Collective Molecular Activities of the Plant: Asarum Heterotropoides
Plant ID: NPO12468
Plant Latin Name: Asarum Heterotropoides
Taxonomy Genus: Asarum
Taxonomy Family: Aristolochiaceae
Plant External Links:
NCBI TaxonomyDB:
366663
Plant-of-the-World-Online:
n.a.
Unknown.
Overview of Ingredients
161 All known Ingredients in Total
Unique ingredients have been isolated from this plant.Plant-Ingredients Associations were manually curated from publications or collected from other databases.
148 Ingredients with Acceptable Bioavailablity
Unique ingredients exhibit acceptable human oral bioavailablity, according to the criteria of SwissADME [PMID: 28256516] and HobPre [PMID: 34991690]. The criteria details:SwissADME: six descriptors are used by SwissADME to evaluate the oral bioavailability of a natural product:
☑ LIPO(Lipophility): -0.7 < XLOGP3 < +5.0
☑ SIZE: 150g/mol < MW < 500g/mol
☑ POLAR(Polarity): 20Ų < TPSA < 130Ų
☑ INSOLU(Insolubility): -6 < Log S (ESOL) < 0
☑ INSATU(Insaturation): 0.25 < Fraction Csp3 < 1
☑ FLEX(Flexibility): 0 < Num. rotatable bonds < 9
If 6 descriptors of a natural plant satisfy the above rules, it will be labeled high HOB.
HobPre: A natural plant ingredient with HobPre score >0.5 is labeled high human oral availability (HOB)
93 Ingredients with experimental-derived Activity
Unique ingredients have activity data available.Ingredient Structrual Cards

Ingredient ID: NPC99886

Ingredient ID: NPC97870
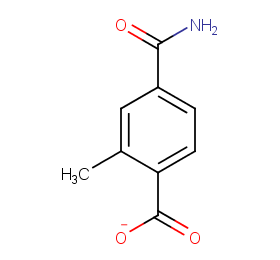
Ingredient ID: NPC97128
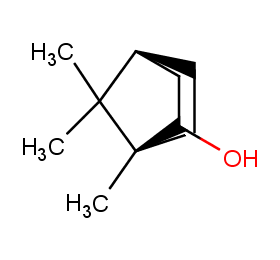
Ingredient ID: NPC96630
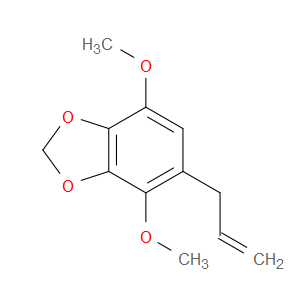
Ingredient ID: NPC92869

Ingredient ID: NPC90105
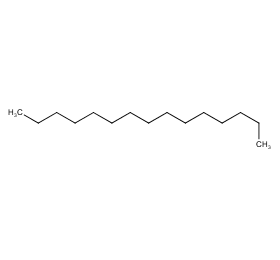
Ingredient ID: NPC89422
Ingredient ID: NPC89069
Ingredient ID: NPC88640
Ingredient ID: NPC88566
Ingredient ID: NPC87682
Ingredient ID: NPC85665
Ingredient ID: NPC85661
Ingredient ID: NPC84856
Ingredient ID: NPC78551
Ingredient ID: NPC76145
Ingredient ID: NPC75158
Ingredient ID: NPC74295

Ingredient ID: NPC73964
Ingredient ID: NPC7343
Ingredient ID: NPC71853

Ingredient ID: NPC71703

Ingredient ID: NPC71645
Ingredient ID: NPC71506
Ingredient ID: NPC69213
Ingredient ID: NPC67790
Ingredient ID: NPC64370
Ingredient ID: NPC64176
Ingredient ID: NPC57471
Ingredient ID: NPC56271
Ingredient ID: NPC54264

Ingredient ID: NPC52464
Ingredient ID: NPC51700
Ingredient ID: NPC50629
Ingredient ID: NPC46679
Ingredient ID: NPC46330
Ingredient ID: NPC46114
Ingredient ID: NPC45727
Ingredient ID: NPC44751
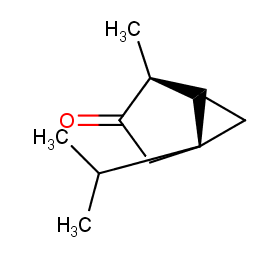
Ingredient ID: NPC44671
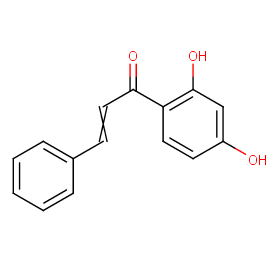
Ingredient ID: NPC43143
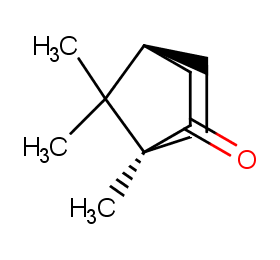
Ingredient ID: NPC40249

Ingredient ID: NPC37871
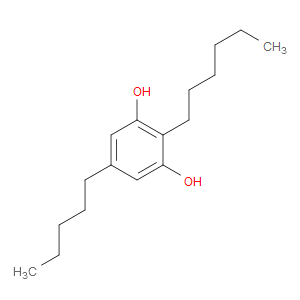
Ingredient ID: NPC37802
Ingredient ID: NPC3649
Ingredient ID: NPC34764
Ingredient ID: NPC33664
Ingredient ID: NPC33611
Ingredient ID: NPC32441
Ingredient ID: NPC31279
Ingredient ID: NPC308218
Ingredient ID: NPC307123
Ingredient ID: NPC29883
Ingredient ID: NPC296742
Ingredient ID: NPC296337
Ingredient ID: NPC293343
Ingredient ID: NPC292869

Ingredient ID: NPC292792

Ingredient ID: NPC287731
Ingredient ID: NPC287660
Ingredient ID: NPC287331

Ingredient ID: NPC287191

Ingredient ID: NPC286683
Ingredient ID: NPC284731
Ingredient ID: NPC283823
Ingredient ID: NPC283655
Ingredient ID: NPC281131
Ingredient ID: NPC280958
Ingredient ID: NPC271397
Ingredient ID: NPC26906
Ingredient ID: NPC268862
Ingredient ID: NPC268647
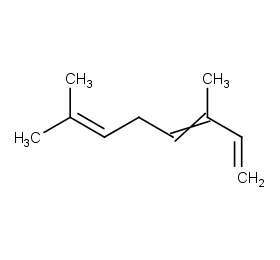
Ingredient ID: NPC262789
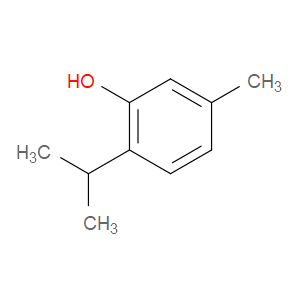
Ingredient ID: NPC259512
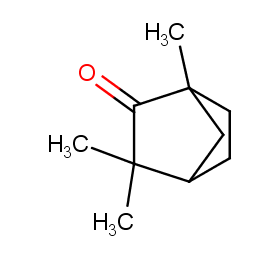
Ingredient ID: NPC258672
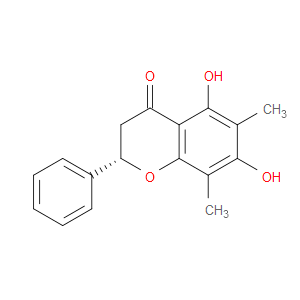
Ingredient ID: NPC248372

Ingredient ID: NPC247922

Ingredient ID: NPC247786
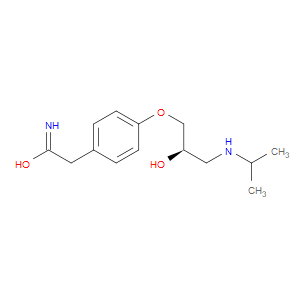
Ingredient ID: NPC247018

Ingredient ID: NPC246620
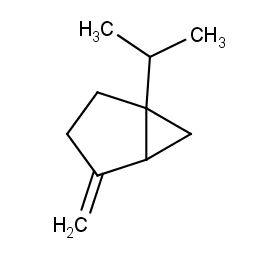
Ingredient ID: NPC246165
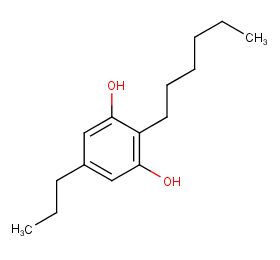
Ingredient ID: NPC244513
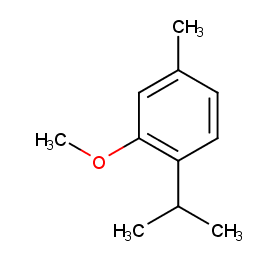
Ingredient ID: NPC24327
Ingredient ID: NPC24294
Ingredient ID: NPC240702
Ingredient ID: NPC240684
Ingredient ID: NPC239039
Ingredient ID: NPC232375
Ingredient ID: NPC227670
Ingredient ID: NPC227458
Ingredient ID: NPC22229
Ingredient ID: NPC222001
Ingredient ID: NPC221733

Ingredient ID: NPC218879
Ingredient ID: NPC217191
Ingredient ID: NPC217002
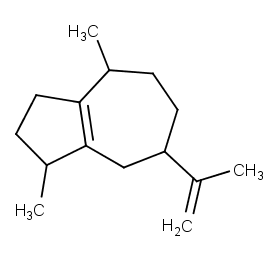
Ingredient ID: NPC216900

Ingredient ID: NPC215756
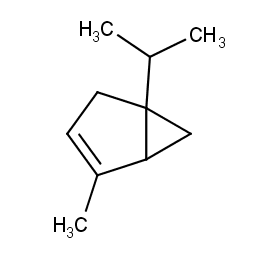
Ingredient ID: NPC215632
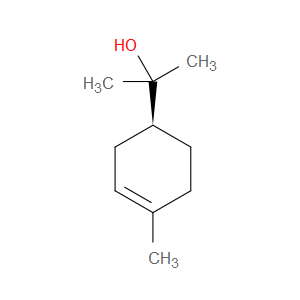
Ingredient ID: NPC214584
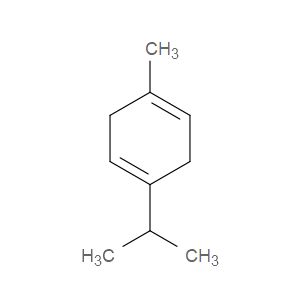
Ingredient ID: NPC213749

Ingredient ID: NPC208151
Ingredient ID: NPC20791
Ingredient ID: NPC207643
Ingredient ID: NPC207561
Ingredient ID: NPC204120
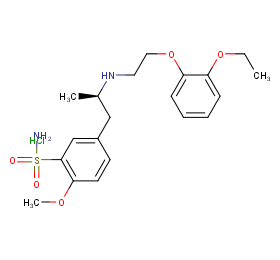
Ingredient ID: NPC203424
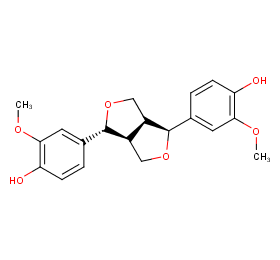
Ingredient ID: NPC200903
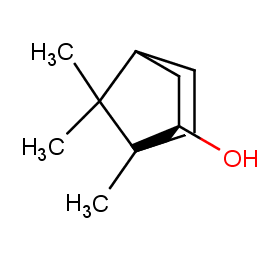
Ingredient ID: NPC196490

Ingredient ID: NPC193666

Ingredient ID: NPC19319
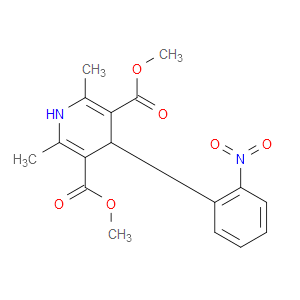
Ingredient ID: NPC190945
Ingredient ID: NPC190810
Ingredient ID: NPC189036
Ingredient ID: NPC188238

Ingredient ID: NPC187796
Ingredient ID: NPC186903
Ingredient ID: NPC186098
Ingredient ID: NPC180684
Ingredient ID: NPC180466
Ingredient ID: NPC179950
Ingredient ID: NPC178223
Ingredient ID: NPC177112
Ingredient ID: NPC173996
Ingredient ID: NPC171928
Ingredient ID: NPC168855
Ingredient ID: NPC166894
Ingredient ID: NPC166362
Ingredient ID: NPC165386
Ingredient ID: NPC164542
Ingredient ID: NPC161557

Ingredient ID: NPC158526
Ingredient ID: NPC157298
Ingredient ID: NPC156139
Ingredient ID: NPC152989

Ingredient ID: NPC152618
Ingredient ID: NPC150892
Ingredient ID: NPC150329
Ingredient ID: NPC149412
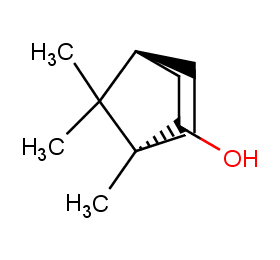
Ingredient ID: NPC14682
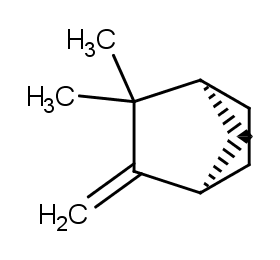
Ingredient ID: NPC141777
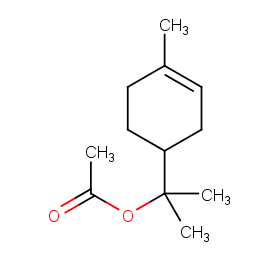
Ingredient ID: NPC141699
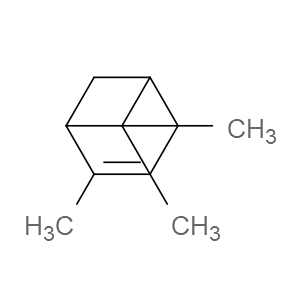
Ingredient ID: NPC13991
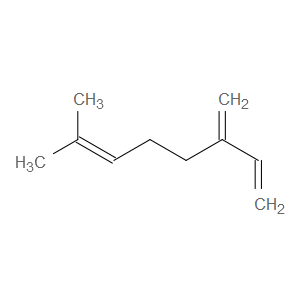
Ingredient ID: NPC138113
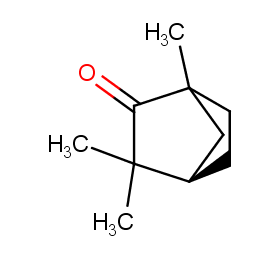
Ingredient ID: NPC136232
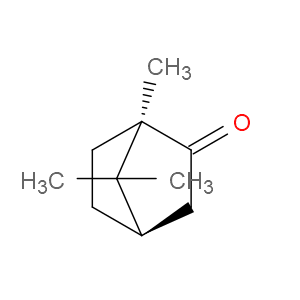
Ingredient ID: NPC131981
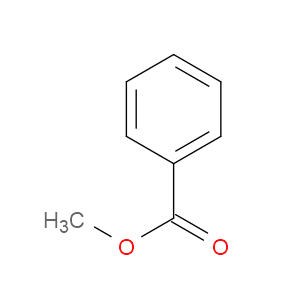
Ingredient ID: NPC130398
Ingredient ID: NPC129687
Ingredient ID: NPC128027
Ingredient ID: NPC127326
Ingredient ID: NPC123526
Ingredient ID: NPC119949
Ingredient ID: NPC119579
Ingredient ID: NPC118284
Ingredient ID: NPC115499
Ingredient ID: NPC114239
Ingredient ID: NPC113101

