monotherapy in Pediatric Patients With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT04615351 | A Pragmatic Approach to Lowering the Risk of Diabetes Mellitus After A Diagnosis of Gestational Diabetes Mellitus | gestational diabetes | Metformin (NPC230775) | |
| NCT00673465 | Effect of SCH 497079 on Metabolic Parameters and Influence of Race/Ethnic Origin on Therapeutic Response (Study P05338)(COMPLETED) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01089166 | Study to Compare the Bioavailability of Extended Release Metformin HCl Tablet Versus Glucophage XR® Tablets in Healthy Human Volunteers Under Fasting Condition | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02844803 | Study of Metformin and S. Baicalensis Combination Therapy in Type 2 Diabetes Mellitus Patients | diabetes mellitus | Metformin (NPC230775) | |
| NCT03824002 | Dulaglutide in Diabetic Patients, Relationship Between Arterial Stiffness, Endothelial Function, Clinical and Laboratory Variables | diabetes mellitus | Metformin (NPC230775) | |
| NCT01589367 | Neoadjuvant Letrozole Plus Metformin vs Letrozole Plus Placebo for ER-positive Postmenopausal Breast Cancer | breast cancer | Metformin (NPC230775) | |
| NCT04511416 | Preventing Cognitive Decline With Metformin | Mental deterioration | Metformin (NPC230775) | |
| NCT00571519 | Randomized, Double-blind, Active-controlled, Study of Rivoglitazone in Type 2 Diabetes Mellitus | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT04792749 | Clinical Effects of Metformin on Fertility-sparing Treatment for Early Endometrial Cancer | endometrial cancer | Metformin (NPC230775) | |
| NCT01137812 | The CANTATA-D2 Trial (CANagliflozin Treatment And Trial Analysis - DPP-4 Inhibitor Second Comparator Trial) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00960661 | A Trial Comparing Two Therapies: Basal Insulin/Glargine, Exenatide and Metformin Therapy (BET) or Basal Insulin/Glargine, Bolus Insulin Lispro and Metformin Therapy (BBT) in Subjects With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00614120 | Effect of Liraglutide or Glimepiride Added to Metformin on Blood Glucose Control in Subjects With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01245166 | A Phase III Randomized, Double-blind, Parallel-group Study to Evaluate the Efficacy and Safety of Acarmet (Metformin HCl 500 mg Plus Acarbose 50 mg Tablets) Versus Acarbose Alone in Subjects With Type 2 Diabetes Mellitus | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT03054519 | Improve PAD PERformance With METformin | peripheral arterial disease | Metformin (NPC230775) | |
| NCT01677923 | Obesity in Children and Adolescents: Associated Risks and Early Intervention | obesity | Metformin (NPC230775) | |
| NCT00184574 | Comparison of Biphasic Insulin Aspart 70/30, 50/50, and 30/70 in Subjects With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT03008551 | Empagliflozin vs Metformin in PCOS | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT01816659 | An Open-Labeled Pilot Study of Biomarker Response Following Short-Term Exposure to Metformin | colorectal carcinoma | Metformin (NPC230775) | |
| NCT03994744 | Assessing Safety and Efficacy of Sintilimab and Metformin Combination Therapy in SCLC | small cell lung carcinoma | Metformin (NPC230775) | |
| NCT01370707 | A Study to Evaluate the Efficacy and Safety of CJ-30001 and CJ-30002 in Type 2 Diabetes Mellitus Patients | diabetes mellitus | Metformin (NPC230775) | |
| NCT03243851 | Study on Low Dose Temozolomide Plus Metformin or Placebo in Patient With Recurrent or Refractory Glioblastoma | glioblastoma multiforme | Metformin (NPC230775) | |
| NCT00620191 | Metformin in Amnestic Mild Cognitive Impairment | Cognitive impairment | Metformin (NPC230775) | |
| NCT02040376 | Metformin for Brain Repair in Children With Cranial-Spinal Radiation for Medulloblastoma | brain neoplasm | Metformin (NPC230775) | |
| NCT01864681 | Combination of Metformin With Gefitinib to Treat NSCLC | non-small cell lung carcinoma | Metformin (NPC230775) | |
| NCT02437812 | Study of Paclitaxel, Carboplatin and Oral Metformin in the Treatment of Advanced Stage Ovarian Carcinoma | ovarian carcinoma | Metformin (NPC230775) | |
| NCT01210911 | Metformin Combined With Chemotherapy for Pancreatic Cancer | pancreatic carcinoma | Metformin (NPC230775) | |
| NCT04817787 | Exercise Dose and Metformin for Vascular Health in Adults With Metabolic Syndrome | metabolic syndrome | Metformin (NPC230775) | |
| NCT00466622 | Uterine Artery Blood Flow in Pregnant Women With PCOS Treated With Metformin | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT00081328 | Treatment Options for Type 2 Diabetes in Adolescents and Youth (TODAY) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01106677 | The CANTATA-D Trial (CANagliflozin Treatment and Trial Analysis - DPP-4 Inhibitor Comparator Trial) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00814372 | Evaluate Safety and Effectiveness of MBX-102 in Type 2 Diabetes Patients With Poor Glycemic Control on Metformin | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT05183204 | Paxalisib With a High Fat, Low Carb Diet and Metformin for Glioblastoma | glioblastoma multiforme | Metformin (NPC230775) | |
| NCT02506777 | Neoadjuvant FDC With Melatonin or Metformin for Locally Advanced Breast Cancer. | breast cancer | Metformin (NPC230775) | |
| NCT00069836 | Study Of AVANDAMET® With Or Without Insulin In Type II Diabetes Mellitus Patients. AVANDAMET® is a Registered Trademark of the GSK Group of Companies. | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01217073 | A Dose-Range Finding Study in Participants With Type 2 Diabetes (MK-3102-006) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00611884 | Comparison of Two NN1250 Formulations Versus Insulin Glargine, All in Combination With Metformin in Subjects With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01464437 | AMG 151 Amgen Protocol Number 20100761 | diabetes mellitus | Metformin (NPC230775) | |
| NCT01890122 | Efficacy and Safety of Alogliptin and Metformin Fixed-Dose Combination in Participants With Type 2 Diabetes | diabetes mellitus | Metformin (NPC230775) | |
| NCT00376038 | Drug Interactions From Simultaneous Administration Of Metformin And GSK189075 To Subjects With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00838903 | Efficacy and Safety of Albiglutide in Treatment of Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02115464 | Advanced Lung Cancer Treatment With Metformin and Chemo-Radiotherapy | lung cancer | Metformin (NPC230775) | |
| NCT03379909 | Phase II Study of Oral Metformin for Intravesical Treatment of Non-muscle-invasive Bladder Cancer | urinary bladder cancer | Metformin (NPC230775) | |
| NCT04500756 | Limiting AAA With Metformin (LIMIT) Trial | Abdominal Aortic Aneurysm | Metformin (NPC230775) | |
| NCT00316082 | Study of BMS-477118 as Monotherapy With Titration in Subjects With Type 2 Diabetes Who Are Not Controlled With Diet and Exercise | diabetes mellitus | Metformin (NPC230775) | |
| NCT03651531 | Comparison of Insulin Alone to Insulin With Metformin to Treat Gestational Diabetes Mellitus | gestational diabetes | Metformin (NPC230775) | |
| NCT04088448 | The Antidiabetic Metformin as a Novel Adjunct to Antidepressants in Major Depressive Disorder Patients | major depressive disorder | Metformin (NPC230775) | |
| NCT00263965 | ARAMIS: Actions of tesaglitazaR on fAt Metabolism and Insulin Sensitivity | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00762957 | Safety and Efficacy of TAK-559 in Combination With Metformin in Patients With Type 2 Diabetes Mellitus. | diabetes mellitus | Metformin (NPC230775) | |
| NCT03465345 | Study of Metformin Plus Oligomeric Procyanidin Complex for Pharmacologic Manipulation of AGE (Advanced Glycation Endproducts) Levels in Prostate Cancer Patients | prostate cancer | Metformin (NPC230775) | |
| NCT01095653 | A Phase III Study of BMS-512148 (Dapagliflozin) in Asian Patients With Type 2 Diabetes Who Are Not Well Controlled With Diet and Exercise | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00605475 | Safety, Tolerability, Pharmacokinetics and Pharmacodynamics of ACZ885 in Patients With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01561482 | Study of Metformin With Simvastatin for Men With Prostate Carcinoma | prostate carcinoma | Metformin (NPC230775) | |
| NCT01528046 | Metformin in Children With Relapsed or Refractory Solid Tumors | brain neoplasm | Metformin (NPC230775) | |
| NCT00469586 | Efficacy and Safety of Inhaled Insulin Compared to Metformin and Glimepiride in Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02190838 | Comparison of Melatonin or Metformin and Dacarbazine Combination Versus Dacarbazine Alone in Disseminated Melanoma | melanoma | Metformin (NPC230775) | |
| NCT01302002 | The Use of Metformin in Early Breast Cancer Patients Pre-Surgery | breast cancer | Metformin (NPC230775) | |
| NCT00856986 | The Effect of Insulin Detemir in Combination With Liraglutide and Metformin Compared to Liraglutide and Metformin in Subjects With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02457286 | Improving Insulin Resistance To Treat Non-Alcoholic Fatty Liver Disease: A Pilot Study | non-alcoholic fatty liver disease | Metformin (NPC230775) | |
| NCT01352026 | Evaluation of Metformin Activity in Addition to Conventional Treatment of Grade II or III Pulmonary Arterial Hypertension (PAH) | pulmonary arterial hypertension | Metformin (NPC230775) | |
| NCT00479466 | Dose-Range Finding Study for MK0893 (0893-008) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT04224051 | Metformin for Abdominal Aortic Aneurysm Growth Inhibition | Abdominal Aortic Aneurysm | Metformin (NPC230775) | |
| NCT02019979 | Metformin and Carbohydrate Restriction With Platinum Based Chemotherapy In Stage IIIB/IV Non-Squamous Non-Small Cell Lung Cancer (NS-NSCLC) | non-small cell lung carcinoma | Metformin (NPC230775) | |
| NCT05053841 | Implication of Metformin Plus Aromatase Inhibitor in Obese Postmenopausal Breast Cancer Women | breast cancer | Metformin (NPC230775) | |
| NCT05093959 | Metformin for Older Patients With Heart Failure With Preserved Ejection Fraction Pilot Study | heart failure | Metformin (NPC230775) | |
| NCT03204058 | Locally Delivered Metformin & Rosuvastatin for Treatment of Intrabony Defects in Chronic Periodontitis | periodontitis | Metformin (NPC230775) | |
| NCT01167738 | Combination Chemotherapy With or Without Metformin Hydrochloride in Treating Patients With Metastatic Pancreatic Cancer | pancreatic carcinoma | Metformin (NPC230775) | |
| NCT00899470 | Bioequivalence Study of Saxagliptin and Glucophage Combination Formulations in Healthy Subjects (A) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT03017833 | Sapanisertib and Metformin in Treating Patients With Advanced or Metastatic Relapsed or Refractory Cancers | neoplasm | Metformin (NPC230775) | |
| NCT00961480 | A Study to Demonstrate the Bioequivalence of Sitagliptin/Metformin Combination Tablets and Co-administration of Sitagliptin and Metformin as Individual Tablets (0431A-095) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02394652 | The Potential for Metformin to Improve Tumor Oxygenation in Locally Advanced Cervix Cancer: A Phase II Randomized Trial | cervical adenocarcinoma;squamous cell carcinoma | Metformin (NPC230775) | |
| NCT00332488 | Efficacy and Safety of Prandial Inhalation of Technosphere/Insulin in Combination With Metformin or Technosphere/Insulin Alone Versus 2 Oral Anti-Diabetic Agents in Subjects With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00789191 | Effect of Detemir and Sitagliptin on Blood Glucose Control in Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00684528 | A Double-Blind, Randomized, Active-Comparator (Metformin) Controlled, Clinical Trial to Study the Efficacy and Safety of the Strategy to Start Patients With Type 2 Diabetes Mellitus on Janumet™ Compared to Metformin | diabetes mellitus | Metformin (NPC230775) | |