Ingredient ID: NPC111544
Ingredient ID: NPC105509
Ingredient ID: NPC101153

Ingredient ID: NPC100223
Classification of Human Proteins Collectively Targeted by the Plant
Detailed Information of Target Proteins
| Target Type | Protein Class | Gene ID | Protein Name | Uniprot ID | Target ChEMBL ID |
|---|---|---|---|---|---|
| Cytochrome P450 | Cytochrome P450 family 19 | CYP19A1 | Cytochrome P450 19A1 | P11511 | CHEMBL1978 |
| Cytochrome P450 | Cytochrome P450 family 3 | CYP3A4 | Cytochrome P450 3A4 | P08684 | CHEMBL340 |
| Therapeutic Target | Enzyme | CES1 | Acyl coenzyme A:cholesterol acyltransferase | P23141 | CHEMBL2265 |
| Therapeutic Target | Enzyme | RECQL | ATP-dependent DNA helicase Q1 | P46063 | CHEMBL1293236 |
| Therapeutic Target | Enzyme | POLB | DNA polymerase beta | P06746 | CHEMBL2392 |
| Therapeutic Target | Enzyme | NOX4 | NADPH oxidase 4 | Q9NPH5 | CHEMBL1250375 |
| Therapeutic Target | Enzyme | ALOX12 | Arachidonate 12-lipoxygenase | P18054 | CHEMBL3687 |
| Therapeutic Target | Lyase | CA12 | Carbonic anhydrase XII | O43570 | CHEMBL3242 |
| Therapeutic Target | Lyase | CA4 | Carbonic anhydrase IV | P22748 | CHEMBL3729 |
| Therapeutic Target | Lyase | CA7 | Carbonic anhydrase VII | P43166 | CHEMBL2326 |
| Therapeutic Target | Nuclear hormone receptor subfamily 1 | RORC | Nuclear receptor ROR-gamma | P51449 | CHEMBL1741186 |
| Therapeutic Target | Nuclear hormone receptor subfamily 1 | NR1H4 | Bile acid receptor FXR | Q96RI1 | CHEMBL2047 |
| Therapeutic Target | Other cytosolic protein | MAPT | Microtubule-associated protein tau | P10636 | CHEMBL1293224 |
| Therapeutic Target | Oxidoreductase | HSD17B10 | Endoplasmic reticulum-associated amyloid beta-peptide-binding protein | Q99714 | CHEMBL4159 |
| Therapeutic Target | Peptide receptor (family A GPCR) | TSHR | Thyroid stimulating hormone receptor | P16473 | CHEMBL1963 |
| Therapeutic Target | Peptide receptor (family A GPCR) | NPSR1 | Neuropeptide S receptor | Q6W5P4 | CHEMBL5162 |
| Therapeutic Target | Phosphodiesterase | TDP1 | Tyrosyl-DNA phosphodiesterase 1 | Q9NUW8 | CHEMBL1075138 |
| Therapeutic Target | Protein Kinase | MAPK14 | MAP kinase p38 alpha | Q16539 | CHEMBL260 |
| Therapeutic Target | Protein Kinase | IGF1R | Insulin-like growth factor I receptor | P08069 | CHEMBL1957 |
| Therapeutic Target | Protein Kinase | MAPK1 | MAP kinase ERK2 | P28482 | CHEMBL4040 |
| Therapeutic Target | Protein Kinase | PIM1 | Serine/threonine-protein kinase PIM1 | P11309 | CHEMBL2147 |
| Therapeutic Target | Protein Kinase | MET | Hepatocyte growth factor receptor | P08581 | CHEMBL3717 |
| Therapeutic Target | Protein Kinase | MKNK2 | MAP kinase signal-integrating kinase 2 | Q9HBH9 | CHEMBL4204 |
| Therapeutic Target | Protein Kinase | CHEK2 | Serine/threonine-protein kinase Chk2 | O96017 | CHEMBL2527 |
| Therapeutic Target | Protein Kinase | PTK6 | Tyrosine-protein kinase BRK | Q13882 | CHEMBL4601 |
| Therapeutic Target | Protein Kinase | FRK | Tyrosine-protein kinase FRK | P42685 | CHEMBL4223 |
| Therapeutic Target | Protein Kinase | EGFR | Epidermal growth factor receptor erbB1 | P00533 | CHEMBL203 |
| Therapeutic Target | Protein Kinase | GAK | Serine/threonine-protein kinase GAK | O14976 | CHEMBL4355 |
| Therapeutic Target | Protein Kinase | SIK2 | Serine/threonine-protein kinase SIK2 | Q9H0K1 | CHEMBL5699 |
| Therapeutic Target | Protein Kinase | CDK1 | Cyclin-dependent kinase 1 | P06493 | CHEMBL308 |
| Therapeutic Target | Protein Kinase | LYN | Tyrosine-protein kinase Lyn | P07948 | CHEMBL3905 |
| Therapeutic Target | Protein Kinase | YES1 | Tyrosine-protein kinase YES | P07947 | CHEMBL2073 |
| Therapeutic Target | Protein Kinase | KDR | Vascular endothelial growth factor receptor 2 | P35968 | CHEMBL279 |
| Therapeutic Target | Protein Kinase | FLT3 | Tyrosine-protein kinase receptor FLT3 | P36888 | CHEMBL1974 |
| Therapeutic Target | Protein Kinase | ERBB4 | Receptor protein-tyrosine kinase erbB-4 | Q15303 | CHEMBL3009 |
| Therapeutic Target | Protein Kinase | LCK | Tyrosine-protein kinase LCK | P06239 | CHEMBL258 |
| Therapeutic Target | Protein Kinase | RIPK2 | Serine/threonine-protein kinase RIPK2 | O43353 | CHEMBL5014 |
| Therapeutic Target | Protein Kinase | IRAK1 | Interleukin-1 receptor-associated kinase 1 | P51617 | CHEMBL3357 |
| Therapeutic Target | Protein Kinase | ERBB2 | Receptor protein-tyrosine kinase erbB-2 | P04626 | CHEMBL1824 |
| Therapeutic Target | Protein Kinase | AURKB | Serine/threonine-protein kinase Aurora-B | Q96GD4 | CHEMBL2185 |
| Therapeutic Target | Protein Kinase | AXL | Tyrosine-protein kinase receptor UFO | P30530 | CHEMBL4895 |
| Therapeutic Target | Protein Kinase | MAPK10 | c-Jun N-terminal kinase 3 | P53779 | CHEMBL2637 |
| Therapeutic Target | Protein Kinase | SLK | Serine/threonine-protein kinase 2 | Q9H2G2 | CHEMBL4202 |
| Therapeutic Target | Protein Kinase | HCK | Tyrosine-protein kinase HCK | P08631 | CHEMBL3234 |
| Therapeutic Target | Serine protease | DPP4 | Dipeptidyl peptidase IV | P27487 | CHEMBL284 |
| Therapeutic Target | Small molecule receptor (family A GPCR) | ADRB1 | Beta-1 adrenergic receptor | P08588 | CHEMBL213 |
| Therapeutic Target | Small molecule receptor (family A GPCR) | DRD1 | Dopamine D1 receptor | P21728 | CHEMBL2056 |
| Therapeutic Target | Structural protein | LMNA | Prelamin-A/C | P02545 | CHEMBL1293235 |
| Therapeutic Target | Transcription factor | TP53 | Cellular tumor antigen p53 | P04637 | CHEMBL4096 |
| Therapeutic Target | Transcription factor | NFKB1 | Nuclear factor NF-kappa-B p105 subunit | P19838 | CHEMBL3251 |
| Therapeutic Target | Transcription factor | RELA | Nuclear factor NF-kappa-B p65 subunit | Q04206 | CHEMBL5533 |
| Therapeutic Target | Transcription factor | HIF1A | Hypoxia-inducible factor 1 alpha | Q16665 | CHEMBL4261 |
| Drug Transporter | Voltage-gated calcium channel | CACNA1C | Voltage-gated L-type calcium channel alpha-1C subunit | Q13936 | CHEMBL1940 |
Clinical trials associated with plant from natural product (NP) & plant level:
| Clinical trials type | Number of clinical trials | |
|---|---|---|
| 432 | ||
| NCT ID | Title | Condition | Form in clinical use | Associated by plant or compound |
|---|---|---|---|---|
| NCT01350609 | Pivotal Bioequivalence FDC Nifedipine / Candesartan vs. Loose Combination of Single Components, Fed | hypertension | Nifedipine (NPC190945) | |
| NCT00135135 | Therapy for Children With Neuroblastoma | neuroblastoma | Gefitinib (NPC56271) | |
| NCT00328562 | ZD1839 With Hypofractionated Radiation Therapy With an Immobilization Device for Advanced Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00255463 | Phase II Neoadjuvant ER+/PgR + Arimidex +/- Iressa Study | breast cancer | Gefitinib (NPC56271) | |
| NCT00020709 | Combination Chemotherapy and Radiation Therapy With or Without Gefitinib in Treating Patients With Stage III Non-Small Cell Lung Cancer That Cannot Be Removed By Surgery | lung adenocarcinoma;squamous cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00589303 | AV Node Ablation and Pacemaker Therapy Compared to Drug Therapy for Atrial Fibrillation - Pilot Study | atrial fibrillation;heart failure | Atenolol (NPC307123) | |
| NCT00357734 | Iressa Follow-up Trial | breast cancer;lung cancer | Gefitinib (NPC56271) | |
| NCT01030016 | Study of Atenolol Influence on Blood Pressure During Resistance Exercise | hypertension | Atenolol (NPC307123) | |
| NCT00095836 | Gefitinib in Treating Patients With Locally Advanced or Metastatic Thyroid Cancer That Did Not Respond to Iodine Therapy | head and neck malignant neoplasia | Gefitinib (NPC56271) | |
| NCT01830530 | HIGH Altitude CArdiovascular REsearch in the ANDES | hypertension | Nifedipine (NPC190945) | |
| NCT00593463 | Drug Discrimination in Methadone-Maintained Humans Study 1 | drug dependence | Nifedipine (NPC190945) | |
| NCT01071122 | FOCUS (Nifedipine GITS's Effect on Central Pressure Assessed by Applanation Tonometry) | hypertension | Nifedipine (NPC190945) | |
| NCT04028778 | Phase 2 Platform Study in Patients With Advanced Non-Small Lung Cancer Who Progressed on First-Line Osimertinib Therapy (ORCHARD) | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00108082 | The CLEVER Study - Coreg And Left Ventricular Mass Regression | left ventricular hypertrophy | Atenolol (NPC307123) | |
| NCT03292133 | A Study of EGF816 and Gefitinib in TKI-naïve EGFR-mutant Non-Small Cell Lung Cancer | lung cancer | Gefitinib (NPC56271) | |
| NCT00389168 | Irbesartan and Atenolol in Hypertensive Heart Disease | hypertension | Atenolol (NPC307123) | |
| NCT03887871 | Study to Evaluate the Bioequivalence of Astellas Phama Korea Inc. "Harnal-D" in Healthy Volunteers After Meal | benign prostatic hyperplasia | Tamsulosin Hydrochloride (NPC203424) | |
| NCT03989271 | Biological Effects of Quercetin in COPD | chronic obstructive pulmonary disease | Quercetin (NPC20791) | |
| NCT00807066 | Randomized Gefitinib Trial | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT01510990 | First Line Gefitinib by FDG-PET Metabolic Response | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT02088112 | MEDI4736 (Anti PD-L1) Combined With Gefitinib in Subjects With Non-Small Cell Lung Cancer(NSCLC). | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00088907 | Phase III Trial Of Docetaxel Versus Docetaxel Plus ZD1839 In Head And Neck Cancer | head and neck malignant neoplasia | Gefitinib (NPC56271) | |
| NCT00025207 | Gefitinib in Treating Patients With Malignant Mesothelioma | mesothelioma | Gefitinib (NPC56271) | |
| NCT00234429 | A Phase 2 Study of Tomudex & Iressa as Second Line Chemotherapy in Subjects With Colorectal Carcinoma | colorectal carcinoma | Gefitinib (NPC56271) | |
| NCT02641821 | Effect of Nifedipine GITS in Patients With Mild-to-moderate Primary Hypertension | hypertension | Nifedipine (NPC190945) | |
| NCT04239833 | ASK120067 Versus Gefitinib as First-line Treatment for EGFRm Locally Advanced or Metastatic NSCLC | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT02560805 | Role of Sympathetic Overactivity and Angiotensin II in PTSD and CV | post-traumatic stress disorder | Atenolol (NPC307123) | |
| NCT02441842 | Evaluation of Periop Biochemical Stress Factors in Craniotomy Neurosurgical Procedure With Respect to Preop Hypertension | hypertension | Atenolol (NPC307123) | |
| NCT00428896 | A Feasibility Study With Iressa in Resistant Cytokeratin-Positive Tumor Cells Circulating in the Blood of Women With Breast Cancer | breast cancer | Gefitinib (NPC56271) | |
| NCT01864681 | Combination of Metformin With Gefitinib to Treat NSCLC | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00100945 | Gefitinib in Treating Patients With Locally Advanced Esophageal Cancer | esophageal cancer | Gefitinib (NPC56271) | |
| NCT00340704 | PK/PD, Long-term Safety and Efficacy of Tamsulosin Treatment in Children With Neurogenic Bladder | Neurogenic bladder | Tamsulosin Hydrochloride (NPC203424) | |
| NCT00096486 | Gefitinib and Everolimus in Treating Patients With Stage IIIB or Stage IV or Recurrent Non-Small Cell Lung Cancer | lung cancer | Gefitinib (NPC56271) | |
| NCT01498562 | Phase II Study of Gefitinib Plus Nimotuzumab Versus Gefitinib in Non-small Cell Lung Cancer | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT01017874 | A Study of Alimta/Cisplatin/Gefitinib for Asian Non-smoking Participants With Non Small Cell Lung Cancer | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00206492 | Trial of ZD1839 (Iressa) and Tamoxifen in Breast Cancer Patients | breast cancer | Gefitinib (NPC56271) | |
| NCT01196234 | Paclitaxel/Carboplatin (PC) Followed by Gefitinib Versus PC in Advanced Non-small Cell Lung Cancer | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00025116 | ZD 1839 in Treating Patients With Prostate Cancer That Has Not Responded to Hormone Therapy | prostate cancer | Gefitinib (NPC56271) | |
| NCT03949478 | Treating Postictal Symptoms Using Ibuprofen and Nifedipine | partial epilepsy | Nifedipine (NPC190945) | |
| NCT01755923 | Gefitinib or Docetaxel as Second Line Therapy for Wild-type Epidermal Growth Factor Receptor (EGFR) NSCLC | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT03542552 | Nifedipine Versus Magnesium Sulfate for Prevention of Preterm Labor in Symptomatic Placenta Previa | placenta praevia | Nifedipine (NPC190945) | |
| NCT02447419 | Study to Evaluate the Safety and Efficacy of Gefitinib, in Subjects With EFGR Amplification Refractory Solid Tumors | neoplasm | Gefitinib (NPC56271) | |
| NCT01933347 | Third-line Treatment of Gefitinib in NSCLC Patients | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT02342275 | Efficacy and Safety of Propranolol Versus Atenolol on the Proliferative Phase of Infantile Hemangioma | hemangioma | Atenolol (NPC307123) | |
| NCT00234468 | Iressa vs Placebo as Maintenance Therapy in Locally Advanced NSCLC | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00993109 | Low-dose Nifedipine-Valsartan Combination Compared to Up-titrated Valsartan Monotherapy in Essential Hypertension | hypertension | Nifedipine (NPC190945) | |
| NCT05422885 | Safety and Feasibility of Dasatinib and Quercetin in Adults at Risk for Alzheimer's Disease | aging | Quercetin (NPC20791) | |
| NCT00246974 | Phase II Gemcitabine + Cisplatin +/- Iressa Bladder CCT | urinary bladder cancer | Gefitinib (NPC56271) | |
| NCT01466660 | LUX-Lung 7: A Phase IIb Trial of Afatinib(BIBW2992) Versus Gefitinib for the Treatment of 1st Line EGFR Mutation Positive Adenocarcinoma of the Lung | lung neoplasm | Gefitinib (NPC56271) | |
| NCT01066195 | Pemetrexed (ALIMTA) and Gefitinib (IRESSA®) in Never-Smoker and Adenocarcinoma Patients With Non-Small Cell Lung Cancer Previously Treated With Platinum-Based Chemotherapy | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT04143607 | Gefitinib in Combination With Anlotinib or Placebo in Previously Untreated EGFR-mutant NSCLC | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT02296125 | Tremelimumab) With a Sequential Switch to a 2nd IMT (MEDI4736) in Patients With Locally Advanced or Metastatic Non-Small-Cell Lung Cancer | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00087334 | Capecitabine, Oxaliplatin, and Gefitinib in Treating Patients With Metastatic Colorectal Cancer | colorectal carcinoma | Gefitinib (NPC56271) | |
| NCT00206414 | Arimidex/Faslodex/Iressa Study: A Trial Using Arimidex, Faslodex and Iressa in Women With Breast Cancer | breast cancer | Gefitinib (NPC56271) | |
| NCT01912820 | Effect of Quercetin on Green Tea Polyphenol Uptake in Prostate Tissue From Patients With Prostate Cancer Undergoing Surgery | prostate adenocarcinoma | Quercetin (NPC20791) | |
| NCT02527109 | The Effect of Intra-anal Nifedipine, Used As Add-on to Conservative Therapy, on Pain in Patients With Anal Fissure | Anal fissure | Nifedipine (NPC190945) | |
| NCT02824458 | PRe-Operative Gefitinib in Resectable EGFR Mutation Positive Lung Cancer With Sector Sequencing for Biomarker Discovery | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00145210 | Safety and Efficacy of Nebivolol in the Treatment of Hypertension in African Americans | hypertension | Atenolol (NPC307123) | |
| NCT00000478 | Asymptomatic Cardiac Ischemia Pilot (ACIP) Study | Myocardial Ischemia | Nifedipine (NPC190945);Atenolol (NPC307123) | |
| NCT00083057 | Gefitinib, Paclitaxel, and Radiation Therapy in Treating Patients With Advanced or Recurrent Squamous Cell Carcinoma of the Head and Neck | head and neck malignant neoplasia | Gefitinib (NPC56271) | |
| NCT03866499 | Abivertinib Maleate Versus Geifitinib in Patients With Advanced Non-small Cell Lung Cancer With Sensitive EGFR Mutation | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00026364 | ZD 1839 Plus Combination Chemotherapy in Treating Patients With Locally Advanced, Locally Recurrent, or Metastatic Colorectal Cancer | colorectal carcinoma | Gefitinib (NPC56271) | |
| NCT01158170 | Prophylactic Cranial Irradiation in Erlotinib/Gefitinib-responders With Non-small Cell Lung Cancer (NSCLC) (RT1001) | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT01544179 | A Study of IRESSA Treatment Beyond Progression in Addition to Chemotherapy Versus Chemotherapy Alone | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00049556 | Gefitinib in Treating Patients With Cervical Cancer | cervical cancer;fallopian tube cancer;ovarian cancer;peritoneum cancer | Gefitinib (NPC56271) | |
| NCT00223717 | Treatment of Supine Hypertension in Autonomic Failure | hypertension | Tamsulosin Hydrochloride (NPC203424) | |
| NCT03122717 | Apatinib Combine With EGFR-TKI for Advanced EGFR-TKI-resistant Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00411047 | Gefitinib in Treating Patients With Previously Untreated Stage IIIB or Stage IV Non-Small Cell Lung Cancer | lung cancer | Gefitinib (NPC56271) | |
| NCT00062062 | Gefitinib With or Without Carboplatin and Paclitaxel in Treating Older Patients With Unresectable or Metastatic Non-Small Cell Lung Cancer | lung cancer | Gefitinib (NPC56271) | |
| NCT01404260 | Intercalating and Maintenance Use of Iressa Versus Chemotherapy in Selected Advanced Non Small Cell Lung Cancer | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT01310855 | Cediranib Maleate With or Without Gefitinib in Treating Patients With Recurrent or Progressive Glioblastoma | glioblastoma multiforme | Gefitinib (NPC56271) | |
| NCT00169221 | Post-operative Radiotherapy With Cisplatin Alone or in Combination With Iressa in Upper Aerodigestive Tract Carcinomas | head and neck malignant neoplasia | Gefitinib (NPC56271) | |
| NCT00084786 | Gefitinib and Docetaxel in Treating Patients With Advanced Solid Tumors | neoplasm | Gefitinib (NPC56271) | |
| NCT00066378 | Anastrozole With or Without Gefitinib in Treating Postmenopausal Women With Metastatic or Locally Recurrent Breast Cancer | breast cancer | Gefitinib (NPC56271) | |
| NCT00014170 | Gefitinib in Treating Patients With Newly Diagnosed Glioblastoma Multiforme | gliosarcoma | Gefitinib (NPC56271) | |
| NCT00103051 | Gemcitabine, Cisplatin, and Gefitinib in Treating Patients Who Are Undergoing Surgery for Stage III Non-Small Cell Lung Cancer | lung cancer | Gefitinib (NPC56271) | |
| NCT02303769 | Study to Investigate the Efficacy and Safety of GL2702 GLARS-NF1tablet and Harnal-D - Tablet in BPH Patients With LUTS | benign prostatic hyperplasia | Tamsulosin Hydrochloride (NPC203424) | |
| NCT03239015 | Efficacy and Safety of Precision Therapy in Refractory Tumor | neoplasm | Gefitinib (NPC56271) | |
| NCT02226484 | Can Quercetin Increase Claudin-4 and Improve Esophageal Barrier Function in GERD? | gastroesophageal reflux disease | Quercetin (NPC20791) | |
| NCT01788358 | Open-Label Long-Term Safety and Efficacy Study of Fixed Dose Combination of Nifedipine Gastrointestinal Therapeutic System and Candesartan Cilexetil in Subjects With Moderate to Severe Essential Hypertension | hypertension | Nifedipine (NPC190945) | |
| NCT01513174 | Study With Gefitinib in Combination With Olaparib (AZD2281) Versus Gefitinib Alone | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT01203917 | Efficacy, Safety, Tolerability of Gefitinib as 1st Line in Caucasian Patients With EGFR Mutation Positive Advanced NSCLC | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT01604811 | Exploratory Study of L.S.E.S.r. (LipidoSterolic Extract of Serenoa Repens)(PERMIXON® 160 mg Hard Capsule) Versus Tamsulosine LP Activity on Inflammation Biomarkers in Urinary Symptoms Related to BPH (Benign Prostatic Hyperplasia) | benign prostatic hyperplasia | Tamsulosin Hydrochloride (NPC203424) | |
| NCT01037998 | UFUR (Tegafur/Uracil) Plus Iressa in Non-small-cell Lung Cancer | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT01518855 | Controlled Release Nifedipine and Valsartan Combination Therapy in Patients With Essential Hypertension | hypertension | Nifedipine (NPC190945) | |
| NCT00040781 | Gefitinib in Treating Children With Refractory Solid Tumors | neoplasm | Gefitinib (NPC56271) | |
| NCT01829893 | PK Comparison of GL2701 With Finasteride and Tamsulosin in Combination | benign prostatic hyperplasia | Tamsulosin Hydrochloride (NPC203424) | |
| NCT00212108 | A Study Of Nasopharyngeal Carcinoma (NPC) Treated With Celecoxib And ZD1839 | nasopharyngeal neoplasm | Gefitinib (NPC56271) | |
| NCT00502060 | Phase I Study of AZD2171 co-Administered With Fixed Multiple Oral Doses of ZD1839 in Patients With Advanced Cancer | neoplasm | Gefitinib (NPC56271) | |
| NCT00049543 | Gefitinib in Treating Patients With Stage IB, II, or IIIA Non-small Cell Lung Cancer That Was Completely Removed by Surgery | bronchoalveolar adenocarcinoma | Gefitinib (NPC56271) | |
| NCT00040794 | Combination Chemotherapy, Radiation Therapy, and Gefitinib in Treating Patients With Stage III Non-Small Cell Lung Cancer | lung adenocarcinoma | Gefitinib (NPC56271) | |
| NCT03333343 | Gefitinib With Chemotherapy or Anti-angiogenesis in NSCLC Patients With Bim Deletion or Low EGFR Mutation Abundance | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT02513927 | The Effects of Nifedipine and Metoprolol on Blood Pressure Variability in Northern Chinese | hypertension | Nifedipine (NPC190945) | |
| NCT00158093 | A Safety Evaluation of ECG Intervals and Blood Pressure in Normal Healthy Volunteers After Use of Nebivolol, Atenolol, Moxifloxacin, or Placebo | hypertension | Atenolol (NPC307123) | |
| NCT02893332 | Hypofractionated Brain Radiation In EGFR Mutated Adenocarcinoma Cranial Disease (Hybrid) | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT04887818 | Diltiazem Ointment With Lidocaine vs. Nifedipine Ointment With Lidocaine Ointment for Treatment of Chronic Anal Fissure | anus disease | Nifedipine (NPC190945) | |
| NCT00497250 | Phase I Study of Iressa and CRT/IMRT in Chinese Patients With IIIB/IV NSCLC After Failure of Platinum-Based Chemotherapy | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00455416 | Dietary Intervention in Follicular Lymphoma | follicular lymphoma | Quercetin (NPC20791) | |
| NCT01715207 | Comparison of Aliskiren vs Negative Controls on Aortic Stiffness in Patients With MFS | Marfan syndrome | Atenolol (NPC307123) | |
| NCT03943459 | Sirtuin-1 and Advanced Glycation End-products in Postmenopausal Women With Coronary Disease | coronary artery disease | Quercetin (NPC20791) | |
| NCT01119534 | Comparative Efficacy of the Suppository Versus Guaiacol Suppository Versus Guaifenesin Syrup in Pediatric Patients With Cough Due the Infectious Origin | Cough | Eucalyptol (NPC287331) | |
| NCT00130702 | Study of Iressa in Patients With Relapsed or Refractory Acute Myelogenous Leukemia | acute myeloid leukemia | Gefitinib (NPC56271) | |
| NCT00234403 | A Trial to Evaluate the Combination of Iressa & Faslodex® in Patients With Advanced or Metastatic Breast Cancer | breast cancer | Gefitinib (NPC56271) | |
| NCT00239304 | Gefitinib (Iressa) in Combo With Chemoradiation in Patients With Locally Advanced Head & Neck Cancer | upper aerodigestive tract neoplasm | Gefitinib (NPC56271) | |
| NCT00052208 | Gefitinib and Radiation Therapy in Treating Patients With Glioblastoma Multiforme | gliosarcoma | Gefitinib (NPC56271) | |
| NCT00536107 | Study to Assess Safety/Tolerability/Efficacy of Gefitinib Versus Docetaxel in Locally Advanced or Metastatic Non-Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00080743 | Gefitinib With or Without Tamoxifen in Treating Patients With Tamoxifen-Resistant Metastatic Breast Cancer | breast cancer | Gefitinib (NPC56271) | |
| NCT00185185 | Olmesartan Medoxomil in Atherosclerosis | essential hypertension | Atenolol (NPC307123) | |
| NCT00238797 | A Phase II Exploratory, Multicentre, Open-label, Non-comparative Study of ZD1839 (Iressa) and Radiotherapy in the Treatment of Patients With Glioblastoma Multiforme | glioblastoma multiforme | Gefitinib (NPC56271) | |
| NCT00483561 | Gefitinib and Etoposide in Treating Patients With Advanced Prostate Cancer That Did Not Respond to Hormone Therapy | prostate cancer | Gefitinib (NPC56271) | |
| NCT03599518 | Phase III Study of Nazartinib (EGF816) Versus Erlotinib/Gefitinib in First-line Locally Advanced / Metastatic NSCLC With EGFR Activating Mutations | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT02804776 | A Study of ASP8273 vs. Erlotinib or Gefitinib in First-line Treatment of Patients With Stage IIIB/IV Non-small Cell Lung Cancer Tumors With EGFR Activating Mutations | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00113529 | Study Of SU011248 Plus Gefitinib (Iressa) In Patients With Advanced Renal Cell Carcinoma | renal cell carcinoma | Gefitinib (NPC56271) | |
| NCT02940548 | Nifedipine GITS and Amlodipine Besylate on Recovery of Blood Pressure Rhythm and Arterial Stiffness | hypertension | Nifedipine (NPC190945) | |
| NCT00713739 | Alfuzosin for Medical Expulsion Therapy of Ureteral Stones | nephrolithiasis | Nifedipine (NPC190945) | |
| NCT00098462 | Gefitinib and Sirolimus in Treating Patients With Recurrent or Refractory Stage IIIB or Stage IV Non-Small Cell Lung Cancer | lung cancer | Gefitinib (NPC56271) | |
| NCT00608868 | SELINE: Second-Line Iressa Phase IV Study in NSCLC Patients | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00239291 | Safety Study to Explore Combination of Gefitinib (ZD1839, Iressa) and Radiotherapy in Non-Metastatic Prostate Cancer | metastatic prostate cancer | Gefitinib (NPC56271) | |
| NCT00252707 | Iressa 2nd Line Phase III Study in Japan | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT03050411 | A Phase II Study of Fruquintinib in Combination With Gefitinib in Patients With Advanced Non-small Cell Lung Cancer | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00310154 | Gefitinib, Docetaxel, and Radiation Therapy in Treating Patients With Stage III Non-Small Cell Lung Cancer | lung cancer | Gefitinib (NPC56271) | |
| NCT00857090 | Pharmacokinetic, Safety and Efficacy Study of OMS201 in Subjects Undergoing Retrograde Ureteroscopic Removal of Upper Urinary Tract Stones | nephrolithiasis | Nifedipine (NPC190945) | |
| NCT00039390 | Gefitinib and Capecitabine in Treating Patients With Advanced Solid Tumors | neoplasm | Gefitinib (NPC56271) | |
| NCT03758287 | To Evaluate the Efficacy and Safety of Gefitinib in Adjuvant Chemotherapy for Lung Adenocarcinoma | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT03849768 | Alflutinib Mesylate Versus Gefitinib in Patients With Locally Advanced or Metastatic Non-small Cell Lung Cancer (FLAG) | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT01732276 | The Safety and Effects of Gefitinib in Triple-negative,EGFR Positive Metastatic Breast Cancer | breast cancer | Gefitinib (NPC56271) | |
| NCT00635856 | Open Label Trial to Assess Iressa in Prostate Cancer Patients | prostate cancer | Gefitinib (NPC56271) | |
| NCT00171132 | VALENCE: Valsartan Versus Atenolol on Exercise Capacity in Hypertensive Overweight Postmenopausal Women | hypertension | Atenolol (NPC307123) | |
| NCT00290719 | Gefitinib, Cisplatin, Irinotecan, and Radiation Therapy Before Surgery in Treating Patients With Esophageal Cancer or Gastroesophageal Junction Cancer That Can Be Removed By Surgery | esophageal cancer | Gefitinib (NPC56271) | |
| NCT00198380 | Treatment of Lung Adenocarcinoma With Bronchioloalveolar Feature | lung adenocarcinoma | Gefitinib (NPC56271) | |
| NCT01589263 | Effectiveness of onaBoNT-A vs Oral Tamsulosin in Men With BPH and LUTS | benign prostatic hyperplasia | Tamsulosin Hydrochloride (NPC203424) | |
| NCT00669279 | Effect of Carvedilol Controlled-Release (CR) and Atenolol on Central Blood Pressure | hypertension | Atenolol (NPC307123) | |
| NCT01017679 | Gefitinib 500mg Versus 250mg in Patients With NSCLC With Stable Disease After a Month Treatment of Gefitinib 250mg | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT01796522 | Utility of Tocolytic Therapy for Maintenance Tocolysis in the Management of Threatened Preterm Delivery | premature birth | Nifedipine (NPC190945) | |
| NCT01313702 | Single Pill to Avert Cardiovascular Events | cardiovascular disease | Atenolol (NPC307123) | |
| NCT01251146 | A Randomized Controlled Study to Assess the Effects of Bisoprolol and Atenolol on Resting Heart Rate and Sympathetic Nervous System's Activity in Subjects With Essential Hypertension | hypertension | Atenolol (NPC307123) | |
| NCT04853082 | Limonene on Regulating Metabolism-related Fatty Liver Disease (MAFLD) and Analysis of TCM Constitution | Hepatic steatosis | Limonene (NPC76145) | |