| NCT02848508 | Metformin in Moderate and Severe Renal Failure (CKD 3-4): A Follow-up Study | chronic kidney disease | Metformin (NPC230775) | |
| NCT01051011 | A Study to Compare Taspoglutide and Insulin Glargine in Insulin-Naïve Patients With Type 2 Diabetes Mellitus Inadequately Controlled on Metformin and Sulfonylurea Combination Therapy | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01211197 | Bioavailability of a Fixed Dose Combination Tablet With Empagliflozin (BI 10773) and Metformin Compared With the Monocomponents and Effect of Food on Bioavailability | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02308228 | Metformin to Augment Strength Training Effective Response in Seniors (MASTERS) | aging | Metformin (NPC230775) | |
| NCT01996696 | Prevention of Metabolic Syndrome and Increased Weight Using Metformin Concurrent to Androgen Deprivation Therapy and Radiotherapy for Locally Advanced Adenocarcinoma of the Prostate | prostate adenocarcinoma | Metformin (NPC230775) | |
| NCT02778776 | Combination of Metformine/Inulin Versus Metformin on Prostate Benign Hyperplasia in Metabolic Syndrome | metabolic syndrome | Metformin (NPC230775) | |
| NCT02696941 | SGLT2 Inhibitors and Metformin on Metabolism and Non-Alcoholic SteatoHepatitis | non-alcoholic fatty liver disease | Metformin (NPC230775) | |
| NCT00755287 | A Study of the Safety, Tolerability and Effect on Glycemic Control of Taspoglutide Versus Insulin Glargine in Insulin Naive Type 2 Diabetic Patients Inadequately Controlled With Metformin Plus Sulphonylurea. | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01442870 | Evaluation of Clinical Safety of Combining Metformin With Anticancer Chemotherapy | cancer | Metformin (NPC230775) | |
| NCT03848533 | Effect of Melatonin and Metformin on Glycemic Control Genotoxicity and Cytotoxicity Markers in Patients With Prediabetes | prediabetes syndrome | Metformin (NPC230775) | |
| NCT02461667 | Alendronate Compared to Metformin in Chronic Periodontitis | periodontitis | Metformin (NPC230775) | |
| NCT01093794 | Bioequivalence Study of Sitagliptin/Metformin Combination Tablet (MK0431A-122) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00698230 | Safety and Efficacy of INCB013739 Plus Metformin Compared to Metformin Alone on Glycemic Control in Type 2 Diabetics | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT03006172 | To Evaluate the Safety, Tolerability, and Pharmacokinetics of Inavolisib Single Agent in Participants With Solid Tumors and in Combination With Endocrine and Targeted Therapies in Participants With Breast Cancer | breast cancer | Metformin (NPC230775) | |
| NCT03056274 | Metformin in Intrahepatic Cholestasis of Pregnancy | pregnancy | Metformin (NPC230775) | |
| NCT00337610 | Sitagliptin Metformin Add-on Study in Patients With Type 2 Diabetes Mellitus | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01192139 | Bioequivalence Study of the Fixed Dose Combination of 5 mg Saxagliptin and 500 mg Metformin XR Tablet (Manufactured in Mt Vernon, IN) Relative to 5 mg Saxagliptin Tablet and 500 mg Metformin XR Tablet (Manufactured in Evansville, IN) | diabetes mellitus | Metformin (NPC230775) | |
| NCT00525330 | A Randomized, Double-Blind, Placebo-Controlled Study to Assess the Safety and Efficacy of KRP-104 in Patients With Type 2 Diabetes Inadequately Controlled on Metformin Alone | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT04209075 | Prebiotics and Metformin Improve Gut and Hormones in Type 2 Diabetes in Youth (MIGHTY-fiber) | diabetes mellitus | Metformin (NPC230775) | |
| NCT00349635 | A Controlled-Study on the Effect of a Combination of Fenofibrate and Metformin on Weight Loss and Weight Loss-Maintenance, in Obese Patients. | obesity | Metformin (NPC230775) | |
| NCT01089192 | Bioequivalence Study of Metformin Hydrochloride Extended Release 750 mg Tablets Versus Glucophage XR® 750 mg Tablet in Healthy Volunteers Under Fasting Conditions. | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT03053544 | Metformin With Neoadjuvant Chemoradiation to Improve Pathologic Responses in Rectal Cancer | rectum cancer | Metformin (NPC230775) | |
| NCT05373680 | Efficacy and Safety of Metformin Versus Empagliflozin on Chronic Kidney Disease Progression | chronic kidney disease | Metformin (NPC230775) | |
| NCT00374907 | A Study Assessing Saxagliptin Treatment in Subjects With Type 2 Diabetes Who Are Not Controlled With Diet and Exercise | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01106625 | The CANTATA-MSU Trial (CANagliflozin Treatment And Trial Analysis - Metformin and SUlphonylurea) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT03117517 | Treatment With Metformin and Combination of Metformin and Pioglitazone in Polycystic Ovarian Syndrome | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT01622257 | Cavitation Ultrasound in Treatment of Patients With PCOS | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT00767000 | Dose Range Finding Study of MK-0941 in Patients With Type 2 Diabetes Mellitus on Insulin (MK-0941-007 AM3 EXT1 AM1)(TERMINATED) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00881725 | A Study of Pre-operative Metformin in Prostate Cancer | prostate cancer | Metformin (NPC230775) | |
| NCT03764605 | Metformin vs Tolvaptan for Treatment of Autosomal Dominant Polycystic Kidney Disease | Autosomal dominant polycystic kidney disease | Metformin (NPC230775) | |
| NCT04196868 | Methotrexate and Metformin in Rheumatoid Arthritis Patients | rheumatoid arthritis | Metformin (NPC230775) | |
| NCT03086005 | Study to Assess the Effect of Metformin Supplementation on IVF Outcome in Patients With Polycystic Ovarian Syndrome. | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT00099866 | Efficacy and Safety of Vildagliptin Compared to Metformin in Drug Naive Patients With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01796028 | Metformin-Docetaxel Association in Metastatic Hormone-refractory Prostate Cancer | prostate adenocarcinoma | Metformin (NPC230775) | |
| NCT02516085 | Improved Muscle Function in Duchenne Muscular Dystrophy Through L-Arginine and Metformin | Duchenne muscular dystrophy | Metformin (NPC230775) | |
| NCT04264897 | Antecedent Metabolic Health and Metformin Aging Study | aging;insulin resistance | Metformin (NPC230775) | |
| NCT00699036 | Rosiglitazone Versus Rosiglitazone and Metformin Versus Rosiglitazone and Avandia in the Treatment of Nonalcoholic Steatohepatitis (NASH) | non-alcoholic steatohepatitis | Metformin (NPC230775) | |
| NCT00885352 | Sitagliptin (MK-0431) vs. Placebo in Patients With Inadequate Glycemic Control on Metformin With Pioglitazone (MK-0431-128) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01243385 | Metformin Hydrochloride as First-Line Therapy in Treating Patients With Locally Advanced or Metastatic Prostate Cancer | prostate cancer | Metformin (NPC230775) | |
| NCT00617240 | Strategies to Reduce Antipsychotic-Associated Weight Gain in Youth | body weight gain | Metformin (NPC230775) | |
| NCT00383578 | Efficacy and Safety of Vildagliptin Compared to Metformin in Elderly Drug Naive Patients With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02773927 | Combination of Metformin/Inulin vs Inulin on Adiponectin in Metabolic Syndrome | metabolic syndrome | Metformin (NPC230775) | |
| NCT02472353 | Use of Metformin to Reduce Cardiac Toxicity in Breast Cancer | breast cancer | Metformin (NPC230775) | |
| NCT00900146 | Dose Finding, Safety and Efficacy of Monthly Subcutaneous Canakinumab Administration in Metformin Monotherapy Treated Type 2 Diabetic Patients | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00266253 | A Study of GK Activator (2) in Patients With Type 2 Diabetes Mellitus Treated With a Stable Dose of Metformin | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01117584 | A Study to Evaluate the Effect of ASP1941 in Combination With Metformin in Adult Patients With Type 2 Diabetes Mellitus | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00918138 | Study To Evaluate 24 Hour Blood Sugar Control (24-hour Mean Weighted Glucose) In Subjects That Are Taking Saxagliptin 5 mg Added Onto Metformin XR 1500 XR mg Compared To Subjects Taking Metformin XR 1500 mg Up-titrated To Metformin XR 2000 mg | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00481663 | A Study of Different Doses of Sitagliptin (MK-0431) in Participants With Type 2 Diabetes Mellitus (MK-0431-014) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00086502 | Pioglitazone Add-on Study in Patients With Type 2 Diabetes Mellitus | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00348712 | Efficacy and Safety of Inhaled Pre-prandial Human Insulin in Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00839527 | A Study to Determine the Safety and Efficacy of Albiglutide in Subjects With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02498522 | Effect of Metformin on Early Pregnancy Loss in Pregnant Women With Polycystic Ovarian Syndrome | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT00268476 | Systemic Therapy in Advancing or Metastatic Prostate Cancer: Evaluation of Drug Efficacy | prostate cancer | Metformin (NPC230775) | |
| NCT02143050 | Study of Dabrafenib, Trametinib and Metformin for Melanoma Patients | melanoma | Metformin (NPC230775) | |
| NCT03722290 | Metformin in Children and Adults With Fragile X Syndrome | fragile X syndrome | Metformin (NPC230775) | |
| NCT00359112 | AVANDAMET Versus Metformin And Sulphonylurea In People With Poorly Controlled Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01465113 | Vitamin D Supplementation on 15-Prostaglandin Dehydrogenase Expression in Barrett's Esophagus | Barrett's esophagus | Metformin (NPC230775) | |
| NCT05305898 | "MetCool ACS"- Metformin "Cooling" Effect on Metformin-naive Patients Treated With PCI Because of Acute Coronary Syndrome | acute coronary syndrome | Metformin (NPC230775) | |
| NCT00375388 | The HOME Trial: Hyperinsulinaemia: the Outcome of Its Metabolic Effects, a Randomized Controlled Trial | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00097279 | Comparison of Biphasic Insulin Aspart 70/30 With Anti-Diabetic Drugs in Subjects With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT04581447 | Extending Time Without Diabetes After Bariatric Surgery: a Trial Comparing the Metformin Addition or Not to Standard Care | diabetes mellitus | Metformin (NPC230775) | |
| NCT02312661 | Study of Metformin With Carboplatin/Paclitaxel Chemotherapy in Patients With Advanced Ovarian Cancer | ovarian cancer | Metformin (NPC230775) | |
| NCT04530058 | The Effects of Metformin on Morbidity and Mortality in Elderly Patients | burn | Metformin (NPC230775) | |
| NCT00067951 | A Study To Evaluate The Safety And Efficacy Of An Investigational Diabetes Drug In Poorly Controlled Type II Diabetics | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02484209 | DiaFrail: A Short Duration Study in Older People ( DIAFRAIL Study) | diabetes mellitus | Metformin (NPC230775) | |
| NCT03674385 | Efficacy of Vit E in PCOS Resistant to Clomiphene Citrate | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT00627445 | Effect of Biphasic Insulin Aspart 50 on Blood Glucose Control in Subjects With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00544518 | Impact of Benfluorex Versus Metformin on Glucose Control and Insulin Secretion in Chinese Type 2 Diabetic Patients | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT03258723 | Diabetes Prevention With Lifestyle Intervention and Metformin Escalation | diabetes mellitus | Metformin (NPC230775) | |
| NCT00361868 | Fenofibrate and Metformin Fixed Combination vs Rosiglitazone - FAME ROSI | Disorder of lipid metabolism | Metformin (NPC230775) | |
| NCT04220021 | Safety and Therapeutic Potential of the FDA-approved Drug Metformin for C9orf72 ALS/FTD | frontotemporal dementia | Metformin (NPC230775) | |
| NCT00631033 | DZX Mediated Insulin Suppression in Obese Men | obesity | Metformin (NPC230775) | |
| NCT01750567 | A Pilot Study of Metformin Therapy in Patients With Relapsed Chronic Lymphocytic Leukemia (CLL) and Untreated CLL | chronic lymphocytic leukemia | Metformin (NPC230775) | |
| NCT00770445 | Efficacy of Pioglitazone and Metformin on Cardiovascular Risk in Subjects With Insulin-Treated Type 2 Diabetes Mellitus. | diabetes mellitus | Metformin (NPC230775) | |
| NCT03108963 | L-carnitine and Metformin in Obese PCOS Women. | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT00712673 | GLP-1 Receptor Agonist Lixisenatide (Morning or Evening) in Patients With Type 2 Diabetes for Glycemic Control and Safety Evaluation, on Top of Metformin | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02903511 | Feasibility Study of Metformin Therapy in ADPKD | Autosomal dominant polycystic kidney disease | Metformin (NPC230775) | |
| NCT00789035 | 12 Weeks Treatment With 3 Different Doses of BI 10773 in Type 2 Diabetic Patients | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01717482 | Metformin as a Chemoprevention Agent in Non-small Cell Lung Cancer | non-small cell lung carcinoma | Metformin (NPC230775) | |
| NCT00700817 | The Effect of Liraglutide Compared to Sitagliptin, Both in Combination With Metformin on Glycaemic Control in Subjects With Type 2 Diabetes Mellitus | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00743002 | A Trial of TT223 in Patients With Type 2 Diabetes Who Are Taking Metformin and/or Thiazolidinedione | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01095666 | A Phase III Study of BMS-512148 (Dapagliflozin) in Asian Patients With Type 2 Diabetes Who Are Not Well Controlled on Metformin Alone | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01154348 | Study to Assess Safety and Pharmacokinetics of Double-Blind S-707106 Alone and in Combination With Open-Label Metformin in Patients With Type 2 Diabetes Mellitus | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT04867252 | Effects of Combined Resveratrol and Myo-inositol on Altered Metabolic, Endocrine Parameters and Perceived Stress in Patients With Polycystic Ovarian Syndrome | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT00809536 | Investigation Of How PD 0332334 And Metformin Are Eliminated From The Body When They Are Administered At The Same Time | generalized anxiety disorder | Metformin (NPC230775) | |
| NCT00295633 | A Study Assessing Saxagliptin Treatment in Type 2 Diabetic Subjects Who Are Not Controlled With TZD Therapy Alone | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00063232 | Treating Nonalcoholic Steatohepatitis (NASH) With Metformin | Hepatitis | Metformin (NPC230775) | |
| NCT00631488 | A Study to Test the Effectiveness and Safety of MK0893 in Combination With Other Drugs Used to Treat Type 2 Diabetes (0893-015) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT04750135 | Assessment of Metformin as Adjuvant Therapy in Patients With Ulcerative Colitis | ulcerative colitis | Metformin (NPC230775) | |
| NCT00348725 | Acceptability of a Fixed Combination of Fenofibrate and Metformin | metabolic disease | Metformin (NPC230775) | |
| NCT00875394 | Study to Assess the Efficacy and Safety of Sitagliptin Added to the Regimen of Patients With Type 2 Diabetes Mellitus With Inadequate Glycemic Control on Metformin (0431-189) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01302145 | Drug to Drug Interaction Study With ASP1941 and Metformin | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02819869 | The Combination Effect of Statin Plus Metformin on Relapse-free | hepatocellular carcinoma | Metformin (NPC230775) | |
| NCT05440591 | Effects of Dapagliflozin and Metformin on Vascular Function in Newly-Diagnosed Treatment-Naive Type 2 Diabetes | diabetes mellitus | Metformin (NPC230775) | |
| NCT01292993 | A Study to Evaluate the Interaction of LX4211 and Metformin in Healthy Subjects | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02719756 | Effect of SGLT-2 Inhibitor Dapagliflozin on Glycemic Variability in Patients With Diabetes Mellitus Type 2 | diabetes mellitus | Metformin (NPC230775) | |
| NCT01334125 | Adjunctive Metformin Therapy in Double Diabetes | diabetes mellitus | Metformin (NPC230775) | |
| NCT00102466 | Vildagliptin Compared to Gliclazide in Combination With Metformin in Patients With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00111631 | A Study of DPP-IV Inhibitor in Patients With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT03833466 | Metformin in Combined With Cisplatin Plus Paclitaxel With Advanced Esophageal Squamous Cell Carcinoma (ECMTPneo) | esophageal squamous cell carcinoma | Metformin (NPC230775) | |
| NCT00860288 | Efficacy and Long-Term Safety of Vildagliptin as Add-on Therapy to Metformin in Patients With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02372656 | Simvastatin and Metformin in Chronic Periodontitis | periodontitis | Metformin (NPC230775) | |
| NCT01578551 | Study of Metformin Plus Paclitaxel/Carboplatin/Bevacizumab in Patients With Adenocarcinoma. | lung adenocarcinoma | Metformin (NPC230775) | |
| NCT02254512 | Metformin and Carbohydrate Restriction With Platinum Based Chemotherapy In Stage IIIB/IV Non-Squamous Non-Small Cell Lung Cancer (NS-NSCLC) | carcinoma | Metformin (NPC230775) | |
| NCT02285855 | Metformin in Non Small Cell Lung Cancer (NSCLC) | lung cancer | Metformin (NPC230775) | |
| NCT00468039 | A Sub-study to LMF237A2302 to Assess the Effect of 24 Weeks Treatment With Initial Combination of Vildagliptin Plus Metformin | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02810132 | Effects of Metformin Treatment on Myocardial Efficiency in Patients With Heart Failure | systolic heart failure | Metformin (NPC230775) | |
| NCT04926155 | The Effect of Metformin in Patients With Metastatic Castration-resistant Prostate Cancer | metastatic prostate cancer | Metformin (NPC230775) | |
| NCT00279045 | Diabetes Study With Rosiglitazone Monotherapy Versus Metformin Or Glyburide/Glibenclamide | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00969566 | Predictive Parameters for Efficacy of Sitagliptin and Metformin Combination | diabetes mellitus | Metformin (NPC230775) | |
| NCT02283554 | Platelet Rich Fibrin With 1% Metformin Gel for Treatment of Intrabony Defects | periodontitis | Metformin (NPC230775) | |
| NCT00097500 | Effects of Exenatide and Insulin Glargine in Subjects With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT04767841 | The AMPK Modulator Metformin as a Novel Adjunct to Conventional Therapy in Patients With Knee Osteoarthritis | osteoarthritis | Metformin (NPC230775) | |
| NCT05236530 | Drug-drug Interaction Study of Ganaplacide and Lumefantrine With Midazolam, Repaglinide, Dextromethorphan, Metformin, Rosuvastatin and Dolutegravir | malaria | Metformin (NPC230775) | |
| NCT02519543 | Treating Insulin Resistance as a Strategy to Improve Outcome in Refractory Bipolar Disorder | bipolar disorder | Metformin (NPC230775) | |
| NCT00541775 | Safety/Efficacy of Sitagliptin in Patient w/ Type 2 Diabetes (0431-801) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01472367 | A Pooled Analysis of the Safety and Efficacy of MK-0431A and MK-0431A XR in Pediatric Participants With Type 2 Diabetes Mellitus With Inadequate Glycemic Control on Metformin Therapy (Alone or in Combination With Insulin) (MK-0431A-170/MK-0431A-289) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00892073 | Hypothalamic Obesity Following Craniopharyngioma Surgery: A Pilot Trial of Combined Metformin and Diazoxide Therapy | obesity | Metformin (NPC230775) | |
| NCT00432276 | Efficacy of Alogliptin and Pioglitazone in Subjects With Type 2 Diabetes Mellitus | diabetes mellitus | Metformin (NPC230775) | |
| NCT00754403 | Efficacy of Pioglitazone and Fortamet Combination Therapy in Subjects With Type 2 Diabetes | diabetes mellitus | Metformin (NPC230775) | |
| NCT00286442 | Efficacy and Safety of Alogliptin Combined With Metformin in Participants With Type 2 Diabetes Mellitus | diabetes mellitus | Metformin (NPC230775) | |
| NCT03378297 | IMPACT: A Randomized WOO Study of Novel Therapeutic Agents in Women Triaged to Primary Surgery for EOC | ovarian cancer | Metformin (NPC230775) | |
| NCT02042495 | Endometrial Cancer Biomarker Changes Following Exposure to Metformin | endometrial cancer | Metformin (NPC230775) | |
| NCT01084486 | Effect of Treatment With Insulin Sensitizer on Arterial Properties, Metabolic Parameters and Liver Function in Patients With Nonalcoholic Fatty Liver Disease | non-alcoholic fatty liver disease | Metformin (NPC230775) | |
| NCT02274090 | 1% Metformin in Moderate and Severe Periodontitis | periodontitis | Metformin (NPC230775) | |
| NCT02614339 | Effect of Adjunctive Metformin on Recurrence of Non-DM Colorectal Cancer Stage II High-risk/ III Colorectal Cancer | colorectal carcinoma | Metformin (NPC230775) | |
| NCT01710527 | BE Study of Metformin GSK 500mg | diabetic foot | Metformin (NPC230775) | |
| NCT01725490 | The Chemopreventive Effect of Metformin in Patients With Familial Adenomatous Polyposis: Double Blinded Randomized Controlled Study | Familial adenomatous polyposis | Metformin (NPC230775) | |
| NCT05394142 | A Clinical Trial to Evaluate the Efficacy, Tolerability, and Safety of a Fixed Dose Combination of Spironolactone, Pioglitazone & Metformin (SPIOMET) in Polycystic Ovary Syndrome (PCOS) | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT02473094 | Neoadjuvant Metformin in Association to Chemoradiotherapy for Locally Advanced Rectal Cancer | rectum cancer | Metformin (NPC230775) | |
| NCT03651895 | Metformin to Reduce Airway Glucose in COPD Patients | chronic obstructive pulmonary disease | Metformin (NPC230775) | |
| NCT01126580 | A Study in Participants With Type 2 Diabetes Mellitus (AWARD-3) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02149459 | Treatment of Recurrent Brain Tumors: Metabolic Manipulation Combined With Radiotherapy | brain neoplasm | Metformin (NPC230775) | |
| NCT00617058 | Improving Metabolic Parameters of Antipsychotic Child Treatment With Ziprasidone, Aripiprazole, and Clozapine | body weight gain | Metformin (NPC230775) | |
| NCT01620593 | Castration Compared to Castration Plus Metformin as First Line Treatment for Patients With Advanced Prostate Cancer | prostate cancer | Metformin (NPC230775) | |
| NCT04691960 | A Pilot Study of Ketogenic Diet and Metformin in Glioblastoma: Feasibility and Metabolic Imaging | glioblastoma multiforme | Metformin (NPC230775) | |
| NCT00349128 | Fenofibrate in Dyslipidemia and Metformin-Controlled Diabetes | metabolic disease | Metformin (NPC230775) | |
| NCT04530747 | Metabolic Effects of Metformin Therapy in Obstructive Sleep Apnea | obstructive sleep apnea | Metformin (NPC230775) | |
| NCT02249910 | Investigating the Influence of Oral Semaglutide on Pharmacokinetics of Metformin and Digoxin in Healthy Subjects | diabetes mellitus | Metformin (NPC230775) | |
| NCT00728351 | Efficacy and Safety of Fixed Combination Therapy of Vildagliptin and Metformin (25/1000 mg Bid) in Patients With Type 2 Diabetes Inadequately Controlled With Prior Metformin Monotherapy (HbA1c 7.0-9.5%) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01632020 | Effect of Metformin on Biomarkers of Colorectal Tumor Cell Growth | colorectal neoplasm | Metformin (NPC230775) | |
| NCT02755844 | Safety and Efficacy of Metronomic Cyclophosphamide, Metformin and Olaparib in Endometrial Cancer Patients | endometrial cancer | Metformin (NPC230775) | |
| NCT00362323 | Fenofibrate and Metformin Fixed Combination vs Metformin - FAME METFO | metabolic disease | Metformin (NPC230775) | |