| NCT00186979 | Study of ZD1839 Combined With Irinotecan and Vincristine in Pediatric Patients With Refractory Solid Tumors | neoplasm | Gefitinib (NPC56271) | |
| NCT00772499 | Vascular Improvement With Olmesartan Medoxomil Study | hypertension | Atenolol (NPC307123) | |
| NCT00014144 | S0031, ZD 1839 in Treating Patients With Advanced Cancer of the Urinary Tract | urethra cancer;urinary bladder cancer | Gefitinib (NPC56271) | |
| NCT00478049 | Iressa as Second Line Therapy in Advanced NSCLC-Asia | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT01291823 | Gefitinib Combined With Radiotherapy in Elderly Patients With Esophageal Cancer | esophageal cancer | Gefitinib (NPC56271) | |
| NCT01192243 | Study of Gefitinib Compared With Pemetrexed/Cisplatin in Advanced Non-Small Cell Lung Cancer Patients | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT02039674 | A Study of Pembrolizumab (MK-3475) in Combination With Chemotherapy or Immunotherapy in Participants With Non-small Cell Lung Cancer (MK-3475-021/KEYNOTE-021) | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT03216876 | A Study Of Ursolic Acid For Primary Sclerosing Cholangitis | sclerosing cholangitis | Ursolic Acid (NPC51700) | |
| NCT00452244 | Gefitinib With or Without Simvastatin in Non-Small Cell Lung Cancer (NSCLC) | lung cancer | Gefitinib (NPC56271) | |
| NCT00409006 | Chemotherapy for Patients With Non-Small Cell Lung Cancer Who Are Non-Smokers | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT03374280 | Study of EGF816 in Combination With Selected Targeted Agents in EGFR-mutant NSCLC | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT05432284 | Behavioral Pharmacology of THC and Beta-Myrcene | cannabis dependence | Myrcene (NPC138113) | |
| NCT04907253 | Quercetin in Coronary Artery By-pass Surgery | coronary artery disease | Quercetin (NPC20791) | |
| NCT00177242 | Study of Gefitinib and Docetaxel as Salvage Therapy in Advanced Pancreatic Carcinoma | cancer | Gefitinib (NPC56271) | |
| NCT00809237 | Hydroxychloroquine and Gefitinib to Treat Lung Cancer | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00429364 | Comparison of Two Medications Aimed at Slowing Aortic Root Enlargement in Individuals With Marfan Syndrome--Pediatric Heart Network | Marfan syndrome | Atenolol (NPC307123) | |
| NCT00322452 | First Line IRESSA™ Versus Carboplatin/Paclitaxel in Asia | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT03529084 | S-1 Plus Gefitinib Versus Gefitinib Monotherapy in Patients With EGFR-sensitive Mutation Advanced Non-squamous NSCLC | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00016991 | ZD 1839 in Treating Patients With Glioblastoma Multiforme in First Relapse | Central Nervous System Neoplasm | Gefitinib (NPC56271) | |
| NCT00015964 | ZD 1839 in Treating Patients With Metastatic or Recurrent Cancer of the Head and Neck | head and neck malignant neoplasia | Gefitinib (NPC56271) | |
| NCT00651235 | A Randomized, Open-label, LOSARTAN Therapy on the Progression of Aortic Root Dilation in Patients With Marfan Syndrome | Marfan syndrome | Atenolol (NPC307123) | |
| NCT00319787 | Combination Casodex® and Iressa™ in Locally Advanced Prostate Cancer | prostate cancer | Gefitinib (NPC56271) | |
| NCT00173667 | A Study of Nifecardia SRFC and Adalat OROS in the Treatment of Patients With Essential Hypertension | hypertension | Nifedipine (NPC190945) | |
| NCT02151721 | Phase I of Vorinostat-Iressa Combined Therapy on Resistance by BIM Polymorphysim in EGFR Mutant Lung Cancer | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00295542 | Ambulatory Blood Pressure Monitoring in the Prediction of Cardiovascular Events and Effects of Chronotherapy | hypertension | Atenolol (NPC307123) | |
| NCT04403568 | Testing the Synergism of Phytonutrients, Curcumin and Ursolic Acid, to Target Molecular Pathways in the Prostate | prostate cancer | Ursolic Acid (NPC51700) | |
| NCT01982955 | Tepotinib With Gefitinib in Participants With Locally Advanced or Metastatic NSCLC (INSIGHT) | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT01556191 | Lung Cancer in Women Treated With Anti-oestrogens anD Inhibitors of EGFR | lung cancer | Gefitinib (NPC56271) | |
| NCT00140959 | Losartan and HCTZ and Amlodipine vs Atenolol and Amlodipine (0954A-309)(COMPLETED) | hypertension | Atenolol (NPC307123) | |
| NCT00242749 | IRESSA in Combination With Cisplatin & Radiotherapy in Patients With Advanced Head & Neck Carcinoma | head and neck malignant neoplasia | Gefitinib (NPC56271) | |
| NCT00228488 | Effect of Iressa With/Without Concurrent Chemoradiotherapy on Tumor Gene Expression Profiles in Patients With Advanced Non-Nasopharyngeal Head and Neck Carcinoma | head and neck malignant neoplasia | Gefitinib (NPC56271) | |
| NCT00913965 | To Demonstrate the Relative Bioavailability of Atenolol Tablets | hypertension | Atenolol (NPC307123) | |
| NCT00541684 | Health Assessment Study (0954-946) | hypertension | Atenolol (NPC307123) | |
| NCT01287260 | High Dose BAYA1040_Nifedipine: a Dose-comparative Study | hypertension | Nifedipine (NPC190945) | |
| NCT00229723 | IRESSA™ (Gefitinib) With Cisplatin Plus Radiotherapy for the Treatment of Previously Untreated Unresected Late Stage III/IV Non-Metastatic Head and Neck Squamous Cell Carcinoma | neoplasm | Gefitinib (NPC56271) | |
| NCT00344773 | First-line Treatment for Adenocarcinoma Patients With Epidermal Growth Factor Receptor (EGFR) Mutation | lung carcinoma | Gefitinib (NPC56271) | |
| NCT01294592 | Comparative Efficacy of Dutasteride Plus Tamulosin With Lifestyle Advice Versus Watchful Waiting Plus Lifestyle Advice in the Management of Treatment naïve Men With Moderately Symptomatic Benign Prostatic Hyperplasia and Prostate Enlargement | benign prostatic hyperplasia | Tamsulosin Hydrochloride (NPC203424) | |
| NCT02319577 | Intercalating and Maintenance Gefitinib in Combination With Chemotherapy for Advanced EGFR-mutant NSCLC | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00000514 | Systolic Hypertension in the Elderly Program (SHEP) | cerebrovascular disorder;heart disease;hypertension | Atenolol (NPC307123) | |
| NCT00068653 | Gefitinib and Celecoxib in Treating Patients With Refractory Non-Small Cell Lung Cancer | lung cancer | Gefitinib (NPC56271) | |
| NCT02047019 | Monotherapy-Controlled Study of Nifedipine Gastrointestinal Therapeutic System and Candesartan Cilexetil in Combination in Subjects With Essential Hypertension Inadequately Controlled on Candesartan Cilexetil | hypertension | Nifedipine (NPC190945) | |
| NCT00817037 | Sitaxsentan in Proteinuric Chronic Kidney Disease | chronic kidney disease;Proteinuria;hypertension | Nifedipine (NPC190945) | |
| NCT02976116 | Gefitinib Combined With Chemotherapy or Antiangiogensis in Patients With Bim Deletion or Low EGFR Mutation Abundance | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT03237637 | Comparative Study to Evaluate the Effectiveness of Atenolol and Propranolol in the Treatment of Infantile Hemangiomas | hemangioma | Atenolol (NPC307123) | |
| NCT00242788 | IRESSA™ In Combo With Xeloda™ in Advanced Colorectal Cancer Patients After 1st-Line Chemo Failure | colorectal carcinoma | Gefitinib (NPC56271) | |
| NCT00144066 | A Randomized Phase III Trial of Chemotherapy Alone Versus Chemotherapy Followed by Gefitinib in Stage IIIB/IV Non-Small Cell Lung Cancer | lung cancer | Gefitinib (NPC56271) | |
| NCT02023047 | An Open Label Multi-Dose Study to Examine the Effect of Coated Nifedipine Suppository on Anal Fissure Pain and Healing | Anal fissure | Nifedipine (NPC190945) | |
| NCT00006049 | ZD 1839 Plus Chemotherapy in Treating Patients With Non-Small Cell Lung Cancer | lung cancer | Gefitinib (NPC56271) | |
| NCT00750113 | Study Evaluating the Efficacy of Nifedipine GITS - Telmisartan Combination in Blood Pressure Control. | hypertension | Nifedipine (NPC190945) | |
| NCT00188617 | Phase 2 Study Of Neoadjuvant Iressa Treatment In Stage 1 NSCLC | lung cancer | Gefitinib (NPC56271) | |
| NCT00137501 | Two Dose Regimens of Nifedipine for the Management of Preterm Labor | premature birth | Nifedipine (NPC190945) | |
| NCT03656393 | DS-1205c With Gefitinib for Metastatic or Unresectable Epidermal Growth Factor Receptor (EGFR)-Mutant Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT03123484 | Osimertinib and Gefitinib in EGFR Inhibitor naïve Advanced EGFR Mutant Lung Cancer | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00683306 | Open Label Extension Study With Gefitinib (IRESSA™) for Completing Trial Patients Who May Benefit From Further Treatment | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00529750 | Effect of the Angiotensin II Receptor Antagonist Irbesartan on Biochemical and Functional Markers of Endothelial Dysfunction in Patients With Hypertension | hypertension | Atenolol (NPC307123) | |
| NCT00352079 | BCG With or Without Gefitinib in Treating Patients With High-Risk Bladder Cancer | urinary bladder cancer | Gefitinib (NPC56271) | |
| NCT01579630 | Genius Study Study to Compare Efficacy and Safety of Gefitinib/ Pemetrexed With Pemetrexed Alone as Maintenance Therapy in Patients With Stage IV EGFR Mutation Negative or T790M Single Mutation Who Respond to Pemetrexed/ Platinum as First-line Therapy | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT02446795 | Isoquercetin as an Adjunct Therapy in Patients With Kidney Cancer Receiving First-line Sunitinib: a Phase I/II Trial | renal cell carcinoma | Isoquercetin (NPC179950) | |
| NCT00126555 | Gefitinib in Treating Patients Who Are Undergoing Surgery and/or Radiation Therapy for Locally Advanced or Recurrent Squamous Cell Skin Cancer | skin cancer;squamous cell carcinoma | Gefitinib (NPC56271) | |
| NCT00198393 | Randomized Phase 2 Study of 3 Therapeutic Modalities in PS 2/3 Patients With NSCLC Stage IIIB/IV | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT01185158 | A Phase II Study of 250-mg ZD1839 Monotherapy in Recurrent or Metastatic or Both Recurrent and Metastatic Squamous Cell Carcinoma | head and neck malignant neoplasia | Gefitinib (NPC56271) | |
| NCT00027625 | Gefitinib Plus Temozolomide in Treating Patients With Malignant Primary Glioma | Central Nervous System Neoplasm | Gefitinib (NPC56271) | |
| NCT03170180 | Exploratory Study of Drug Sensitivity Prediction Software (IRCR-DReSS) With Patient-derived Tumor Cells of Metastatic Gastric Cancer | stomach neoplasm | Gefitinib (NPC56271) | |
| NCT00195078 | Safety Trial of IRESSA, Cisplatin and Radiation Therapy for Patients With Head and Neck Cancer | head and neck malignant neoplasia | Gefitinib (NPC56271) | |
| NCT00723801 | Effects of Losartan Versus Atenolol on Aortic and Cardiac Muscle Stiffness in Adults With Marfan Syndrome | Marfan syndrome | Atenolol (NPC307123) | |
| NCT00972907 | An Open Label Study to Examine the Effect of Coated Nifedipine Suppository on Anal Fissure Pain and Healing in Human Subjects | Anal fissure | Nifedipine (NPC190945) | |
| NCT01762436 | Effects of Bisoprolol and Atenolol on Sympathetic Nervous Activity and Central Aortic Pressure in Patients With Essential Hypertension | hypertension | Atenolol (NPC307123) | |
| NCT00005806 | Combination Chemotherapy in Treating Patients With Advanced Non-Small Cell Lung Cancer | lung cancer | Gefitinib (NPC56271) | |
| NCT02426177 | Comparison of Tab.Labetalol and Tab.Nifedipine for Management of Postpartum Hypertension | hypertension | Nifedipine (NPC190945) | |
| NCT04552431 | Chronic Prostatitis Collaborative Research Network Clinical Trial- Ciprofloxacin and Tamsulosin | prostatitis | Tamsulosin Hydrochloride (NPC203424) | |
| NCT02244242 | Study to Evaluate the Symptomatic Relief Effects of FLOMAX® in Patients With Signs and Symptoms of Benign Prostatic Hyperplasia | benign prostatic hyperplasia | Tamsulosin Hydrochloride (NPC203424) | |
| NCT00173875 | Iressa as a First-Line Treatment in Chemonaive Patients With Inoperable Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00057941 | Anastrozole and ZD1839 Compared With Fulvestrant and ZD1839 in Postmenopausal Women w/ Metastatic Breast Cancer | breast cancer | Gefitinib (NPC56271) | |
| NCT03267654 | β-elemene Combine With EGFR-TKI for Advanced EGFR-TKI-resistant NSCLC | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT01530334 | Iressa Re-Challenge in Advanced NSCLC EGFR M+ Patients Who Responded to Gefitinib USed as 1st Line or Previous Treatment | lung cancer | Gefitinib (NPC56271) | |
| NCT01429545 | premature birth | Nifedipine (NPC190945) | ||
| NCT00486824 | Indomethacin Versus Nifedipine for Preterm Labor Tocolysis | premature birth | Nifedipine (NPC190945) | |
| NCT00238251 | Radiation Therapy Combined With Either Gefitinib or Temozolomide in Pats With NSCLC and Brain Metastases | lung cancer | Gefitinib (NPC56271) | |
| NCT00127829 | Study Evaluating Gefitinib (IRESSA®) in Subjects With Solid Malignancies That Are Locally Advanced, Recurrent or Metastatic | neoplasm | Gefitinib (NPC56271) | |
| NCT00034879 | Iressa Expanded Access Program (EAP) | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00338260 | Losartan Intervention For Endpoint Reduction in Hypertension (LIFE) Study (0954-133) | left ventricular hypertrophy;hypertension | Atenolol (NPC307123) | |
| NCT02509104 | Bioavailability Study of Fixed Dose Combination (FDC) Dutasteride and Tamsulosin Hydrochloride (HCl) Relative to One Dutasteride and One Tamsulosin HCl Tablet in Healthy Male Subjects | benign prostatic hyperplasia | Tamsulosin Hydrochloride (NPC203424) | |
| NCT02411448 | Gefitinib Combined With Thalidomide to Treat NSCLC | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT01568918 | Use of Tamsulosin to Reduce the Incidence and Duration of Postoperative Urinary Retention Following Spine Surgery | post-operative sign or symptom | Tamsulosin Hydrochloride (NPC203424) | |
| NCT01405079 | Gefitinib Versus Vinorelbine/Platinum as Adjuvant Treatment in Stage II-IIIA(N1-N2) NSCLC With EGFR Mutation | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00090675 | Maintenance ZD1839 (IRESSA®) Following Completion of Front Line, Platinum-Based, Double Chemotherapy in Subjects With Advanced or Metastatic Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT02518802 | Pemetrexed Combined With Synchronous Gefitinib as Adjuvent Therapy in Patient With EGFR Mutant Lung Adenocarcinoma | lung neoplasm | Gefitinib (NPC56271) | |