| NCT01076088 | Safety and Efficacy of Co-Administration of Sitagliptin and Metformin in China (MK-0431-121) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01336322 | Metformin and Sitagliptin in Women With Previous Gestational Diabetes | prediabetes syndrome | Metformin (NPC230775) | |
| NCT01911247 | Preoperative Window Study of Metformin for the Treatment of Endometrial Cancer | endometrial cancer | Metformin (NPC230775) | |
| NCT02042664 | Effect of GLP-1 Receptor (GLP-1R) Agonists on Cardiac Function and on Epicardial Adipose Tissue (EAT) Volume and on Myocardial TG Content in Obese Diabetics | diabetes mellitus | Metformin (NPC230775) | |
| NCT02656017 | Metformin as a Novel Therapy for Autosomal Dominant Polycystic Kidney Disease | Autosomal dominant polycystic kidney disease | Metformin (NPC230775) | |
| NCT05131828 | CCMR Two: A Phase IIa, Randomised, Double-blind, Placebo-controlled Trial of the Ability of the Combination of Metformin and Clemastine to Promote Remyelination in People With Relapsing-remitting Multiple Sclerosis Already on Disease-modifying Therapy | multiple sclerosis | Metformin (NPC230775) | |
| NCT01808690 | EMERALD: Effects of Metformin on Cardiovascular Function in Adolescents With Type 1 Diabetes | type 1 diabetes mellitus | Metformin (NPC230775) | |
| NCT01266486 | Effect of Metformin on Breast Cancer Metabolism | breast cancer | Metformin (NPC230775) | |
| NCT01981525 | A Pilot Study of Metformin in Patients With a Diagnosis of Li-Fraumeni Syndrome | Li-Fraumeni syndrome | Metformin (NPC230775) | |
| NCT02026869 | Vaginal Administration of Metformin in PCOS Patients. | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT00698789 | Evaluate the Safety and Tolerability of INCB019602 When Administered With Metformin in Patients With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00528372 | A Phase III Study of BMS-512148 (Dapagliflozin) in Patients With Type 2 Diabetes Who Are Not Well Controlled With Diet and Exercise | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01135433 | A Study to Assess the Efficacy and Safety of ASP1941 in Combination With Metformin in Type 2 Diabetic Patients | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01089205 | Bioequivalence Study of Metformin Hydrochloride Extended Release 750 mg Tablets Versus Glucophage XR® 750 mg Tablet in Healthy Volunteers Under Fed Conditions. | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02360618 | Metformin and Simvastatin Use in Bladder Cancer | urinary bladder cancer | Metformin (NPC230775) | |
| NCT03184493 | Celebrex and Metformin for Postoperative Hepatocellular Carcinoma | liver cancer | Metformin (NPC230775) | |
| NCT01368081 | Empagliflozin (BI 10773) Comprehensive add-on Study in Japanese Subjects With Type 2 Diabetes Mellitus | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00643851 | An Efficacy & Safety Study of BMS-512148 in Combination With Metformin Extended Release Tablets | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00414245 | Metformin for the Treatment of Diabetes in Pregnancy | gestational diabetes;type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT04852510 | Amelioration of Polycystic Ovary Syndrome Related Disorders by Supplementation of Thymoquinone and Metformin | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT00350779 | Sitagliptin Metformin/PPARg Agonist Combination Therapy Add-on (0431-052) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01383356 | Comparison of the Bioavailability of Metformin Between Medium Dose Linagliptin/Metformin Tablets and Medium Dose Glucophage Tablet Given With Linagliptin Tablet | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00086515 | Metformin Add-on Study in Patients With Type 2 Diabetes Mellitus (0431-020)(COMPLETED) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT04113889 | Effects of Triple Drug Cocktail Therapy on Metabolic, Endocrine Alterations and Perceived Stress in Patients With Poly Cystic Ovary Syndrome | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT00661362 | Evaluate Efficacy and Safety of Saxagliptin in Combination With Metformin in Adult Patients With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00959101 | Comparison of Repaglinide and Metformin Combination Tablet Versus Repaglinide and Metformin as Separate Tablets in Healthy Volunteers | diabetes mellitus | Metformin (NPC230775) | |
| NCT00783744 | Insulin Glargine Combination Therapies in Type II Diabetics | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00382096 | Efficacy of Fixed Combination Therapy of Vildagliptin and Metformin Compared to the Individual Monotherapy Components in Drug Naive Patients With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00830076 | A Study of the Effects of Co-Administration of Sitagliptin (MK-0431) and Metformin on Incretin Hormone Concentrations (MK-0431-110) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT04143282 | Evaluation of the Effect of Metformin on Metastatic Breast Cancer as Adjuvant Treatment | breast cancer | Metformin (NPC230775) | |
| NCT03862950 | A Trial of Metformin in Individuals With Fragile X Syndrome (Met) | fragile X syndrome;intellectual disability;Intellectual disability;neurobehavioral manifestations;sex chromosome disorder of sex development | Metformin (NPC230775) | |
| NCT00399711 | Effect of Repaglinide and Metformin Combination Tablet or Rosiglitazone and Metformin in Fixed Dose Combination on Blood Glucose Control in Patients With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00871936 | A Comparison of SLx-4090 in Combination With Metformin Versus Metformin Therapy Alone in Patients With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01477060 | Modulation of Response to Hormonal Therapy With Lapatinib and/or Metformin in Patients With Metastatic Breast Cancer | breast cancer | Metformin (NPC230775) | |
| NCT00032487 | Glycemic Control and Complications in Diabetes Mellitus Type 2 (VADT) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01034111 | A Local Experiential Study With Sitagliptin (an Antihyperglycemic Drug) in 30 Patients With Type 2 Diabetes Mellitus (0431-178) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01107717 | Durability of Combination Therapy With Exenatide/Pioglitazone/Metformin vs. Conventional Therapy in New Onset T2DM | diabetes mellitus | Metformin (NPC230775) | |
| NCT01915550 | Metformin as Adjuvant to Insulin Therapy in Insulin-resistant Diabetes Mellitus With Pregnancy | diabetes mellitus | Metformin (NPC230775) | |
| NCT03179254 | Type 2 Diabetes and Ambulatory Surgery Patients | diabetes mellitus | Metformin (NPC230775) | |
| NCT00971659 | Adjunctive Therapy of Exenatide or Sitagliptin to Insulin Glargine in Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT03861767 | REMAP Trial for Optimizing Surgical Outcomes at UPMC | aging | Metformin (NPC230775) | |
| NCT00106340 | Vildagliptin Compared to Glimepiride in Combination With Metformin in Patients With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00401973 | Assessment of Safety and Efficacy of Therapy for the Prevention of Weight Gain Associated With Olanzapine | schizoaffective disorder;schizophrenia | Metformin (NPC230775) | |
| NCT00318461 | To Compare the Effect of Liraglutide When Given Together With Metformin With the Effect of Metformin Given Alone and With the Effect of Glimepiride and Metformin Given Together | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00975052 | A Study of the Effects of Sitagliptin (MK0431) and Metformin on Incretin Hormone Concentrations (0431-050)(COMPLETED) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT04170959 | The Addition of Metformin to Definitive Radiotherapy in Patients With Stage III NSCLC | non-small cell lung carcinoma | Metformin (NPC230775) | |
| NCT00108615 | Effects of Insulin Sensitizers in Subjects With Impaired Glucose Tolerance | diabetes mellitus | Metformin (NPC230775) | |
| NCT01997775 | Metformin in Stage IV Lung Adenocarcinoma | non-small cell lung carcinoma | Metformin (NPC230775) | |
| NCT02437656 | Combination of Metformin to Neoadjuvant Radiochemotherapy in the Treatment of Locally Advanced Rectal Cancer (METCAP). | rectum cancer | Metformin (NPC230775) | |
| NCT01068717 | Bioequivalence Study of 2.5-mg Saxagliptin and 500-mg Glucophage in Tablets and a Fixed-dose Combination Tablet in Healthy Participants | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01459809 | Efficacy and Safety Comparison of Metformin/Glimepiride Combination Versus Each Compound Alone in New Diagnosed Type 2 Diabetes Patients | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01071850 | A Study to Evaluate the Effect of ASP1941 in Adult Patients With Type 2 Diabetes Mellitus | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT04148183 | Effect of Metformin and Rosiglitazone Over no Diabetic With Metabolic Syndrome Patients. | insulin resistance | Metformin (NPC230775) | |
| NCT00454233 | A Safety and Efficacy Study With YM543 in Type 2 Diabetes Mellitus Subjects | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01627067 | Exemestane-RAD001-Metformin | breast cancer | Metformin (NPC230775) | |
| NCT03681197 | Metformin Use and Clinical Pregnancy Rate in Women With Unexplained Infertility | infertility | Metformin (NPC230775) | |
| NCT00362440 | Combination of Insulin Sensitizer and Leptin as Treatment for the HAART -Induced Metabolic Syndrome | HIV-Associated Lipodystrophy Syndrome | Metformin (NPC230775) | |
| NCT00754988 | A Study of Taspoglutide Versus Sitagliptin for the Treatment of Patients With Type 2 Diabetes Mellitus Inadequately Controlled With Metformin. | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01462266 | Study of Sitagliptin for the Treatment of Type 2 Diabetes Mellitus With Inadequate Glycemic Control on Insulin (MK-0431-260) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01381900 | A Efficacy, Safety, and Tolerability Study of Canagliflozin in Patients With Type 2 Diabetes Mellitus With Inadequate Glycemic Control on Metformin Alone or in Combination With a Sulphonylurea | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00283816 | Impact of Metformin in Teens With Polycystic Ovary Syndrome (PCOS) on Oral Contraceptive Therapy | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT00763451 | GLP-1 Receptor Agonist Lixisenatide in Patients With Type 2 Diabetes for Glycemic Control and Safety Evaluation, on Top of Metformin | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00159536 | Metformin in Pregnant PCOS Women | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT01864096 | The Metformin Active Surveillance Trial (MAST) Study | prostate cancer | Metformin (NPC230775) | |
| NCT00327015 | A Phase 3 Study of BMS-477118 in Combination With Metformin in Subjects With Type 2 Diabetes Who Are Not Controlled With Diet and Exercise | diabetes mellitus | Metformin (NPC230775) | |
| NCT01566799 | Metformin Plus Neoadjuvant Chemotherapy in Breast Cancer | neoplasm | Metformin (NPC230775) | |
| NCT03086733 | Phase II Lung Metcore - Preoperative Metformin for Lung Cancer | non-small cell lung carcinoma | Metformin (NPC230775) | |
| NCT01205672 | Evaluation of the Molecular Effects of Metformin on the Endometrium in Patients With Endometrial Cancer | endometrial cancer | Metformin (NPC230775) | |
| NCT03047837 | A Randomized, 2x2 Factorial Design Biomarker Prevention Trial of Low-dose Aspirin and Metformin in Stage I-III Colorectal Cancer Patients. | malignant colon neoplasm | Metformin (NPC230775) | |
| NCT00313313 | A Study of Saxagliptin in Subjects With Type 2 Diabetes Who Have Inadequate Blood Sugar Control With Sulfonylureas | diabetes mellitus | Metformin (NPC230775) | |
| NCT00715624 | GLP-1 Receptor Agonist Lixisenatide in Patients With Type 2 Diabetes for Glycemic Control and Safety Evaluation, on Top of Basal Insulin | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01488552 | Gemcitabine+Nab-paclitaxel and FOLFIRINOX and Molecular Profiling for Patients With Advanced Pancreatic Cancer | pancreatic carcinoma | Metformin (NPC230775) | |
| NCT00613951 | Comparison of Two NN5401 Formulations Versus Biphasic Insulin Aspart 30, All in Combination With Metformin in Subjects With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00961857 | A Study to Demonstrate the Bioequivalence of Sitagliptin/Metformin Combination Tablets and Concomitant Administration of Sitagliptin and Metformin as Individual Tablets (0431A-048) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00701090 | A Study to Test the Safety and Efficacy of Sitagliptin Compared to Glimepiride in Patients With Type 2 Diabetes on a Stable Dose of Metformin (0431-803)(COMPLETED) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT03479476 | A Trial of Metformin in Individuals With Fragile X Syndrome | fragile X syndrome;Intellectual disability;neurobehavioral manifestations;sex chromosome disorder of sex development | Metformin (NPC230775) | |
| NCT05262257 | Evaluation and Intervention of Cognitive Function in Patients With Diabetes Mellitus. | diabetes mellitus | Metformin (NPC230775) | |
| NCT03889795 | Phase IB Metformin, Digoxin, Simvastatin in Solid Tumors | pancreatic carcinoma | Metformin (NPC230775) | |
| NCT01650506 | Study of Erlotinib and Metformin in Triple Negative Breast Cancer | breast cancer | Metformin (NPC230775) | |
| NCT02815397 | DA-EPOCH-Rituximab/Metformin (RM) for Double Hit Lymphoma | diffuse large B-cell lymphoma | Metformin (NPC230775) | |
| NCT02945813 | SAKK 08/15 - PROMET - Salvage Radiotherapy +/- Metformin for Patients With Prostate Cancer After Prostatectomy | prostate cancer | Metformin (NPC230775) | |
| NCT02946996 | Study of Pharmacologic Manipulation of AGE (Advanced Glycation Endproducts) Levels in Prostate Cancer Patients Receiving Androgen Deprivation Therapy | prostate cancer | Metformin (NPC230775) | |
| NCT00856284 | Efficacy and Safety of Alogliptin Plus Metformin Compared to Glipizide Plus Metformin in Patients With Type 2 Diabetes Mellitus | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00598793 | Biphasic Insulin Aspart 30 in Type 2 Diabetes Failing OAD Therapy | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT04972396 | ALT-801 DDI Study in Healthy Volunteers | non-alcoholic steatohepatitis | Metformin (NPC230775) | |
| NCT01365091 | Bioequivalence Study of Fixed-dose Combinations and Coadministered Individual Tablets of Saxagliptin/Metformin-Brazil | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00744367 | A Study of Taspoglutide Versus Placebo for the Treatment of Patients With Type 2 Diabetes Mellitus Inadequately Controlled With Metformin Plus Pioglitazone. | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00151411 | Lifestyle Intervention and Metformin for Women With Polycystic Ovary Syndrome (PCOS) | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT01076075 | A Study to Evaluate the Safety and Efficacy of Sitagliptin 100 mg in Participants With Type 2 Diabetes Mellitus Who Have Inadequate Glycemic Control (MK-0431-229) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT04973891 | CANA/Met in Non-diabetic Women With PCOS | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT00131664 | Avandia™ + Amaryl™ or Avandamet™ Compared With Metformin (AVALANCHE™ Study) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00063635 | Treatment of Nonalcoholic Fatty Liver Disease in Children (TONIC) | Hepatic steatosis | Metformin (NPC230775) | |
| NCT03713801 | Impact of Metformin on Immunity | aging | Metformin (NPC230775) | |
| NCT01482286 | Effect of Weight and Insulin Sensitivity on Reproductive Function in PCOS | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT01434186 | A Multicenter, Randomized, Double-blind, Placebo-controlled Study to Evaluate the Efficacy and Safety of Saxagliptin (BMS-477118) in Combination With Metformin IR or Metformin XR in Pediatric Patients With Type 2 Diabetes Who Have Inadequate Glycemic Control on Metformin Alone | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02048761 | Locally Delivered 1% Metformin Gel in the Treatment of Intrabony Defects in Chronic Periodontitis | periodontitis | Metformin (NPC230775) | |
| NCT03030300 | Remission Rate of Newly Diagnosed Type 2 Diabetes Outpatients Treated With Short-term Intensive Insulin Therapy | diabetes mellitus | Metformin (NPC230775) | |
| NCT01215331 | Gestational Diabetes: Insulin or Oral Hypoglycemic Agents? | gestational diabetes | Metformin (NPC230775) | |
| NCT02045290 | Insulin Clamp Ancillary Study for Assessment of Insulin Resistance | type 1 diabetes mellitus | Metformin (NPC230775) | |
| NCT00331851 | Effect of Liraglutide on Blood Glucose Control in Subjects With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02088034 | Intervention to Promote Weight Loss in Latinas At-risk for Diabetes | diabetes mellitus | Metformin (NPC230775) | |
| NCT00425009 | Therapeutic Effects of Berberine in Patients With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00792935 | A Study to Test MK-0941 in Patients With Type 2 Diabetes Mellitus With Inadequate Glycemic Control on Metformin (MK-0941-017) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT03905941 | Relative Desirability of Metformin vs. Birth Control Pill in Treating PCOS in Women of Later Reproductive Age | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT02895750 | Efficacity and Safety of Metformin XR in CKD Stage 1 to 3 | chronic kidney disease | Metformin (NPC230775) | |
| NCT04536805 | Relapse in Previously Irradiated Prostate Bed : Stereotactic Ablative Reirradiation Potentiated by Metformin | prostate cancer | Metformin (NPC230775) | |
| NCT03981861 | Metabolic and Neuro-Endocrine Effect of Treating PCOS in Adolescents | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT00121641 | Saxagliptin Treatment in Subjects With Type 2 Diabetes Who Are Not Controlled With Diet and Exercise | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT04050098 | Bioequivalence Study for Gemigliptin/Metformin 50/1000 mg and Coadministration of Gemigliptin 50 mg and Metformin 1000 mg. | diabetes mellitus | Metformin (NPC230775) | |
| NCT00543361 | Study MK0767 and Metformin in Type 2 Diabetic Patients (0767-020) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00798161 | Safety and Efficacy of Linagliptin (BI 1356) Plus Metformin in Type 2 Diabetes, Factorial Design | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01529593 | Temsirolimus in Combination With Metformin in Patients With Advanced Cancers | cancer | Metformin (NPC230775) | |
| NCT01733459 | Efficacy and Safety of DLBS3233 in Subjects With Polycystic Ovary Syndrome (PCOS) | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT02848950 | The Effect of Metformin on Fecundability in an ART(Assisted Reproductive Technology) Setting - the Correlation to Uterine Flows | infertility | Metformin (NPC230775) | |
| NCT04998032 | A Study of Efficacy and Safety of Supaglutide in Type 2 Diabetes Patients On Metformin Treatment | diabetes mellitus | Metformin (NPC230775) | |
| NCT04275713 | Altered Tumor Oxygenation by Metformin, a Potential Step in Overcoming Radiotherapy Resistance in LACC | cervical cancer | Metformin (NPC230775) | |
| NCT03204071 | Locally Delivered Metformin and Aloe Vera for Treatment of Intrabody Defects in Chronic Periodontitis | periodontitis | Metformin (NPC230775) | |
| NCT01440127 | Impact of Pretreatment With Metformin on Colorectal Cancer Stem Cells (CCSC) and Related Pharmacodynamic Markers | malignant colon neoplasm | Metformin (NPC230775) | |
| NCT01995032 | L-citrulline and Metformin in Duchenne's Muscular Dystrophy | muscular dystrophy | Metformin (NPC230775) | |
| NCT01318135 | Long-term Safety Study of Alogliptin Used in Combination With Sulfonylurea or Metformin in Participants With Type 2 Diabetes in Japan | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00251953 | GALLANT 8 Tesaglitazar Add-on to Metformin | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT05298670 | Drug Repurposing Using Metformin for Improving the Therapeutic Outcome in Multiple Sclerosis Patients | multiple sclerosis | Metformin (NPC230775) | |
| NCT05349474 | Metformin Treatment in Progressive Multiple Sclerosis | primary progressive multiple sclerosis;secondary progressive multiple sclerosis | Metformin (NPC230775) | |
| NCT00722371 | MK0431 and Pioglitazone Co-Administration Factorial Study in Patients With Type 2 Diabetes Mellitus (0431-102 AM2) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01068730 | Bioequivalence Study of 500 mg and 1000 mg Glucophage (Metformin) Tablets in Healthy Subjects | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01046422 | Safety Study of BMS-770767 in Subjects With Type 2 Diabetes | diabetes mellitus | Metformin (NPC230775) | |
| NCT00690456 | Effect of Rimonabant and Metformin Combination on Glycemic Control in Patients With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02981329 | Fetal Hemoglobin Induction Treatment Metformin | sickle cell anemia | Metformin (NPC230775) | |
| NCT03863405 | Metformin Use in Rheumatoid Arthritis | rheumatoid arthritis | Metformin (NPC230775) | |
| NCT04177303 | Incretin Hormones in Type 1 Diabetes Mellitus | type 1 diabetes mellitus | Metformin (NPC230775) | |
| NCT00971295 | Effect of Eslicarbazepine Acetate on the Pharmacokinetics of Metformin in Healthy Volunteers | neuropathic pain | Metformin (NPC230775) | |
| NCT02956044 | Interaction of Bexagliflozin With Metformin, Glimepiride and Sitagliptin | diabetes mellitus | Metformin (NPC230775) | |
| NCT00499707 | Efficacy and Safety Study of Rosiglitazone/Metformin Therapy vs Rosiglitazone and Metformin in Type 2 Diabetes Subjects | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00763815 | GLP-1 Receptor Agonist Lixisenatide in Patients With Type 2 Diabetes for Glycemic Control and Safety Evaluation, on Top of Pioglitazone | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01068743 | Bioequivalence Study of Fixed Dose Combination of 2.5 mg Saxagliptin/850 mg Metformin Tablet Relative to 2.5 mg Onglyza and 850 mg Glucophage Tablets Co-Administered | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT04559308 | The Effect of Metformin on Breast Cancer Patients | breast cancer | Metformin (NPC230775) | |
| NCT04248998 | Calorie Restriction With or Without Metformin in Triple Negative Breast Cancer | triple-negative breast cancer | Metformin (NPC230775) | |
| NCT02531308 | Metformin in Combination With Standard Induction Therapy for Large B-cell Lymphoma (DLBCL) | diffuse large B-cell lymphoma | Metformin (NPC230775) | |
| NCT01329016 | Glyburide and Metformin for Gestational Diabetes Mellitus (GDM) | gestational diabetes | Metformin (NPC230775) | |
| NCT02562664 | Metformin Improves Clinical Pregnancy Rate in Polycystic Ovarian Syndrome Patients | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT01087866 | Metformin Versus Insulin in the Treatment of Gestational Diabetes Mellitus | gestational diabetes | Metformin (NPC230775) | |
| NCT00121667 | Study Assessing Saxagliptin Treatment In Type 2 Diabetic Subjects Who Are Not Controlled With Metformin Alone | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01075282 | A Study in Participants With Type 2 Diabetes Mellitus (AWARD-2) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00713830 | GLP-1 Receptor Agonist Lixisenatide in Patients With Type 2 Diabetes for Glycemic Control and Safety Evaluation, on Top of Sulfonylurea | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02083692 | Pilot Study of Metformin in Head and Neck Squamous Cell Cancer and Its Effects on Stromal-epithelial Metabolic Uncoupling | squamous cell carcinoma | Metformin (NPC230775) | |
| NCT04141163 | Metformin in Patients With Fragile X | fragile X syndrome | Metformin (NPC230775) | |