| NCT01391260 | Radiotherapy Combined With Iressa for EGFR Mutation Positive Patients With Locally Advanced Non-small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00264498 | Phase II Iressa & Carbo/Gem in NSCLC | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00455936 | First-line Gefitinib Versus Chemotherapy for Lung Adenocarcinoma in Never Smoker | lung cancer | Gefitinib (NPC56271) | |
| NCT04931108 | The Effect of Nitrendipine/Atenolol Combination on Blood Pressure Variability. | hypertension | Atenolol (NPC307123) | |
| NCT01833572 | Preoperative Gefitinib for EGFR Mutant II-IIIa NSCLC | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT02848131 | Senescence in Chronic Kidney Disease | chronic kidney disease | Quercetin (NPC20791) | |
| NCT00242918 | Safety and Effectiveness Study of Docetaxel and ZD1839 Followed by Removal of the Prostate to Treat Prostate Cancer | prostate cancer | Gefitinib (NPC56271) | |
| NCT00509002 | Iressa Study in Patients With Salivary Gland Cancer | salivary gland cancer | Gefitinib (NPC56271) | |
| NCT00252798 | ZD1839 (Iressa™) and Concurrent Chemo-Radiation in Patients With Locally Advanced Non Small Cell Lung Cancer | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00162318 | A Phase I Study of Cetuximab in Combination With Gefitinib in Patients With Advanced/Metastatic Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00770588 | Assess the Efficacy, Safety and Tolerability of Gefitinib (Iressa® 250mg) as Maintenance Therapy in Locally Advanced or Metastatic (Stage IIIB/IV) Non Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00593710 | Losartan Versus Atenolol for the Treatment of Marfan Syndrome | Marfan syndrome | Atenolol (NPC307123) | |
| NCT00084708 | Calcitriol and Gefitinib With or Without Dexamethasone in Treating Patients With Advanced Solid Tumors | neoplasm | Gefitinib (NPC56271) | |
| NCT00054691 | ZD1839 (Iressa) for Recurrent or Metastatic Squamous Cell Carcinoma of the Skin | skin cancer | Gefitinib (NPC56271) | |
| NCT03399669 | A Phase III, Randomized, Multi-center Study to Determine the Efficacy of the Intercalating Combination Treatment of Chemotherapy and Gefitinib or Chemotherapy as Adjuvant Treatment in NSCLC With Common EGFR Mutations. | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00258323 | Radiotherapy,Chemotherapy,Before and After Surgery in Advanced Esophageal or Gastroesophageal Junction Cancer | esophageal cancer | Gefitinib (NPC56271) | |
| NCT00637026 | Ph II Early BC Pre-Surgical Biologic Study | breast cancer | Gefitinib (NPC56271) | |
| NCT00206219 | Head and Neck Phase III Iressa Versus Methotrexate Refractory: Iressa Versus Methotrexate (IMEX) | head and neck squamous cell carcinoma | Gefitinib (NPC56271) | |
| NCT01610336 | A Safety and Efficacy Study of INC280 and Gefitinib in Patients With EGFR Mutated, c-MET-amplified NSCLC Who Have Progressed After EGFRi Treatment | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT04987892 | Investigating Medication vs. Prostatic Urethral Lift: Assessment and Comparison of Therapies for BPH | benign prostatic hyperplasia | Tamsulosin Hydrochloride (NPC203424) | |
| NCT00231465 | Phase II Study of Docetaxel + ZD1839 in Elderly Patients With Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT02029118 | Acupoint Application in Patients With Stable Angina Pectoris (AASAP) | angina pectoris | Borneol (NPC283655) | |
| NCT04404686 | Vaginal Indomethacin for Preterm Labor | premature birth | Nifedipine (NPC190945) | |
| NCT02374645 | Induction Therapy With Intercalated Tyrosine Kinase Inhibitor (TKI) and Chemotherapy in NSCLC With Activating Epidermal Growth Factor Receptor (EGFR) Mutation in Stages II-IIIB | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT01720147 | Quercetin in Children With Fanconi Anemia; a Pilot Study | Fanconi anemia | Quercetin (NPC20791) | |
| NCT01749072 | Vinorelbine-ifosfamide Versus Gefitinib for EGFR Gene Mutation Negative Non-small Cell Lung Cancer Patients | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00217698 | Gefitinib as First-Line Therapy Followed by Gemcitabine and Cisplatin in Treating Patients With Stage III or Stage IV Non-Small Cell Lung Cancer | lung cancer | Gefitinib (NPC56271) | |
| NCT00986284 | Epidermal Growth Factor Receptor (EGFR) Status Based Gefitinib Neoadjuvant Therapy in Non Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00317772 | Topotecan and Gefitinib (Iressa) for Ovarian, Peritoneal, or Fallopian Tube Cancer | fallopian tube cancer;ovarian cancer;peritoneal neoplasm | Gefitinib (NPC56271) | |
| NCT01708278 | Beneficial Effects of Quercetin in Chronic Obstructive Pulmonary Disease (COPD) | chronic obstructive pulmonary disease | Quercetin (NPC20791) | |
| NCT00255489 | A Phase I Study of ZD1839 and Palliative Thoracic Radiotherapy in Patients With Non-small-cell Lung Cancer | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT01024712 | Paclitaxel, Carboplatin, and Gefitinib in Treating Patients With Advanced Non-Small Cell Lung Cancer | lung cancer | Gefitinib (NPC56271) | |
| NCT00052754 | Gefitinib in Treating Patients With Locally Advanced or Metastatic Synovial Sarcoma | sarcoma | Gefitinib (NPC56271) | |
| NCT01360034 | Nifedipine Versus Indomethacin in the Treatment of Preterm Labour | premature birth | Nifedipine (NPC190945) | |
| NCT03887858 | Study to Evaluate the Bioequivalence of Astellas Phama Korea Inc. "Harnal-D" in Healthy Volunteers | benign prostatic hyperplasia | Tamsulosin Hydrochloride (NPC203424) | |
| NCT00519077 | Phase II Study of Skin Toxicity Dosing of IRESSA (Gefitinib) in Squamous Cell Carcinoma of the Head and Neck | upper aerodigestive tract neoplasm | Gefitinib (NPC56271) | |
| NCT00234416 | IRESSA Combined With Radiotherapy & Gemcitabine as First-Line Treatment in Locally Advanced Pancreatic Cancer | pancreatic carcinoma | Gefitinib (NPC56271) | |
| NCT00256711 | Phase II Iressa Versus Vinorelbine (INVITE) | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00066339 | Gefitinib in Treating Patients With Metastatic Breast Cancer That Has Progressed After Antiestrogen and Nonsteroidal Aromatase Inhibitor Therapy | breast cancer | Gefitinib (NPC56271) | |
| NCT04853199 | Quercetin In The Treatment Of SARS-COV 2 | COVID-19 | Quercetin (NPC20791) | |
| NCT03040973 | Study to Allow Patients Previously Participating in a Novartis Sponsored Trial to Continue Receiving Capmatinib Treatment as Single Agent or in Combination With Other Treatments or the Combination Treatment Alone | neoplasm | Gefitinib (NPC56271) | |
| NCT00661895 | Black Education and Treatment of Hypertension (BEAT HTN) | hypertension | Atenolol (NPC307123);Nifedipine (NPC190945) | |
| NCT00025675 | Gefitinib in Treating Patients With Recurrent or Progressive CNS Tumors | Central Nervous System Neoplasm | Gefitinib (NPC56271) | |
| NCT00352105 | Cisplatin, Fluorouracil, Iressa, and Radiation Therapy Patients With Locally Advanced Head and Neck Cancer | head and neck malignant neoplasia | Gefitinib (NPC56271) | |
| NCT01254071 | A Study to Determine the Bioavailability of a Fixed Dose Combination Product of Dutasteride (0.5mg) and Tamsulosin Hydrochloride (0.2mg) Relative to Co-administration of the Individual Components in Healthy Male Subjects of North East Asian and Non-Asian Ancestry. | benign prostatic hyperplasia | Tamsulosin Hydrochloride (NPC203424) | |
| NCT02031601 | Intercalated Combination of Chemotherapy and Tyrosine Kinase Inhibitors as First-line Treatment for Patients With Non-Small-Cell Lung Cancer | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00232882 | Pharmacodynamic Influences of Candesartan, Atenolol, Hydrochlorothiazide and Drug Combinations in Hypertensive Patients. | hypertension | Atenolol (NPC307123) | |
| NCT00189358 | A Phase II Study of ZD1839 and Tamoxifen in Patients With Epithelial Ovarian Carcinoma, Cancer of the Fallopian Tube or the Peritoneum Refractory to Platinum- and Taxane-based Therapy | Fallopian Tube Carcinoma;ovarian cancer;peritoneum cancer | Gefitinib (NPC56271) | |
| NCT01839344 | Effects of Quercetin on Blood Sugar and Blood Vessel Function in Type 2 Diabetes. | type 2 diabetes mellitus | Quercetin (NPC20791) | |
| NCT00049062 | Anastrozole and ZD 1839 in Treating Postmenopausal Women With Metastatic Breast Cancer | breast cancer | Gefitinib (NPC56271) | |
| NCT00193336 | Iressa in Poor Performance Status Patients With Previously Untreated Advanced Non-Small Cell Lung Cancer | lung cancer | Gefitinib (NPC56271) | |
| NCT00588445 | Phase II Trial to Correlate Radiographic Response Induced By Gefitinib With Mutations in the Protein-Tyrosine Kinase Domain of the EGF Receptor Gene | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT02321293 | A Open-label Prospective Cohort Trial of Curcumin Plus Tyrosine Kinase Inhibitors (TKI) for EGFR -Mutant Advanced NSCLC | lung cancer | Gefitinib (NPC56271) | |
| NCT00027690 | Gefitinib in Treating Patients With Persistent or Recurrent Endometrial Cancer | uterine neoplasm | Gefitinib (NPC56271) | |
| NCT03381066 | Pemetrexed/Cisplatin Intercalating Gefitinib Treating EGFR Wild NSCLC | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00062270 | Neoadjuvant Chemoradiotherapy With or Without Gefitinib in Treating Patients With Stage IIIA or Stage IIIB Non-Small Cell Lung Cancer | lung cancer | Gefitinib (NPC56271) | |
| NCT01985594 | Utrogestan Versus Nifedipine as Tocolysis for Preterm Labor: a Randomised Controlled Trial | premature birth | Nifedipine (NPC190945) | |
| NCT00239343 | Phase II Trial of Preoperative Therapy With Gefitinib and Chemotherapy in Patients With ERneg Breast Cancer | breast cancer | Gefitinib (NPC56271) | |
| NCT00229697 | Phase II Metastatic ER+/PgR+ Nolvadex +/- Iressa Study | breast neoplasm | Gefitinib (NPC56271) | |
| NCT00796614 | Study to Evaluate the Efficacy and Safety of Tamsulosin in Children With Neurogenic Bladder | Neurogenic bladder | Tamsulosin Hydrochloride (NPC203424) | |
| NCT00955695 | Radiation Therapy to the Brain or Observation in Preventing Brain Metastases in Patients With Advanced Non-Small Cell Lung Cancer | lung cancer | Gefitinib (NPC56271) | |
| NCT01485809 | Efficacy and Safety Study of Gefitinib in Squamous NSCLC Patients Who Failed First-Line Chemotherapy | squamous cell carcinoma | Gefitinib (NPC56271) | |
| NCT04733651 | Study to Investigate the Clinical Efficacy of Isoquercetin in Patients With COVID-19 | COVID-19 | Isoquercetin (NPC179950) | |
| NCT01955421 | Erlotinib 100mg qd Versus Gefitinib 250mg qd for EGFR Mutant Nsclc | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00000530 | Raynaud's Treatment Study (RTS) | Raynaud disease | Nifedipine (NPC190945) | |
| NCT03944772 | A Study of BPI-7711 Capsule in Non-small Cell Lung Cancer Patients | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00681967 | Postoperative Radiotherapy Plus Iressa or Radiotherapy Plus Cisplatin and Iressa for Advanced Head & Neck Cancer | head and neck malignant neoplasia | Gefitinib (NPC56271) | |
| NCT02856893 | A Study of Gefitinib With or Without Apatinib in Patients With Advanced Non-squamous Non-Small-Cell Lung Cancer Harboring EGFR Mutations | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00082667 | Gefitinib Followed By Surgery in Treating Women With Ductal Carcinoma In Situ of the Breast | breast cancer | Gefitinib (NPC56271) | |
| NCT04248829 | A Study of SH-1028 Tablets Versus Gefitinib in Patients With Locally Advanced or Metastatic Non-small Cell Lung Cancer | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT04536090 | Study of Isoquercetin (IQC-950AN) Plus Standard of Care Versus Standard of Care Only for the Treatment of COVID-19 | COVID-19 | Isoquercetin (NPC179950) | |
| NCT03314025 | Prophylactic Tamsulosin in Prevention of Post-operative Urinary Retention in Men After Transanal Endoscopic Microsurgery | Urinary retention | Tamsulosin Hydrochloride (NPC203424) | |
| NCT00026299 | Oxaliplatin With or Without Gefitinib in Treating Patients With Metastatic or Locally Recurrent Colorectal Cancer | colorectal carcinoma | Gefitinib (NPC56271) | |
| NCT01522950 | A Study Examining the Effects of Nebivolol Compared to Atenolol on Endothelial Function | hypertension | Atenolol (NPC307123) | |
| NCT03147092 | Matão Controlling Hypertension (MatCH Study): Rationale and Design | hypertension | Atenolol (NPC307123) | |
| NCT04514510 | Fixed Dose Flavonoid Isoquercetin on Thrombo-Inflammatory Biomarkers in Subjects With Stable Sickle Cell Disease | sickle cell anemia | Isoquercetin (NPC179950) | |
| NCT00616499 | Gefitinib in Treating Patients With Stage IIIA Non-Small Cell Lung Cancer | lung cancer | Gefitinib (NPC56271) | |
| NCT04622865 | Masitinib Combined With Isoquercetin and Best Supportive Care in Hospitalized Patients With Moderate and Severe COVID-19 | COVID-19 | Isoquercetin (NPC179950) | |
| NCT00080340 | Study of TLK286 (Telcyta) vs. Gefitinib in Locally Advanced or Metastatic Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT02467400 | Dose Response and Receptor Selectivity of Beta-blocker Effects on Bone Metabolism | osteoporosis | Atenolol (NPC307123) | |
| NCT01022463 | Effect of Ivabradine on Heart Rate & Effort Tolerance in Mitral Stenosis in Sinus Rhythm | mitral valve stenosis | Atenolol (NPC307123) | |
| NCT00252811 | A Study of ZD1839 Effects on Cell Proliferation in Breast Cancer | breast cancer | Gefitinib (NPC56271) | |
| NCT00193284 | Chemotherapy Plus Gefitinib Followed by Chemotherapy, Radiation Therapy, and Gefitinib For Head and Neck Cancer | head and neck malignant neoplasia | Gefitinib (NPC56271) | |
| NCT00824746 | Study of Gefitinib Retreatment in Non-Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00012337 | ZD 1839 in Treating Patients With Metastatic Kidney Cancer | kidney cancer | Gefitinib (NPC56271) | |
| NCT01314859 | Nifedipine Treatment in Preterm Labor | premature birth | Nifedipine (NPC190945) | |
| NCT01145612 | Atenolol Versus Losartan in the Prevention of Progressive Dilation of the Aorta in Marfan Syndrome | Marfan syndrome | Atenolol (NPC307123) | |
| NCT01167153 | Effectiveness of Valsartan/Amlodipine (EXforge®) and Nifedipine treAtment coMparison in Treating Chinese Hypertensive Patients | hypertension | Nifedipine (NPC190945) | |