| NCT00971243 | Efficacy and Safety of MP-513 in Combination With Metformin in Patients With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00120146 | Glaser Obesity Study | obesity | Metformin (NPC230775) | |
| NCT00000620 | Action to Control Cardiovascular Risk in Diabetes (ACCORD) | atherosclerosis;coronary artery disease;Hypercholesterolemia;hypertension;type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02682121 | Mitochondrial-related Platelet Transcript Expression Levels in Pre-diabetic Subjects Randomized to Metformin or Placebo | prediabetes syndrome | Metformin (NPC230775) | |
| NCT01779375 | RISE Pediatric Medication Study | prediabetes syndrome | Metformin (NPC230775) | |
| NCT01169779 | Efficacy and Safety of Lixisenatide in Patients With Type 2 Diabetes Mellitus Insufficiently Controlled by Metformin | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02022007 | Saxagliptin + Metformin Compared to Saxagliptin or Metformin Monotherapy in PCOS Women With Impaired Glucose Homeostasis | polycystic ovary syndrome | Metformin (NPC230775) | |
| NCT00482729 | MK0431A Comparative Study in Patients With Type 2 Diabetes (0431A-079)(COMPLETED) | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01881828 | Metformin Therapy for Overweight Adolescents With Type 1 Diabetes | type 1 diabetes mellitus | Metformin (NPC230775) | |
| NCT00379769 | RECORD: Rosiglitazone Evaluated for Cardiac Outcomes and Regulation of Glycaemia in Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00734474 | A Study of LY2189265 Compared to Sitagliptin in Participants With Type 2 Diabetes Mellitus on Metformin | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT02949700 | Phase I/II Study OF Metformin in Combination With Cisplatin and Radiation in Head and Neck Squamous Cell Carcinoma | head and neck squamous cell carcinoma | Metformin (NPC230775) | |
| NCT04939935 | Implementation of Metformin theraPy to Ease Decline of Kidney Function in Polycystic Kidney Disease (IMPEDE-PKD) | Autosomal dominant polycystic kidney disease | Metformin (NPC230775) | |
| NCT00528879 | A Phase III Study of BMS-512148 (Dapagliflozin) in Patients With Type 2 Diabetes Who Are Not Well Controlled on Metformin Alone | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01310231 | A Trial of Standard Chemotherapy With Metformin (vs Placebo) in Women With Metastatic Breast Cancer | breast cancer | Metformin (NPC230775) | |
| NCT02593097 | Metformin for the Prevention of Episodic Migraine (MPEM) | migraine disorder | Metformin (NPC230775) | |
| NCT00842400 | A Drug Interaction Study of VI-0521 With Metformin, Sitagliptan and Probenecid in Healthy Subjects. | obesity | Metformin (NPC230775) | |
| NCT00261352 | GALLANT 14 Tesaglitazar vs. Metformin and Fenofibrate | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT03192293 | Metformin and Simvastatin in Addition to Fulvestrant | breast cancer | Metformin (NPC230775) | |
| NCT02823691 | The MetNET-2 Trial | neuroendocrine neoplasm | Metformin (NPC230775) | |
| NCT01318109 | Efficacy and Safety of Alogliptin Used Combination With Metformin in Participants With Type 2 Diabetes in Japan | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT00683735 | Quantification of the Dipeptidyl Peptidase (DPP)-4 Inhibition-mediated Enhancement of the Activity of the Entero-insular Axis | diabetes mellitus | Metformin (NPC230775) | |
| NCT01111955 | Safety Study of BMS-823778 in Subjects With Type 2 Diabetes | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT04865835 | A Clinical Trial Study to Determine the Effect of an Investigational Drug (SEP-363856) Has on the Way That the Drug Metformin Travels Through the Body in People With Schizophrenia. | schizophrenia | Metformin (NPC230775) | |
| NCT02497638 | LIpitor and biGuanide to Androgen Delay Trial | prostate cancer | Metformin (NPC230775) | |
| NCT01396564 | Pioglitazone and Metformin in Diabetic Children | type 2 diabetes mellitus | Metformin (NPC230775) | |
| NCT01885013 | Myocet + Cyclophosphamide + Metformin Vs Myocet + Cyclophosphamide in 1st Line Treatment of HER2 Neg. Metastatic Breast Cancer Patients | breast carcinoma | Metformin (NPC230775) |
❱❱❱ Associated Human Diseases and Detailed Association Evidence
How do we define the Plant-Targeted Human Disease Association?
Associated human diseases of an individual plant are summurized based on FOUR types of association evidence, these include:
❶ Association by Therapeutic Target: Bioactive protein targets of the plant were defined in "Molecular Targets" section, target-disease associations collected from TTD database were subsequently used to build the associations between the plant and its targeted human diseases.
❷ Association by Disease Gene Reversion: Plant and a specific disease will be associated when >= 1 plant target gene overlaped with disease's DEGs.
❸ Association by Clinical Trials of Plant: Plant and a specific disease will be associated when >= 1 clinical trial (the plant is the intervetion) can be matched in ClinicalTrials.gov database.
❹ Association by Clinical Trials of Plant Ingredients: Plant and a specific disease will be associated when >= 1 clinical trial (the plant ingredient is the intervetion) can be matched in ClinicalTrials.gov database.
Associated Disease of the Plant |
Association Type & Detailed Evidence |
|---|---|
Solid tumour/cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A00-2F9Z |
CA4,GSK3B,XDH,CSNK2A1,MKNK1,FLT3,MKNK2,HIF1A,CA12
|
Diabetes mellitus arising in pregnancyDisease Category: 18.Pregnancy, childbirth or the puerperiumDisease ICD-11 Code: JA63.2 |
NCT00414245,NCT03320694,NCT01329016,NCT01215331,NCT03651531,NCT02275845,NCT03841591,NCT04615351,NCT01087866
|
Malignant neoplasms of biliary tract, distal bile ductDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C15 |
CSNK2A3,LMNA,CA4,AKR1B1,CSNK2A1,MMP12,HIF1A,NOX4,ESR2
|
Germ cell tumour of testisDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C80.2 |
CA12,DPP4,CA4,AKR1B1,ACHE,XDH,HIF1A,TTR,NOX4
|
Periodontal diseaseDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DA0C |
NCT03204071,NCT02274090,NCT02372656,NCT03204058,NCT02048761,NCT02283554,NCT02580331,NCT02461667
|
Serous cystadenoma,borderline malignancy of ovaryDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C73.4 |
CA12,CSNK2A3,CSNK2A1,MMP12,XDH,CYP1A2,NOX4,PSMB5
|
Adenocarcinoma of pancreasDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C10.0 |
DPP4,LMNA,ACHE,XDH,CYP1B1,CA7,TTR,HIF1A
|
Mesothelioma of pleuraDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C26.0 |
CA12,DPP4,LMNA,MMP12,XDH,FLT3,CSNK2A2,CA7
|
Hepatocellular carcinoma of liverDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C12.02 |
CA12,CA4,MMP12,CYP19A1,NOX4,CA12,MMP12,CYP19A1
|
Endometrial cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C76 |
NCT02755844,NCT02990728,NCT01911247,NCT04792749,NCT01205672,NCT02042495,NCT01797523
|
Intermediate hyperglycaemia, unspecifiedDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A40.Z |
NCT04742751,NCT01336322,NCT02682121,NCT03848533,NCT01804049,NCT01779375,NCT01779362
|
Carcinosarcoma of uterusDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C76.43 |
DPP4,AKR1B1,MMP12,XDH,CA7,CYP19A1,PSMB5
|
Malignant neoplasms of thyroid gland, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D10.Z |
CA12,DPP4,MMP12,XDH,CYP1B1,CA7,NOX4
|
Adenocarcinoma of bronchus or lungDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25.0 |
CA12,THPO,MMP12,ACHE,XDH,CA7,TTR
|
Malignant neoplasms of thymusDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C27 |
DPP4,AKR1B1,MMP12,ACHE,FLT3,CYP1B1,PSMB5
|
Malignant neoplasms of adrenal glandDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D11 |
CA12,DPP4,CA4,MMP12,CA7,CYP19A1,NOX4
|
Colorectal cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B91 |
FLT3,MKNK1,MKNK2
NCT01632020,NCT02614339,NCT01816659 |
Non-small cell lung cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25 |
NCT01864681,NCT03086733,NCT01717482,NCT02019979,NCT01997775,NCT04170959
|
Glioblastoma, primary, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH0MB1 |
NCT05183204,NCT03151772,NCT04945148,NCT02780024,NCT03243851,NCT04691960
|
PheochromocytomaDisease Category: X.Extension CodesDisease ICD-11 Code: XH3854 |
CA4,MMP12,ACHE,XDH,TTR,NOX4
|
HepatoblastomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C12.01 |
CA12,CA4,AKR1B1,MMP12,CYP19A1,NOX4
|
Glioblastoma of brainDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A00.00 |
CA12,CSNK2A3,DPP4,LMNA,CYP1B1,CYP19A1
|
Diabetes mellitus, type unspecifiedDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A14 |
DPP4
NCT00108615,NCT02088034,NCT05262257,NCT02484209,NCT03670043,NCT01370707,NCT00313313,NCT00727857,NCT01915550,NCT01613755,NCT03039075,NCT00327015,NCT00772174,NCT00286442,NCT01464437,NCT04581447,NCT01890122,NCT04998032,NCT00316082,NCT00959101,NCT00279240,NCT00770445,NCT04416269,NCT00770835,NCT01107717,NCT00038727,NCT02956044,NCT01046422,NCT00683735,NCT01766778,NCT03030300,NCT03824002,NCT02719756,NCT03258723,NCT01814748,NCT04050098,NCT00820573,NCT02042664,NCT01874080,NCT00432276,NCT05440591,NCT02249910,NCT00684528,NCT00046462,NCT02844803,NCT00969566,NCT01192152,NCT01192139,NCT01334125,NCT00762957,NCT00754403,NCT03179254,NCT04209075 |
Diffuse large B-cell lymphomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A81 |
FLT3
NCT03200015,NCT03600363,NCT02815397,NCT02531308 |
Rheumatoid arthritisDisease Category: 15.Diseases of the musculoskeletal system or connective tissueDisease ICD-11 Code: FA20 |
AKR1B1
NCT01405118,NCT03863405,NCT04068246,NCT04196868 |
Fragile X chromosomeDisease Category: 20.Developmental anomaliesDisease ICD-11 Code: LD55 |
GSK3B
NCT03722290,NCT03479476,NCT03862950,NCT04141163 |
Type1diabetesmellitusDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A10 |
NCT01881828,NCT01808690,NCT02045290,NCT01483560,NCT04177303
|
Squamous cell carcinomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B60-2D01 |
NCT03833466,NCT02083692,NCT02394652,NCT03618654,NCT03076281
|
Non-alcoholic fatty liver diseaseDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DB92 |
NCT02696941,NCT00699036,NCT04972396,NCT01084486,NCT02457286
|
Ageing associated decline in intrinsic capacityDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: MG2A |
NCT03072485,NCT03713801,NCT04264897,NCT03861767,NCT02308228
|
LeishmaniasisDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1F54 |
DPP4,AKR1B1,MMP12,FLT3,NOX4
|
Squamous cell carcinoma of bronchus or lungDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25.2 |
CA12,HSD17B1,MMP12,XDH,CA7
|
Superficial ovarian endometriosisDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GA10.B4 |
DPP4,LMNA,CA4,ACHE,HSD17B1
|
Chronic kidney diseaseDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GB61 |
XDH
NCT05373680,NCT02895750,NCT02848508 |
PsoriasisDisease Category: 14.Diseases of the skinDisease ICD-11 Code: EA90 |
DPP4,FLT3
MMP12 NCT02644954 |
GlaucomaDisease Category: 09.Diseases of the visual systemDisease ICD-11 Code: 9C61 |
AKR1B1,CA4,ACHE
NCT05426044 |
Liver cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C12 |
CSNK2A1,MKNK2,MKNK1
NCT03184493 |
Lung cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25 |
FLT3,MMP12
NCT02115464,NCT02285855 |
Pulmonary hypertensionDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BB01 |
HIF1A
NCT00000620,NCT01352026,NCT03617458 |
MelanomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C30 |
NCT02143050,NCT02190838,NCT01638676,NCT03311308
|
Malignant neoplasm metastasis in lymph nodes of head, face or neckDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D60.0 |
MMP12,NOX4
NCT04414540,NCT02949700 |
Neuroendocrine carcinomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C34 |
NCT02294006,NCT02279758,NCT02823691,NCT02254512
|
Malignant neoplasm of pancreas, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C10.Z |
NCT01488552,NCT01167738,NCT01210911,NCT03889795
|
Autosomal dominant polycystic kidney diseaseDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GB81 |
NCT02656017,NCT02903511,NCT03764605,NCT04939935
|
Adenocarcinoma of prostateDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C82.0 |
TTR,NOX4
NCT01996696,NCT01796028 |
Other specified malignant neoplasms of kidney, except renal pelvisDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C90.Y |
ACHE,XDH,CYP19A1,MMP12
|
Other specified malignant neoplasms of bronchus or lungDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25.Y |
CA12,MMP12,XDH,HSD17B1
|
Urothelial carcinoma of bladderDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C94.2 |
CYP1A2,TTR,AKR1B1,HSD17B1
|
Cytomegaloviral diseaseDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D82 |
THPO,ACHE,ESR2,CYP1A1
|
Chikungunya virus diseaseDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D40 |
LMNA,XDH,CYP19A1,HIF1A
|
Renal cell carcinoma, chromophobe typeDisease Category: X.Extension CodesDisease ICD-11 Code: XH6153 |
CA12,CYP1A1,CYP1A2,TTR
|
Carpal tunnel syndromeDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8C10.0 |
DPP4,THPO,CYP1B1,NOX4
|
Polycystic ovary syndromeDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A80.1 |
MMP12
NCT01581814,NCT03008551,NCT02244567,NCT02026869,NCT01733459,NCT02689843,NCT04113889,NCT04852510,NCT00344851,NCT03086005,NCT00466622,NCT03905941,NCT01622257,NCT04867252,NCT00714233,NCT03981861,NCT04973891,NCT00151411,NCT02086526,NCT02498522,NCT00159536,NCT03259919,NCT05394142,NCT03108963,NCT02562664,NCT01482286,NCT02022007,NCT00948402,NCT03117517,NCT01775176,NCT03229057,NCT03674385,NCT00283816 |
Breast cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C60-2C6Y |
CA12,ESR2,CA4,CYP19A1,MKNK2,MKNK1
NCT04559308,NCT01627067,NCT01477060,NCT02488564,NCT04300790,NCT02506777,NCT02506790,NCT02472353,NCT03006172,NCT01266486,NCT02028221,NCT01042379,NCT03238495,NCT01980823,NCT05053841,NCT04899349,NCT01302002,NCT00930579,NCT04248998,NCT00909506,NCT01650506,NCT03192293,NCT04143282,NCT02360059,NCT01310231,NCT01589367 |
Ovarian cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C73 |
FLT3
NCT02312661,NCT03378297 |
Mature T-cell lymphomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A90 |
MKNK1,HIF1A,MKNK2
|
Alzheimer diseaseDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8A20 |
ESR2,FLT3,ACHE
|
SchizophreniaDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6A20 |
AKR1B1
NCT00401973,NCT04865835 |
Heart failureDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BD10-BD1Z |
XDH
NCT02810132,NCT05093959 |
AdenocarcinomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D40 |
NCT02186847,NCT01877564,NCT01578551
|
Rectum cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B92 |
NCT02437656,NCT02473094,NCT03053544
|
NeoplasmsDisease Category: 02.NeoplasmsDisease ICD-11 Code: 02 |
NCT04428086,NCT01566799,NCT03017833
|
Multiple sclerosisDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8A40 |
NCT05298670,NCT04121468,NCT05131828
|
Unspecified malignant neoplasms of unspecified sitesDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D4Z |
NCT01442870,NCT01529593,NCT01087983
|
Male infertilityDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GB04 |
NCT03681197,NCT03135301,NCT02848950
|
Malignant neoplasm metastasis in male genital organsDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2E06 |
NCT01677897,NCT04925063,NCT04926155
|
Primary neoplasms of brainDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A00 |
NCT01528046,NCT02149459,NCT02040376
|
Malignant neoplasms of prostate, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C82.Z |
NCT02614859,NCT02640534,NCT01561482
|
Adenocarcinoma of stomachDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B72.0 |
MMP12,CYP19A1,NOX4
|
West Nile virus infectionDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D46 |
DPP4,CYP19A1,ACHE
|
Other specified malignant neoplasms ofcolonDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B90.Y |
MMP12,NOX4,CYP1A1
|
Malignant neoplasms of oesophagusDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B70 |
MMP12,ACHE,NOX4
|
Malignant neoplasms of corpus uteri, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C76.Z |
MMP12,XDH,CYP1A1
|
Invasive carcinoma of breastDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C61 |
CA12,THPO,MMP12
|
Type 2 diabetes mellitusDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A11 |
DPP4
NCT00359112,NCT01302145,NCT00814372,NCT01368081,NCT00425009,NCT00251953,NCT00469586,NCT00614120,NCT01002807,NCT01111955,NCT00374907,NCT00614055,NCT00272064,NCT00086515,NCT00661362,NCT01472367,NCT01126580,NCT00672386,NCT00960661,NCT01289119,NCT00707031,NCT01068717,NCT01154348,NCT00528372,NCT00482729,NCT00961857,NCT01353391,NCT00885378,NCT00295633,NCT00383578,NCT00000620,NCT00643851,NCT01381900,NCT01383356,NCT00086502,NCT00575588,NCT01340664,NCT00541775,NCT00975052,NCT01240759,NCT01089166,NCT01245166,NCT01137812,NCT00069836,NCT00102466,NCT00081328,NCT00859898,NCT00318461,NCT00348712,NCT00673465,NCT00868790,NCT00901979,NCT00121641,NCT01211197,NCT00261352,NCT00856986,NCT00263276,NCT01357876,NCT00103857,NCT00789035,NCT01075282,NCT01089192,NCT01071850,NCT00712673,NCT00871936,NCT00605475,NCT00611884,NCT00333151,NCT00396357,NCT01076075,NCT00519480,NCT00481663,NCT01093794,NCT00382096,NCT01095666,NCT00767000,NCT01068743,NCT00839527,NCT01462266,NCT01175473,NCT01117584,NCT00715624,NCT00087516,NCT00266253,NCT01217892,NCT01217073,NCT00337610,NCT00279045,NCT01068730,NCT00690456,NCT00961909,NCT00263965,NCT01023581,NCT00698789,NCT01459809,NCT01305551,NCT00754689,NCT00328172,NCT01028391,NCT00755287,NCT00975286,NCT00743002,NCT00543361,NCT01296412,NCT01089179,NCT00885352,NCT00798161,NCT00106340,NCT00479466,NCT00376038,NCT00881530,NCT00427154,NCT00830076,NCT00789191,NCT00676338,NCT00924573,NCT01095653,NCT00627445,NCT00971243,NCT01135433,NCT00838903,NCT01396564,NCT00571519,NCT00598793,NCT00454233,NCT00544518,NCT01204294,NCT00856284,NCT00121667,NCT01434186,NCT00399711,NCT01318109,NCT00698230,NCT00067951,NCT00331851,NCT01292993,NCT00783744,NCT00499707,NCT00375388,NCT00918138,NCT00792935,NCT00110851,NCT00700817,NCT00722371,NCT00613951,NCT00379769,NCT00734474,NCT00728351,NCT00875394,NCT01064687,NCT00316758,NCT00899470,NCT00449605,NCT01204775,NCT00961480,NCT00971659,NCT00744367,NCT00353691,NCT00032487,NCT00915772,NCT00097279,NCT00420511,NCT00350779,NCT00528879,NCT01332071,NCT01076088,NCT00960076,NCT00713830,NCT01034111,NCT01059825,NCT00332488,NCT00174642,NCT00525330,NCT01169779,NCT00701090,NCT01365091,NCT00855166,NCT01340768,NCT01106690,NCT01318135,NCT00763451,NCT00976937,NCT00099866,NCT00131664,NCT01106677,NCT00900146,NCT00968812,NCT00414245,NCT00851903,NCT00860288,NCT00763815,NCT01051011,NCT01106625,NCT01089205,NCT00097500,NCT00468039,NCT00754988,NCT00184574,NCT00631488,NCT00111631 |
AsthmaDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CA23 |
CA4,MMP12
|
ParkinsonismDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8A00 |
ACHE,GSK3B
|
Myeloproliferative neoplasmDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A20 |
FLT3,GSK3B
|
NADisease Category: NADisease ICD-11 Code: NA |
NOX4
NCT05132439 |
Acute myeloid leukaemiaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A60 |
FLT3,GSK3B
|
Bacterial infectionDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1A00-1C4Z |
CA4,CA12
|
Seborrhoeic dermatitisDisease Category: 14.Diseases of the skinDisease ICD-11 Code: EA81 |
CA12,CA4
|
Cushing syndromeDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A70 |
CYP19A1,ESR2
|
Myocardial infarctionDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA41-BA43 |
DPP4,THPO
|
Malignant haematopoietic neoplasmDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B33 |
DPP4,FLT3
|
Acute diabete complicationDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A2Y |
AKR1B1,DPP4
|
DementiaDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6D80-6D8Z |
ACHE
NCT04220021 |
NeuropathyDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8C0Z |
AKR1B1,CYP19A1
|
Pain, unspecifiedDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: MG3Z |
ACHE,AKR1B1
|
Multiple myelomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A83 |
FLT3
NCT02967276 |
Human immunodeficiency virus disease without mention of associated disease or condition, clinical stage unspecifiedDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1C62.Z |
NCT02659306,NCT02064374
|
Breast in situ carcinomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2E65 |
NCT01885013,NCT02278965
|
Disorders of intellectual development, unspecifiedDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6A00.Z |
NCT03862950,NCT03479476
|
Acute viral hepatitisDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1E50 |
NCT00063232,NCT03000218
|
Hepatocellular carcinoma, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH4W48 |
NCT02819869,NCT02672488
|
Malignant neoplasms of bladder, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C94.Z |
NCT02360618,NCT03379909
|
COVID-19Disease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D6Y |
NCT04604678,NCT04510194
|
Neurobehavioral disorderDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8A00-8A0Z |
NCT03479476,NCT03862950
|
Other specified malignant neoplasms of colonDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B90.Y |
NCT01926769,NCT01725490
|
Malignant neoplasms of colonDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B90 |
NCT01440127,NCT03047837
|
Other specified non-alcoholic fatty liver diseaseDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DB92.Y |
NCT00303537,NCT00063635
|
Insulin-resistance syndromesDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A44 |
NCT04148183,NCT04264897
|
Sex development malformative disorderDisease Category: 20.Developmental anomaliesDisease ICD-11 Code: LD2A |
NCT03862950,NCT03479476
|
Symptoms and signs involving cognition, unspecifiedDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: MB21.Z |
NCT04098666,NCT00620191
|
Atrial fibrillationDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BC81.3 |
CA7
NCT02931253 |
Muscular dystrophyDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8C70 |
NCT02018731,NCT01995032
|
Head and neck cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D42 |
NCT02402348,NCT02325401
|
Aneurysm/dissectionDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BD50 |
NCT04500756,NCT04224051
|
Endometrial glandular hyperplasiaDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GA16.0 |
NCT04607252,NCT01685762
|
Inflammatory bowel diseases, unspecifiedDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DD7Z |
MMP12,NOX4
|
Melanoma of skinDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C30 |
TTR,NOX4
|
Pneumonia due to PseudomonasaeruginosaDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CA40.05 |
DPP4,MMP12
|
Chronic rhinosinusitisDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CA0A |
MMP12,CYP1A1
|
Systemic lupus erythematosusDisease Category: 04.Diseases of the immune systemDisease ICD-11 Code: 4A40.0 |
ACHE,CYP1A1
|
Gliomas of brainDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A00.0 |
MMP12,NOX4
|
Prostate cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C82 |
ESR2
NCT04536805,NCT01620593,NCT00268476,NCT03137186,NCT02945813,NCT01864096,NCT01733836,NCT01243385,NCT01215032,NCT02946996,NCT00881725,NCT03465345,NCT02497638 |
ObesityDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5B80-5B81 |
NCT00631033,NCT00941382,NCT03349775,NCT02063802,NCT03000218,NCT00120146,NCT00349635,NCT04839237,NCT01725126,NCT00892073,NCT01677923,NCT00842400
|
Metabolic disorderDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5C50-5D2Z |
DPP4
NCT02773927,NCT00703755,NCT00349128,NCT02778776,NCT04817787,NCT02629627,NCT01234649,NCT01046500,NCT00362323,NCT00348725 |
Diffuse large B-cell lymphomasDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A81 |
CA12,LMNA,THPO,AKR1B1,CSNK2A1,ACHE,CSNK2A2,NOX4,ESR2,PSMB5
|