| NCT01769066 | Clinical Value of Sequential Gefitinib With Pemetrexed/Platinum for Advanced NSCLC | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00687973 | Effect of the Fixed Dose Combination Amlodipine/Valsartan on Central Aortic Blood Pressure in Uncontrolled Essential Hypertension With Amlodipine 5 mg | hypertension | Atenolol (NPC307123) | |
| NCT00238628 | A Phase I/II Study of Bexarotene in Combination With ZD1839 (IRESSA®) in the Treatment of Non-Small Cell Lung Cancer | lung cancer | Gefitinib (NPC56271) | |
| NCT00025142 | Gefitinib and Combination Chemotherapy in Treating Patients With Advanced Solid Tumors or Colorectal Cancer | colorectal carcinoma | Gefitinib (NPC56271) | |
| NCT00076388 | Iressa Versus Docetaxel (Taxotere) | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00198159 | A Phase II Study of Iressa in Patients With Chemo Refractory Germ Cell Tumors Expressing EGFR | germ cell tumor | Gefitinib (NPC56271) | |
| NCT00237900 | Gefitinib in Combination With Chemoradiation in Resectable Gastric Cancer | gastric carcinoma | Gefitinib (NPC56271) | |
| NCT00250887 | Pre- and Postoperative Use of ZD1839 (Iressa) in Recurrent Glioblastoma, Including Translational Research | glioblastoma multiforme | Gefitinib (NPC56271) | |
| NCT02132533 | Nifedipine for Acute Tocolysis of Preterm Labor | premature birth | Nifedipine (NPC190945) | |
| NCT04474626 | Isoquercetin in Sickle Cell Anemia | sickle cell anemia | Isoquercetin (NPC179950) | |
| NCT02025218 | Iressa Re-challenge in Advanced NSCLC EGFR-mutated Patients | lung cancer | Gefitinib (NPC56271) | |
| NCT01348204 | Nasal Potential Studies Utilizing Cystic Fibrosis Transmembrane Regulator (CFTR) Modulators | cystic fibrosis | Quercetin (NPC20791) | |
| NCT00048087 | Iressa/Docetaxel in Non-Small-Cell Lung Cancer | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00223717 | Treatment of Supine Hypertension in Autonomic Failure | hypertension | Nifedipine (NPC190945) | |
| NCT00258297 | Gefitinib in Treating Patients With Stage I, Stage II, or Stage III Esophageal Cancer That Can Be Removed By Surgery | esophageal cancer | Gefitinib (NPC56271) | |
| NCT02337933 | Effect of Ursolic Acid Administration on Insulin Sensitivity and Metabolic Syndrome | metabolic syndrome | Ursolic Acid (NPC51700) | |
| NCT01871480 | CIK Cell Transfusion Plus Gefitinib As Second Or Third-Line Treatment for Advanced Adenocarcinoma Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT02417831 | Determine the Bioequivalence of Two Formulations of Tamsulosin HCl Capsules in Fasted Male. | benign prostatic hyperplasia | Tamsulosin Hydrochloride (NPC203424) | |
| NCT02326285 | GEfitinib Plus viNOrelbine in Advanced EGFR Mutated NSCLC. GENOA Trial | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT03787992 | Ningetinib (CT053PTSA) Plus Gefitinib in Stage IIIB or IV NSCLC Patients With EGFR Mutation and T790M Negative | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00372515 | High Dose Gefitinib for the Treatment of Carcinomatous Meningitis in Adult Patients With Non-Small Cell Lung Cancer and Known or Suspected EGFR Mutations | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00259064 | Iressa v BSC (Best Supportive Care) in First Line NSCLC | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT05456022 | Therapeutic Efficacy of Quercetin Versus Its Encapsulated Nanoparticle on Tongue Squamous Cell Carcinoma Cell Line | mouth neoplasm | Quercetin (NPC20791) | |
| NCT01637948 | Controlled Clinical Trial of Traditional Chinese Medicine Mouthrinse | gingivitis | Thymol (NPC259512) | |
| NCT00813722 | Effect of Active Telephone Calls in the Compliance of Hypertensive Patients With Treatment | hypertension | Atenolol (NPC307123) | |
| NCT02552888 | Nitrite, Isoquercetin and Endothelial Dysfunction (NICE) Trial | chronic kidney disease | Isoquercetin (NPC179950) | |
| NCT02299765 | AZD9291 Versus Gefitinib or Erlotinib in Patients With Locally Advanced or Metastatic Non-small Cell Lung Cancer | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00268255 | Gefitinib and Radiation Therapy in Treating Patients With Inoperable Stage I or Stage II Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00247481 | ZD1839 (Iressa™) In Combination With Docetaxel As First-Line Treatment In Patients With Metastatic Breast Cancer | breast cancer | Gefitinib (NPC56271) | |
| NCT00519870 | Losartan Therapy in Pulmonary Hypertension | pulmonary hypertension | Nifedipine (NPC190945) | |
| NCT02040064 | Tolerability and Efficacy of Tremelimumab in Combination With Gefitinib in NSCLC Patients | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT04733534 | An Open-Label Intervention Trial to Reduce Senescence and Improve Frailty in Adult Survivors of Childhood Cancer | childhood cancer | Quercetin (NPC20791) | |
| NCT04392622 | d-Limonene +Radiation +Platinum Based Chemo for Xerostomia Prevention in Locally Advanced Head and Neck Squamous Cell Carcinoma | xerostomia | Limonene (NPC76145) | |
| NCT00479089 | Iressa and Taxotere Study in Patients With Metastatic Urothelial Cancer | urinary bladder cancer | Gefitinib (NPC56271) | |
| NCT00000102 | Congenital Adrenal Hyperplasia: Calcium Channels as Therapeutic Targets | congenital adrenal hyperplasia | Nifedipine (NPC190945) | |
| NCT02563405 | The Effects of Doxazosin and Nifedipine on Blood Pressure Variability and Uric Acid in Plasma in Hypertensive Patients | hypertension | Nifedipine (NPC190945) | |
| NCT00059722 | This Study is to Compare the Efficacy of ZD6474 and ZD1839 in Subjects With NSCLC. | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00024154 | Trastuzumab and Gefitinib in Treating Patients With Metastatic Breast Cancer | male breast carcinoma | Gefitinib (NPC56271) | |
| NCT00077025 | Comparing Anastrozole-Placebo to the Combination Anastrozole-ZD1839 in Postmenopausal Patients With Estrogen Receptor and/or Progesterone Receptor Metastatic Breast Cancer | breast cancer | Gefitinib (NPC56271) | |
| NCT01774721 | ARCHER1050: A Study of Dacomitinib vs. Gefitinib in 1st-Line Treatment Of Advanced NSCLC. | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00493025 | Paclitaxel, Cisplatin, Gefitinib, and Radiation Therapy Followed by Surgery and Gefitinib in Treating Patients With Locally Advanced Cancer of the Esophagus or Gastroesophageal Junction That Can Be Removed By Surgery | esophageal cancer | Gefitinib (NPC56271) | |
| NCT01784549 | Customized Neoadjuvant Versus Standard Chemotherapy in NSCL Patients With Resectable Stage IIIA (N2)Disease | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00093652 | Oxaliplatin, Gefitinib, and Radiation Therapy in Treating Patients With Locally Advanced or Metastatic Esophageal Cancer | esophageal cancer | Gefitinib (NPC56271) | |
| NCT00246519 | Pharmacogenomic Evaluation of Antihypertensive Responses | hypertension | Atenolol (NPC307123) | |
| NCT00242801 | Iressa vs Best Supportive Care - 2nd/3rd Line Survival Study | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00635973 | Open-Label Extension of Other SZ1839 (Iressa) Trials | cancer | Gefitinib (NPC56271) | |
| NCT02951637 | Chemotherapy Plus Gefitinib for Advanced Lung Adenocarcinoma and Sensitive EGFR Mutations: a Randomized Controlled Trial | lung adenocarcinoma | Gefitinib (NPC56271) | |
| NCT01736488 | Clinical Study to Evaluate the Antihypertensive Efficacy and Changes of Neurohormonal Markers of Fimasartan and Atenolol With Exaggerated Blood Pressure Response During Exercise in Essential Hypertensive Patients | hypertension | Atenolol (NPC307123) | |
| NCT00171756 | Study to Compare the Effect of Valsartan vs Atenolol on Pro-thrombotic State in Hypertensive Patients. | hypertension | Atenolol (NPC307123) | |
| NCT01375101 | Therapeutic Effect of Quercetin and the Current Treatment of Erosive and Atrophic Oral Lichen Planus | oral lichen planus | Quercetin (NPC20791) | |
| NCT00024089 | Gefitinib in Treating Patients With Recurrent and/or Metastatic Head and Neck Cancer | head and neck malignant neoplasia | Gefitinib (NPC56271) | |
| NCT00004266 | Drugs for High Blood Pressure and High Cholesterol in American Indians With Type 2 Diabetes | diabetic nephropathy | Nifedipine (NPC190945) | |
| NCT02387086 | A Phase I Study of Safety and Pharmacokinetics of Volitinib in Combination With Gefitinib in EGFR(+) NSCLC | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT04358562 | Clinical Trial of YH25448(Lazertinib) as the First-line Treatment in Patients With EGFR Mutation Positive Locally Advanced or Metastatic NSCLC (LASER301) | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT04846621 | Comparative Study Between Nicorandil and Nifedipine for the Treatment of Preterm Labour | premature birth | Nifedipine (NPC190945) | |
| NCT05062486 | A Study to Evaluate the Safety and Efficacy of RQC for AMD | age-related macular degeneration | Quercetin (NPC20791) | |
| NCT03486496 | Gefitinib and Berberine in the First-line Treatment of Lung Adenocarcinoma With EGFR Mutation | lung adenocarcinoma | Gefitinib (NPC56271) | |
| NCT02417844 | Determine the Bioequivalence of Two Formulations of Tamsulosin HCl Capsules in Fed Male. | benign prostatic hyperplasia | Tamsulosin Hydrochloride (NPC203424) | |
| NCT00000513 | Trial of Antihypertensive Intervention Management | heart disease;hypertension | Atenolol (NPC307123) | |
| NCT01018511 | Study of Solifenacin Succinate and Tamsulosin Hydrochloride OCAS in Males With Lower Urinary Tract Symptoms | benign prostatic hyperplasia | Tamsulosin Hydrochloride (NPC203424) | |
| NCT04851821 | The Effectiveness of Phytotherapy in SARS-COV2(COVID-19) | COVID-19 | Quercetin (NPC20791) | |
| NCT02413515 | Blood Pressure Control of Nifedipine GITS 60mg Treatment in Chinese Hypertensive Subjects Uncontrolled on Starting-dose Mono-therapyo-therapy. | hypertension | Nifedipine (NPC190945) | |
| NCT00181688 | Iressa (ZD1839) Plus Anastrozole (Arimidex) in Patients With Ovarian Cancer | carcinoma;ovarian cancer;peritoneal neoplasm | Gefitinib (NPC56271) | |
| NCT01312337 | Iressa for EGFR Mutation Negative Non-small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT01227603 | Single Dose Bioequivalence Study Comparing Nifedipine/Candesartan FDC (Fixed Dose Combination) With Loose Combination of Nifedipine GITS (Gastro-intestinal Therapeutic System) Plus Candesartan and Single Components Under Fasting Conditions | hypertension | Nifedipine (NPC190945) | |
| NCT00319800 | Second-Line Irinotecan or Gefitinib in Docetaxel Pretreated NSCLC | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00091156 | Gefitinib After Chemotherapy in Treating Patients With Stage IIIB or Stage IV Non-Small Cell Lung Cancer | lung cancer | Gefitinib (NPC56271) | |
| NCT04390984 | Gefitinib With Anlotinib in Advanced Non-squamous NSCLC Patients With Uncleared Plasma ctDNA EGFRm After First-line Treatment With Gefitinib | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00268346 | Gefitinib in Treating Patients With Recurrent or Metastatic Esophageal or Gastroesophageal Junction Cancer | esophageal cancer | Gefitinib (NPC56271) | |
| NCT01438320 | Q-Trial in Patients With Hepatitis C | chronic hepatitis C virus infection | Quercetin (NPC20791) | |
| NCT00025350 | Gefitinib in Treating Patients With Recurrent Metastatic Colorectal Cancer | colorectal carcinoma | Gefitinib (NPC56271) | |
| NCT04298034 | Preeclampsia Postpartum Antihypertensive Treatment | preeclampsia | Nifedipine (NPC190945) | |
| NCT00014183 | ZD 1839 in Treating Patients With Stage IV or Recurrent Kidney Cancer | kidney cancer | Gefitinib (NPC56271) | |
| NCT02499822 | REducing Blood Pressure Variability in Essential Hypertension With RAmipril vErsus Nifedipine GITS Trial | hypertension | Nifedipine (NPC190945) | |
| NCT00599664 | Pharmacokinetic and Safety Study of OMS201 in Subjects Undergoing Ureteroscopic Treatment for Removal of Urinary Tract-Located Stones | nephrolithiasis | Nifedipine (NPC190945) | |
| NCT00086957 | Gefitinib, Trastuzumab, and Docetaxel in Treating Patients With Metastatic Breast Cancer | breast cancer | Gefitinib (NPC56271) | |
| NCT01202721 | Beta Blockers and Angiotensin Receptor Blockers in Bicuspid Aortic Valve Disease Aortopathy (BAV Study) | heart disease | Atenolol (NPC307123) | |
| NCT00298688 | A Study of IRESSA in Relapsed and Refractory Small Cell Lung Cancer | small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00319618 | Combination Weekly Taxotere™ With Iressa™ /Placebo in Metastatic Breast Cancer | breast cancer | Gefitinib (NPC56271) | |
| NCT00768560 | Nifedipine (Adalat CR, BAY A1040) High Dose PK/PD Study | hypertension | Nifedipine (NPC190945) | |
| NCT00030524 | ZD 1839 in Treating Patients With Locally Advanced or Metastatic Colorectal Cancer | colorectal carcinoma | Gefitinib (NPC56271) | |
| NCT01783834 | Gefitinib Versus Pemetrexed for Previously Treated NSCLC Patients | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT01294215 | High Dose BAYA1040_Nifedipine: a Long Term Combination Study | hypertension | Nifedipine (NPC190945) | |
| NCT02148380 | Combination of Chemotherapy and Gefitinib as First-line Treatment | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00620724 | Tocolytic Therapy in Conservative Management of Symptomatic Placenta Previa | placenta praevia | Nifedipine (NPC190945) | |
| NCT01777269 | Prospective Sexual Function Study for BPH Subjects | benign prostatic hyperplasia | Tamsulosin Hydrochloride (NPC203424) | |
| NCT00071994 | Gefitinib in Treating Patients With Advanced Unresectable Hepatocellular Carcinoma (Liver Cancer) | hepatocellular carcinoma | Gefitinib (NPC56271) | |
| NCT01021501 | The Pharmacokinetics of Nifedipine Controlled Release Tablets in Hypertensive Patients on Hemodialysis | hypertension | Nifedipine (NPC190945) | |
| NCT00467077 | Gefitinib and PEG-Interferon Alfa-2b in Treating Patients With Unresectable or Metastatic Kidney Cancer | kidney cancer | Gefitinib (NPC56271) | |
| NCT00672113 | Effects of Adalat LA and Coracten on Drug Levels, Blood Pressure, and Heart Rate in Fed Patients With Hypertension | hypertension | Nifedipine (NPC190945) | |
| NCT00029003 | Gefitinib in Treating Patients With Stage IIIB or Stage IV Non-Small Cell Lung Cancer | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT01355367 | High Dose BAYA1040 CR: a Long Term Extension Study | hypertension | Nifedipine (NPC190945) | |
| NCT02179671 | Immune-Modulated Study of Selected Small Molecules (Gefitinib, AZD9291, or Selumetinib + Docetaxel) or a 1st Immune-Mediated Therapy (IMT | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT03856697 | A Study to Evaluate Safety and Efficacy of HS-10296 as First-Line Treatment in Patients | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT04946383 | Safety and Effectivness of Quercetin & Dasatinib on Epigenetic Aging | aging | Quercetin (NPC20791) | |
| NCT03457337 | The Continuation of Gefitinib Treatment Beyond Progression in Non-small Cell Lung Cancer Patients With EGFR Mutation | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT01469000 | A Study of Pemetrexed and Gefitinib Versus Gefitinib in Non-Small Cell Lung Cancer (NSCLC) | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT02929693 | Clinical Study of YYJD Decoction Combined With Gefitinib in Advanced Pulmonary Adenocarcinoma | cancer | Gefitinib (NPC56271) | |
| NCT02882984 | Osimertinib Treatment on EGFR T790M Plasma Positive NSCLC Patients (APPLE) | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT02588261 | A Study of Ramucirumab (LY3009806) in Combination With Erlotinib in Previously Untreated Participants With EGFR Mutation-Positive Metastatic NSCLC (RELAY) | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00885599 | Efficacy of a Naturally-derived Mouthrinse for Gingival Inflammation | gingivitis | Thymol (NPC259512) | |
| NCT00265070 | Trial of Iressa in Prostate Cancer Patients | prostate cancer | Gefitinib (NPC56271) | |
| NCT00396656 | Safety and Efficacy of Valsartan vs Atenolol and Hydrochlorothiazide Combination on Blood Flow in Hypertensive Patients | hypertension | Atenolol (NPC307123) | |
| NCT00891579 | Study of Pemetrexed Versus Gefitinib in Patients With Locally Advanced or Metastatic Non Small Cell Lung Cancer Who Have Previously Received Platinum-Based Chemotherapy Without Epidermal Growth Factor Receptor (EGFR) Mutations | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00006048 | ZD 1839 Plus Combination Chemotherapy in Treating Patients With Non-Small Cell Lung Cancer | lung cancer | Gefitinib (NPC56271) | |
| NCT00787410 | An Open-label, Phase II Trial of ZD1839 (IRESSA) in Patients With Malignant Mesothelioma | mesothelioma | Gefitinib (NPC56271) | |
| NCT01079962 | An Active-controlled, Clinical Trial to Assess Central Hemodynamic Effects of Bisoprolol in Hypertensive Patients (Central Hemodynamic Assessment Measured in Patient With HypertensION [CHAMPION]) | hypertension | Atenolol (NPC307123) | |
| NCT01570296 | A Trial of Gefitinib in Combination With BKM120 in Patients With Advanced Non-Small Cell Lung Cancer, With Enrichment for Patients Whose Tumours Harbour Molecular Alterations of PI3K Pathway and Known to Overexpress EGFR | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00241475 | Gefitinib in the Treatment of the First Relapse of Prostate Cancer Beyond Prostatectomy or Radiotherapy | prostate cancer | Gefitinib (NPC56271) | |
| NCT02930954 | Stereotactic Body Radiation Therapy (SBRT) in Newly Diagnosed Advanced Staged Lung Adenocarcinoma (Sindas) | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT00023699 | Gefitinib in Treating Patients With Recurrent or Persistent Ovarian Epithelial Cancer or Primary Peritoneal Cancer | ovarian cancer;peritoneum cancer | Gefitinib (NPC56271) | |
| NCT01024413 | Phase III Trial to Evaluate the Elortinib vs Gefitinib in Advanced NSCLC With EGFR Exon 19 or 21 Mutations | neoplasm | Gefitinib (NPC56271) | |
| NCT01659580 | Phase III Trial of Dantonic® (T89) Capsule to Prevent and Treat Stable Angina | angina pectoris | Borneol (NPC283655) | |
| NCT01039948 | A Phase 1b/2 Study in Asian Subjects With Non-Small Cell Lung Cancer | carcinoma;lung cancer | Gefitinib (NPC56271) | |
| NCT00333294 | Phase II Iressa + Irradiation Followed by Chemo in NSCLC | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT01303783 | Study of Nifedipine GITS and Candesartan Combination Compared to Monotherapy in Patients With Essential Hypertension | hypertension | Nifedipine (NPC190945) | |
| NCT05276895 | Effect of Natural Senolytic Agents & NLRP3 Inhibitors on Osteoarthritis | osteoarthritis | Quercetin (NPC20791) | |
| NCT01040780 | Safety and Efficacy Study of Icotinb in Non-small Cell Lung Cancer (NSCLC) Patients | non-small cell lung carcinoma | Gefitinib (NPC56271) | |
| NCT01119534 | Comparative Efficacy of the Suppository Versus Guaiacol Suppository Versus Guaifenesin Syrup in Pediatric Patients With Cough Due the Infectious Origin | Cough | Camphor (NPC131981) | |
| NCT00200421 | A Pilot Study Comparing Nebivolol and Atenolol and Its Effects With Exercise in Patients With Mild to Moderate Hypertension | hypertension | Atenolol (NPC307123) |
❱❱❱ Associated Human Diseases and Detailed Association Evidence
How do we define the Plant-Targeted Human Disease Association?
Associated human diseases of an individual plant are summurized based on FOUR types of association evidence, these include:
❶ Association by Therapeutic Target: Bioactive protein targets of the plant were defined in "Molecular Targets" section, target-disease associations collected from TTD database were subsequently used to build the associations between the plant and its targeted human diseases.
❷ Association by Disease Gene Reversion: Plant and a specific disease will be associated when >= 1 plant target gene overlaped with disease's DEGs.
❸ Association by Clinical Trials of Plant: Plant and a specific disease will be associated when >= 1 clinical trial (the plant is the intervetion) can be matched in ClinicalTrials.gov database.
❹ Association by Clinical Trials of Plant Ingredients: Plant and a specific disease will be associated when >= 1 clinical trial (the plant ingredient is the intervetion) can be matched in ClinicalTrials.gov database.
Associated Disease of the Plant |
Association Type & Detailed Evidence |
|---|---|
Malignant haematopoietic neoplasmDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B33 |
KDM1A,TP53,SRC,AURKB,FLT3,CDK1,ERBB4,DPP4,ERBB2
|
Esophageal cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B70 |
TP53
NCT00493025,NCT00268346,NCT00290719,NCT00100945,NCT00258297,NCT00258323,NCT01291823,NCT00093652 |
Adenocarcinoma of stomachDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B72.0 |
NPSR1,NOX4,BLM,CDK1,DRD1,CYP19A1,ALOX15,MET,AURKB
|
Malignant neoplasms of corpus uteri, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C76.Z |
BLM,CA2,CDK1,CYP1A1,AURKB,XDH,ALOX15,PTK6,NPSR1
|
Rheumatoid arthritisDisease Category: 15.Diseases of the musculoskeletal system or connective tissueDisease ICD-11 Code: FA20 |
CA2,ABCG2,RELA,HMGCR,NFKB1,MAPK14,CCR2,ALOX5
|
PsoriasisDisease Category: 14.Diseases of the skinDisease ICD-11 Code: EA90 |
RORC,NFKB1,ALOX5,FLT3,RELA,IRAK4,DPP4,ERBB3
|
Cardiovascular diseaseDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA00-BE2Z |
SRC,KDR,MET,LCK,HMGCR,PTK6,RIPK2
NCT01313702 |
Brain cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A00 |
MET,ERBB2,EPHB4,EGFR,TP53,SRC,HIF1A,KDR
|
Pancreatic cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C10 |
ERBB2,TP53,MAPK1,IGF1R,CCR2,SRC,EGFR,ERBB3
|
West Nile virus infectionDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D46 |
NFKB1,LYN,RIPK2,CYP19A1,CCR2,MET,AXL,DPP4
|
Chikungunya virus diseaseDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D40 |
LMNA,ERBB4,HIF1A,RIPK2,CYP19A1,EGFR,XDH,AXL
|
Renal cell carcinoma, chromophobe typeDisease Category: X.Extension CodesDisease ICD-11 Code: XH6153 |
CA2,CYP1A1,CYP3A4,ADRB1,CYP1A2,CA12,MET,AURKB
|
Mature T-cell lymphomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A90 |
KDM1A,ERBB2,EGFR,MKNK2,ERBB4,IRAK4,HIF1A
|
Multiple myelomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A83 |
FLT3,HMGCR,AXL,LCK,KDR,IGF1R,SRC
|
Malignant neoplasms of oesophagusDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B70 |
NOX4,BLM,AURKB,ALOX15,CDK1,CHEK2,NPSR1
|
Invasive carcinoma of breastDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C61 |
CA12,BLM,BLK,CDK1,PTK6,AURKB,ERBB2
|
Carpal tunnel syndromeDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8C10.0 |
ADRA1B,NOX4,LYN,ADRB1,BLK,CYP1B1,DPP4
|
Gliomas of brainDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A00.0 |
NOX4,BLM,LYN,BLK,HCK,CCR2,CHEK2
|
Pulmonary hypertensionDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BB01 |
HIF1A,DRD1,ADRB1,ADRA1B
NCT00140959,NCT00813722,NCT00232882,NCT00768560,NCT00750113,NCT00158093,NCT00672113,NCT01736488,NCT00913965,NCT02563405,NCT01350609,NCT00246519,NCT01303783,NCT00396656,NCT00529750,NCT02641821,NCT00541684,NCT00171132,NCT01518855,NCT01522950,NCT00145210,NCT01287260,NCT00295542,NCT01251146,NCT02426177,NCT01227603,NCT02047019,NCT01294215,NCT04931108,NCT02940548,NCT01030016,NCT00519870,NCT00173667,NCT01830530,NCT00171756,NCT02441842,NCT00000513,NCT01079962,NCT00000514,NCT00223717,NCT01355367,NCT02413515,NCT01788358,NCT00338260,NCT02513927,NCT03147092,NCT00669279,NCT02499822,NCT01021501,NCT00200421,NCT00389168,NCT00772499,NCT01762436,NCT00687973,NCT00993109,NCT01167153,NCT01071122,NCT00661895,NCT00817037 |
Postoperative inflammationDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1A00-CA43 |
ALOX5,MAPK14,LCK,PTK6,RIPK2,SRC
|
MelanomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C30 |
MET,KDR,CCR2,MAPK1,ADRB1,EGFR
|
Alzheimer diseaseDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8A20 |
MET,FLT3,CACNA1C,MAPT,KDR,MAPK14
|
Liver cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C12 |
IGF1R,MKNK2,MET,KDR,CSNK2A1,EGFR
|
Chronic kidney diseaseDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GB61 |
XDH,IGF1R,CCR2
NCT02848131,NCT00817037,NCT02552888 |
Multiple sclerosisDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8A40 |
CCR2,RORC,RELA,HMGCR,SRC,NFKB1
|
Peritoneal cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C51 |
FLT3,TP53,KDR,SRC
NCT00317772,NCT00181688 |
Marfan syndrome or Marfan-related disordersDisease Category: 20.Developmental anomaliesDisease ICD-11 Code: LD28.0 |
NCT00723801,NCT00651235,NCT01715207,NCT01145612,NCT00429364,NCT00593710
|
AdenocarcinomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D40 |
NCT03486496,NCT02951637,NCT00020709,NCT00049543,NCT00040794,NCT00198380
|
Melanoma of skinDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C30 |
ADRA1B,NOX4,ABCG2,BLK,CCR2,IGF1R
|
Urothelial carcinoma of bladderDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C94.2 |
BLM,CA2,CYP3A4,CDK1,AURKB,CYP1A2
|
Type 2 diabetes mellitusDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A11 |
RELA,MAPK10,NFKB1,DPP4
NCT01839344 |
Stomach cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B72 |
MET,KDR,ERBB2,EGFR,TP53
|
Myelodysplastic syndromeDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A37 |
KDR,EGFR,KDM1A,AXL,TP53
|
Renal cell carcinomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C90 |
EGFR,KDR,MET
NCT00113529,NCT02446795 |
Chronic arterial occlusive diseaseDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BD4Z |
HMGCR,DPP4,TP53,CES1,MET
|
Myeloproliferative neoplasmDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A20 |
ERBB4,FLT3,LCK,KDM1A,SRC
|
Chronic painDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: MG30 |
FLT3,ERBB2,CCR2,EGFR,KDR
|
COVID-19Disease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D6Y |
NCT04536090,NCT04622865,NCT04851821,NCT04733651,NCT04853199
|
Malignant neoplasm metastasis in lymph nodes of head, face or neckDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D60.0 |
ADRA1B,NOX4,AURKB,NPSR1
NCT00206219 |
Superficial ovarian endometriosisDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GA10.B4 |
LMNA,CA4,CA2,SRC,DPP4
|
Lung cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25 |
RORC,EGFR,KDR,FLT3,CDK1,MET,KDM1A,EPHB4,ALOX5,ERBB3,ERBB4,AXL,ERBB2,IGF1R
NCT00098462,NCT03292133,NCT02321293,NCT00006048,NCT00955695,NCT00062270,NCT00452244,NCT00068653,NCT02025218,NCT00103051,NCT00091156,NCT00193336,NCT00455936,NCT00616499,NCT00238628,NCT00238251,NCT00357734,NCT01530334,NCT00188617,NCT00005806,NCT00310154,NCT01556191,NCT00062062,NCT01039948,NCT01024712,NCT00217698,NCT00006049,NCT00411047,NCT00144066,NCT00096486 |
Metastatic lymph node neoplasmDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D60 |
MET,ERBB3,KDR,EGFR
|
Fallopian tube cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C74 |
SRC,TP53
NCT00049556,NCT00317772 |
Acute diabete complicationDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A2Y |
AURKB,ADRB1,DPP4,KDR
|
Heart failureDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BD10-BD1Z |
ADRB1,XDH,CA2
NCT00589303 |
Squamous cell carcinomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B60-2D01 |
ERBB3
NCT00020709,NCT00126555,NCT01485809 |
Chronic obstructive pulmonary diseaseDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CA22 |
CCR2,MAPK14
NCT03989271,NCT01708278 |
GlaucomaDisease Category: 09.Diseases of the visual systemDisease ICD-11 Code: 9C61 |
CA4,ADRB1,MET,CA2
|
Mature B-cell leukaemiaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A82 |
EGFR,AXL,TP53,SRC
|
Pleomorphic sarcomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B54 |
KDR,SRC,MET
NCT00052754 |
Pain, unspecifiedDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: MG3Z |
ALOX5,ADRA1B,HMGCR,IGF1R
|
Atopic eczemaDisease Category: 14.Diseases of the skinDisease ICD-11 Code: EA80 |
CYP3A4,ALOX5,NFKB1,RELA
|
Ischemic heart diseaseDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA40-BA6Z |
MET
NCT00000513,NCT01202721,NCT00000514 |
Malignant neoplasms of bladder, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C94.Z |
NCT00479089,NCT00014144,NCT00352079,NCT00246974
|
Unspecified malignant neoplasms of unspecified sitesDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D4Z |
NCT00177242,NCT02929693,NCT04733534,NCT00635973
|
Other specified malignant neoplasms ofcolonDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B90.Y |
NPSR1,NOX4,CYP1A1,MET
|
Other specified malignant neoplasms of bronchus or lungDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25.Y |