Coronavirus infectionDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D92 |
CSNK2A1
|
Chronic painDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: MG30 |
FLT3
|
Vasomotor/allergic rhinitisDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CA08 |
ESR2
|
Menopausal disorderDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GA30 |
ESR2
|
SepsisDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1G40-1G41 |
ESR2
|
Bone/articular cartilage neoplasmDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2F7B |
FLT3
|
Myotonic disorderDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8C71 |
GSK3B
|
Transplanted organ/tissueDisease Category: 24.Factors influencing health status or contact with health servicesDisease ICD-11 Code: QB63 |
FLT3
|
OsteosarcomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B51 |
GSK3B
|
Mineral deficiencyDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5B5K |
XDH
|
InfluenzaDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1E30-1E32 |
XDH
|
GoutDisease Category: 15.Diseases of the musculoskeletal system or connective tissueDisease ICD-11 Code: FA25 |
XDH
|
CataractDisease Category: 09.Diseases of the visual systemDisease ICD-11 Code: 9B10 |
AKR1B1
|
AmyloidosisDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5D00 |
TTR
|
Oesophageal/gastroduodenal disorderDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DD90 |
ACHE
|
Hepatitis virus infectionDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1E50-1E51 |
ESR2
|
Prostate hyperplasiaDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GA90 |
ESR2
|
Renal cell carcinomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C90 |
MMP12
|
PediculosisDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1G00 |
ACHE
|
Inflammatory arthropathyDisease Category: 15.Diseases of the musculoskeletal system or connective tissueDisease ICD-11 Code: FA2Z |
FLT3
|
Hyper-lipoproteinaemiaDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5C80 |
ESR2
|
Ischaemic/haemorrhagic strokeDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8B20 |
THPO
|
Substance abuseDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6C40 |
ACHE
|
EndometriosisDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GA10 |
CYP19A1
|
Human immunodeficiency virus diseaseDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1C60-1C62 |
FLT3
|
Brain cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A00 |
HIF1A
|
Phakomatoses/hamartoneoplastic syndromeDisease Category: 20.Developmental anomaliesDisease ICD-11 Code: LD2D |
FLT3
|
Unspecific substance harmful effectDisease Category: 22.Injury, poisoning or certain other consequences of external causesDisease ICD-11 Code: NE6Z |
ACHE
|
CardiomyopathyDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BC43 |
TTR
|
Chronic myelomonocytic leukaemiaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A40 |
FLT3
|
Adrenomedullary hyperfunctionDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A75 |
HIF1A
|
Indeterminate colitisDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DD72 |
ESR2
|
MastocytosisDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A21 |
FLT3
|
Peritoneal cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C51 |
FLT3
|
Myasthenia gravisDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8C6Y |
ACHE
|
Mild neurocognitive disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6D71 |
ACHE
|
Inborn purine/pyrimidine/nucleotide metabolism errorDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5C55 |
XDH
|
OesophagitisDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DA24 |
CYP19A1
|
EncephalopathyDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8E47 |
XDH
|
Parasitic worm infestationDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1F90 |
ACHE
|
Idiopathic interstitial pneumonitisDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CB03 |
FLT3
|
Mature B-cell lymphomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A85 |
FLT3
|
Autism spectrum disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6A02 |
DPP4
|
BCR-ABL1-negative chronic myeloid leukaemiaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A41 |
FLT3
|
Chronic arterial occlusive diseaseDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BD4Z |
DPP4
|
Unspecified viral infection of unspecified siteDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D9Z |
FLT3
|
ThrombosisDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DB61-GB90 |
AKR1B1
|
Diabetic foot ulcerDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BD54 |
NCT01710527
|
Precursor cell lymphoblastic leukaemia, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH5J37 |
NCT01324180
|
Ulcerative colitisDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DD71 |
NCT04750135
|
Low back painDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: ME84.2 |
NCT04055012
|
Malignant neoplasm metastasis, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2E2Z |
NCT01840007
|
Follicular lymphomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A80 |
NCT03600363
|
Polyneuropathy, unspecifiedDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8C0Z |
NCT04780854
|
Coronary atherosclerosisDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA80 |
NCT00000620
|
OsteoarthritisDisease Category: 15.Diseases of the musculoskeletal system or connective tissueDisease ICD-11 Code: FA00-FA05 |
NCT04767841
|
HIV-infected patients with tuberculosisDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1B10-1B14 |
NCT05215990
|
Malignant neoplasms of ill-defined sitesDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D42 |
NCT03477162
|
Hidradenitis suppurativaDisease Category: 14.Diseases of the skinDisease ICD-11 Code: ED92 |
NCT04649502
|
HypopituitarismDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A61.0 |
NCT03490019
|
Urogenital cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C95-2C9Z |
NCT02365597
|
Adenocarcinoma, endocervical typeDisease Category: X.Extension CodesDisease ICD-11 Code: XH0GS9 |
NCT02394652
|
Sickle-cell disorderDisease Category: 03.Diseases of the blood or blood-forming organsDisease ICD-11 Code: 3A51 |
NCT02981329
|
Schizoaffective disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6A21 |
NCT00401973
|
Carcinomas of ovaryDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C73.0 |
NCT02437812
|
Pregnancy/childbirth/puerperium maternal infectionDisease Category: 18.Pregnancy, childbirth or the puerperiumDisease ICD-11 Code: JB63 |
NCT03056274
|
Migraine disorder(TM1)Disease Category: 26.Supplementary Chapter Traditional Medicine Conditions - Module IDisease ICD-11 Code: SD10 |
NCT02593097
|
Generalised anxiety disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6B00 |
NCT00809536
|
AngiosarcomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B56 |
NCT01042379
|
Cardiovascular diseaseDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA00-BE2Z |
NCT01037660
|
Severe acute respiratory syndromeDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D65 |
NCT04510194
|
HypercholesterolaemiaDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5C80.0 |
NCT00000620
|
Acute ischaemic heart disease, unspecifiedDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA4Z |
NCT05305898
|
Primary progressive multiple sclerosisDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8A40.1 |
NCT05349474
|
Secondary progressive multiple sclerosisDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8A40.2 |
NCT05349474
|
Chronic lymphocytic leukaemia or small lymphocytic lymphomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A82.0 |
NCT01750567
|
Inborn lipid metabolism errorDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5C52 |
NCT00361868
|
MalariaDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1F40-1F45 |
NCT05236530
|
Chronic arterial occlusive disease, unspecifiedDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BD4Z |
NCT03054519
|
Large cell carcinoma of bronchus or lungDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25.3 |
NCT02186847
|
Cervical cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C77 |
NCT04275713
|
Obstructive sleep apnoeaDisease Category: 07.Sleep-wake disordersDisease ICD-11 Code: 7A41 |
NCT04530747
|
Squamous cell carcinoma of oropharynxDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B6A.0 |
NCT03618654
|
Thyroid cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D10 |
NCT05468554
|
Macular degenerationDisease Category: 09.Diseases of the visual systemDisease ICD-11 Code: 9B75 |
NCT02684578
|
Osteosarcoma of bone and articular cartilage of other specified sitesDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B51.Y |
NCT01981525
|
RetinopathyDisease Category: 09.Diseases of the visual systemDisease ICD-11 Code: 9B71 |
NCT02587741
|
Chronic obstructive pulmonary diseaseDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CA22 |
NCT03651895
|
Small cell carcinoma of bronchus or lungDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25.1 |
NCT03994744
|
BlindnessDisease Category: 09.Diseases of the visual systemDisease ICD-11 Code: 9D90.6 |
NCT03490019
|
Huntington diseaseDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8A01.10 |
NCT04826692
|
Columnar metaplastic epithelium of the oesophagus, unspecifiedDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DA23.Z |
NCT01465113
|
Bipolar disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6A60 |
NCT02519543
|
Single episode depressive disorder, severe, without psychotic symptomsDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6A70.3 |
NCT04088448
|
Lipoatrophy/lipodystrophyDisease Category: 14.Diseases of the skinDisease ICD-11 Code: EF01 |
NCT00362440
|
Neuropathic painDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8E43.0 |
NCT00971295
|
Schizophrenia or other primary psychotic disorders, unspecifiedDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6A2Z |
NCT04511416
|
Spastic cerebral palsyDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8D20 |
NCT03710343
|
Central diabetes insipidusDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A61.5 |
NCT02460354
|
LeprosyDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1B20 |
NCT05243654
|
coronary artery diseaseDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA8Y-BA8Z |
NCT00000620
|
Impaired glucose toleranceDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A40.1 |
NCT01234649
|
Burn and burn infectionDisease Category: 22.Injury, poisoning or certain other consequences of external causesDisease ICD-11 Code: ND90-NE2Z |
NCT04530058
|
Duchenne muscular dystrophyDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8C70.1 |
NCT02516085
|
Crohn diseaseDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DD70 |
MMP12
|
Coeliac diseaseDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DA95 |
CYP1A1
|
Zika virus diseaseDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D48 |
ACHE
|
Osteoarthritis, unspecifiedDisease Category: 15.Diseases of the musculoskeletal system or connective tissueDisease ICD-11 Code: FA0Z |
CA12
|
Idiopathic pulmonary fibrosisDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CB03.4 |
MMP12
|
Myotonic dystrophyDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8C71.0 |
CYP1A1
|
KeratoconusDisease Category: 09.Diseases of the visual systemDisease ICD-11 Code: 9A78.50 |
CA4
|
Pre-eclampsiaDisease Category: 18.Pregnancy, childbirth or the puerperiumDisease ICD-11 Code: JA24 |
CA4
|
Tetralogy of FallotDisease Category: 20.Developmental anomaliesDisease ICD-11 Code: LA88.2 |
CA4
|
Systemic sclerosisDisease Category: 04.Diseases of the immune systemDisease ICD-11 Code: 4A42 |
NOX4
|
Heart failure, unspecifiedDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BD1Z |
CYP1B1
|
Human immunodeficiency virus disease without mention of tuberculosis or malariaDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1C62 |
FLT3
|
Ebola virus diseaseDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D60.01 |
ACHE
|