CA12,XDH,CDK1,AURKB
|
Human immunodeficiency virus disease without mention of tuberculosis or malariaDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1C62 |
FLT3,HCK,MET,AXL
|
Solid tumour/cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A00-2F9Z |
EGFR,KDR,CCR2,CA4,ERBB3,ERBB2,TP53,CHEK2,CSNK2A1,MET,NFKB1,LCK,ERBB4,CYP3A4,LYN,AXL,POLB,CDK1,CA12,KDM1A,XDH,RELA,YES1,SRC,ALOX5,HIF1A,RORC,AURKB,MAPK1,MKNK2,FLT3,IGF1R,CA2,PIM1,MAPK10
|
Breast cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C60-2C6Y |
CA4,CDK1,CA12,ERBB3,EGFR,CA2,SRC,MET,CYP19A1,MKNK2,ERBB2,IGF1R,AXL,KDR
NCT00082667,NCT00357734,NCT00080743,NCT00319618,NCT00077025,NCT00066339,NCT00247481,NCT00255463,NCT00066378,NCT00252811,NCT00049062,NCT00637026,NCT00428896,NCT00086957,NCT00239343,NCT01732276,NCT00234403,NCT00057941,NCT00206492,NCT00206414 |
Mesothelioma of pleuraDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C26.0 |
KDR,LCK,ALOX15,CSNK2A2,CHEK2,CA7,CACNA1F,AURKB,HCK,XDH,CCR2,RECQL,DPP4,PIM1,LMNA,LYN,BLK,RIPK2,EGFR,CDK1,HMGCR,CA12,IRAK1,ALOX5,DRD1,TDP1,FLT3,PTK6,MAP4K4,SRC,MET,AXL
|
Adenocarcinoma of pancreasDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C10.0 |
CACNA1C,BLM,CA2,LCK,ALOX15,CHEK2,CA7,ABCG2,AURKB,HCK,XDH,POLB,CCR2,RECQL,DPP4,ERBB2,PIM1,LMNA,CES1,LYN,BLK,RIPK2,CYP1B1,ADRA1B,ALOX5,DRD1,HIF1A,PTK6,MET,AXL,NPSR1
|
Nasopharyngeal cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B6B |
EGFR,CDK1,ALOX5
|
Seborrhoeic dermatitisDisease Category: 14.Diseases of the skinDisease ICD-11 Code: EA81 |
CA2,CA12,CA4
|
Myocardial infarctionDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA41-BA43 |
HMGCR,MAPK14,DPP4
|
Choreiform disorderDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8A01 |
RELA,NFKB1,MAPT
|
Thyroid cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D10 |
KDR,TSHR,MET
|
Cervical cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C77 |
SRC,TP53
NCT00049556 |
Diabetes mellitus, type unspecifiedDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A14 |
NFKB1,DPP4,RELA
|
ParkinsonismDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8A00 |
DRD1,SRC,MAPT
|
Coronary atherosclerosisDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA80 |
ADRB1,MAPK14,HMGCR
|
Irritable bowel syndromeDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DD91 |
NFKB1,RELA,ABCG2
|
Bacterial infectionDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1A00-1C4Z |
CA2,CA12,CA4
|
Malignant mesenchymal neoplasmDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B5D-2B5Y |
EGFR,ERBB3,ERBB2
|
Essential hypertensionDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA00 |
CA2,ADRB1
NCT00185185 |
AsthmaDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CA23 |
CA4,ADRB1,ALOX5
|
Hepatitis virus infectionDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1E50-1E51 |
HMGCR,EGFR,ERBB2
|
Idiopathic interstitial pneumonitisDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CB03 |
FLT3,ALOX5,MAPK10
|
Malignant digestive organ neoplasmDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C11 |
SRC,KDR,ERBB2
|
Fungal infectionDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1F29-1F2F |
ALOX5,NFKB1,RELA
|
Angina pectorisDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA40 |
EGFR
NCT02029118,NCT01659580 |
Malignant neoplasms of kidney, except renal pelvisDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C90 |
NCT00012337,NCT00014183,NCT00467077
|
CholelithiasisDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DC11 |
NCT00713739,NCT00857090,NCT00599664
|
Anal fissure/fistulaDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DB50 |
NCT02527109,NCT02023047,NCT00972907
|
Malignant neoplasms of retroperitoneumDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C50 |
NCT00049556,NCT00189358,NCT00023699
|
Glioblastoma, primary, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH0MB1 |
NCT00238797,NCT01310855,NCT00250887
|
Central nervous system diseaseDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8A04-8D87 |
NCT00016991,NCT00025675,NCT00027625
|
Adenocarcinoma of prostateDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C82.0 |
NOX4,ALOX15
NCT01912820 |
Coeliac diseaseDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DA95 |
CDK1,CYP1A1,TSHR
|
Myelodysplastic syndromes, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A3Z |
PIM1,ERBB3,TSHR
|
Tetralogy of FallotDisease Category: 20.Developmental anomaliesDisease ICD-11 Code: LA88.2 |
ABCG2,CA4,RORC
|
Ebola virus diseaseDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D60.01 |
MAPK10,MET,AXL
|
Carcinosarcoma of uterusDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C76.43 |
ERBB4,CA2,HSD17B10,CYP19A1,LCK,KDM1A,ALOX15,CHEK2,CA7,HCK,XDH,POLB,ERBB3,DPP4,ERBB2,LYN,ADRB1,RIPK2,RORC,YES1,IRAK1,TSHR,TP53,TDP1,MAP4K4,MET,AURKB,NPSR1
|
Serous cystadenoma,borderline malignancy of ovaryDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C73.4 |
ERBB4,CA2,LCK,ALOX15,CACNA1F,HCK,XDH,CCR2,ERBB2,NOX4,LYN,ADRB1,ALOX12,RIPK2,BLK,RORC,YES1,CYP1A2,CA12,IRAK1,TSHR,ALOX5,CSNK2A3,TP53,FRK,TDP1,PTK6,MET
|
Diffuse large B-cell lymphomasDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A81 |
KDR,CACNA1C,BLM,HSD17B10,KDM1A,CSNK2A2,MAPK10,CHEK2,AURKB,RECQL,NOX4,LMNA,BLK,EGFR,CDK1,CA12,ADRA1B,TSHR,TP53,FRK,TDP1,SRC,MET,AXL
|
Malignant neoplasms of biliary tract, distal bile ductDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C15 |
MAPT,CACNA1C,BLM,ERBB4,MAPK10,CA4,CACNA1F,ERBB3,NOX4,LMNA,ALOX12,RIPK2,CDK1,IRAK1,ALOX5,HIF1A,CSNK2A3,TP53,PTK6,SRC,IGF1R,AURKB
|
Germ cell tumour of testisDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C80.2 |
KDR,BLM,MAPK1,CA4,ABCG2,HCK,XDH,CCR2,ERBB2,DPP4,NOX4,ALOX12,BLK,CDK1,CA12,IRAK1,ALOX5,DRD1,HIF1A,MAP4K4,MET,NPSR1
|
Diabetic foot ulcerDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BD54 |
ADRB1,EGFR
|
Metastatic malignant neoplasmDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D50-2E09 |
AXL,ERBB2
|
Motor neuron diseaseDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8B60 |
SRC,IGF1R
|
Metastatic biliary tract neoplasmsDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C14-2C17 |
EGFR,ERBB2
|
BCR-ABL1-negative chronic myeloid leukaemiaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A41 |
SRC,FLT3
|
InfluenzaDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1E30-1E32 |
ALOX5,XDH
|
Allergic/hypersensitivity disorderDisease Category: 04.Diseases of the immune systemDisease ICD-11 Code: 4A80-4A8Z |
DRD1,ADRB1
|
Mature B-cell lymphomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A85 |
CDK1,FLT3
|
Metastatic tumourDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D50-2E2Z |
CCR2,EPHB4
|
Lupus erythematosusDisease Category: 04.Diseases of the immune systemDisease ICD-11 Code: 4A40 |
RELA,NFKB1
|
Acute/subacute hepatic failureDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DB91 |
NFKB1,RELA
|
NADisease Category: NADisease ICD-11 Code: NA |
MET,NOX4
|
RetinopathyDisease Category: 09.Diseases of the visual systemDisease ICD-11 Code: 9B71 |
IGF1R,CCR2
|
Endometrial cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C76 |
TP53,SRC
|
IschemiaDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8B10-8B11 |
SRC,EGFR
|
Unspecific body region injuryDisease Category: 22.Injury, poisoning or certain other consequences of external causesDisease ICD-11 Code: ND56 |
EGFR,MET
|
Spinal cord/cranial nerve neoplasmDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A02 |
EGFR,ERBB2
|
Skin and skin-structure infectionDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1F28-1G0Z |
RELA,NFKB1
|
ObesityDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5B80-5B81 |
ADRB1,ADRA1B
|
Testicular cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C80 |
ERBB2,EGFR
|
ThrombocytopeniaDisease Category: 03.Diseases of the blood or blood-forming organsDisease ICD-11 Code: 3B64 |
KDR,ALOX5
|
Human immunodeficiency virus diseaseDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1C60-1C62 |
FLT3,CCR2
|
Bladder cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C94 |
ERBB2,TP53
|
Metabolic disorderDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5C50-5D2Z |
DPP4
NCT02337933 |
Gastrointestinal stromal tumourDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B5B |
KDR,SRC
|
Indeterminate colitisDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DD72 |
RELA,NFKB1
|
Hyper-lipoproteinaemiaDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5C80 |
HMGCR,CES1
|
Pituitary gland disorderDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A60-5A61 |
IGF1R,ALOX5
|
Urethral cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C93 |
EGFR
NCT00014144 |
Muscular dystrophyDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8C70 |
RELA,NFKB1
|
Ageing associated decline in intrinsic capacityDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: MG2A |
NCT04946383,NCT05422885
|
coronary artery diseaseDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA8Y-BA8Z |
NCT03943459,NCT04907253
|
Atrial fibrillationDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BC81.3 |
CA7
NCT00589303 |
Neuromuscular dysfunction of bladder, not elsewhere classifiedDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GC01.4 |
NCT00796614,NCT00340704
|
CardiomegalyDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BC45 |
NCT00338260,NCT00108082
|
Retention of urineDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: MF50.3 |
NCT03314025,NCT01568918
|
Sickle-cell disorderDisease Category: 03.Diseases of the blood or blood-forming organsDisease ICD-11 Code: 3A51 |
NCT04514510,NCT04474626
|
HemangiomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2E81-2F2Y |
NCT02342275,NCT03237637
|
Neuroendocrine carcinomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C34 |
NCT01039948,NCT00181688
|
Gingival diseasesDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DA0B |
NCT00885599,NCT01637948
|
GliosarcomaDisease Category: X.Extension CodesDisease ICD-11 Code: XH9RC8 |
NCT00052208,NCT00014170
|
Metastatic lung neoplasmDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D70 |
NCT02518802,NCT01466660
|
Pleural mesotheliomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C26 |
NCT00025207,NCT00787410
|
Pre-eclampsiaDisease Category: 18.Pregnancy, childbirth or the puerperiumDisease ICD-11 Code: JA24 |
CA4
NCT04298034 |
Placenta praevia specified as without haemorrhageDisease Category: 18.Pregnancy, childbirth or the puerperiumDisease ICD-11 Code: JA8B.0 |
NCT03542552,NCT00620724
|
Skin cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C30-2C37 |
NCT00126555,NCT00054691
|
Osteoarthritis, unspecifiedDisease Category: 15.Diseases of the musculoskeletal system or connective tissueDisease ICD-11 Code: FA0Z |
CA12,CDK1
|
Dengue fever, unspecifiedDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D2Z |
AURKB,ABCG2
|
Myotonic dystrophyDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8C71.0 |
ERBB3,CYP1A1
|
Pneumonia due to PseudomonasaeruginosaDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CA40.05 |
ADRA1B,DPP4
|
Systemic lupus erythematosusDisease Category: 04.Diseases of the immune systemDisease ICD-11 Code: 4A40.0 |
CYP1A1,YES1
|
Heart failure, unspecifiedDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BD1Z |
ABCG2,CYP1B1
|
Head and neck cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D42 |
KDR,ERBB2,EGFR,ERBB3,MET,TP53
NCT00681967,NCT00088907,NCT00195078,NCT00193284,NCT00352105,NCT00083057,NCT00242749,NCT00015964,NCT00024089,NCT00228488,NCT00169221,NCT01185158,NCT00095836 |
Glioblastoma of brainDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A00.00 |
KDR,BLM,CA2,CYP19A1,LCK,IRAK4,HCK,CCR2,DPP4,ERBB2,LMNA,LYN,RIPK2,CYP1B1,CA12,IRAK1,TSHR,CSNK2A3,MET
|
Colorectal cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B91 |
TP53,ERBB2,ERBB3,FLT3,KDR,MET,IGF1R,EGFR,MKNK2
NCT00026364,NCT00242788,NCT00026299,NCT00030524,NCT00087334,NCT00234429,NCT00025350,NCT00025142 |
Hepatocellular carcinoma of liverDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C12.02 |
MAPT,CA12,IRAK1,NOX4,CA4,BLM,CYP19A1,CDK1,AURKB,MAPT,CA12,IRAK1,CYP19A1,CDK1,MAP4K4,SRC,AURKB
|
Prostate hyperplasiaDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GA90 |
NCT01018511,NCT01294592,NCT03887871,NCT01254071,NCT01589263,NCT03887858,NCT02509104,NCT04987892,NCT02417831,NCT01604811,NCT02417844,NCT01829893,NCT02244242,NCT02303769,NCT01777269
|
PheochromocytomaDisease Category: X.Extension CodesDisease ICD-11 Code: XH3854 |
MAPT,CACNA1C,BLM,ERBB4,CA2,CA4,CACNA1F,XDH,ERBB3,NOX4,LYN,ADRB1,RORC,PTK6,MET
|
NeoplasmsDisease Category: 02.NeoplasmsDisease ICD-11 Code: 02 |
NCT00239304,NCT00084708,NCT00519077,NCT00039390,NCT00040781,NCT03040973,NCT00127829,NCT00084786,NCT00229723,NCT00186979,NCT02447419,NCT01024413,NCT00502060,NCT03239015
|
Malignant neoplasms of thymusDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C27 |
MAPT,NFKB1,HSD17B10,ALOX15,CHEK2,HCK,CYP3A4,DPP4,CYP1B1,IRAK1,DRD1,FRK,FLT3,PTK6
|
Non-small cell lung cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25 |
EGFR,KDR,MET
NCT01017874,NCT03599518,NCT00255489,NCT00256711,NCT02088112,NCT03457337,NCT02040064,NCT03529084,NCT00770588,NCT00891579,NCT00409006,NCT03656393,NCT00986284,NCT02151721,NCT00608868,NCT01391260,NCT00076388,NCT01933347,NCT01196234,NCT02588261,NCT00497250,NCT03866499,NCT00536107,NCT02148380,NCT00683306,NCT01404260,NCT01784549,NCT01579630,NCT01871480,NCT01192243,NCT02804776,NCT00162318,NCT00080340,NCT01833572,NCT01405079,NCT00478049,NCT03787992,NCT00252798,NCT01040780,NCT01203917,NCT00034879,NCT02824458,NCT01037998,NCT01312337,NCT01066195,NCT02387086,NCT01017679,NCT03050411,NCT00807066,NCT00048087,NCT04239833,NCT01783834,NCT00322452,NCT03381066,NCT03758287,NCT01158170,NCT01513174,NCT01774721,NCT02031601,NCT01498562,NCT03856697,NCT02882984,NCT04358562,NCT04028778,NCT00173875,NCT04390984,NCT02856893,NCT03123484,NCT03374280,NCT03944772,NCT01749072,NCT00029003,NCT00059722,NCT02893332,NCT00231465,NCT00252707,NCT01769066,NCT03267654,NCT02976116,NCT02039674,NCT00090675,NCT02411448,NCT02930954,NCT01955421,NCT04143607,NCT01469000,NCT01755923,NCT01982955,NCT00198393,NCT01510990,NCT00242801,NCT01864681,NCT03399669,NCT01570296,NCT00319800,NCT00264498,NCT00333294,NCT00809237,NCT02296125,NCT02299765,NCT02326285,NCT02374645,NCT02319577,NCT00234468,NCT03849768,NCT04248829,NCT00268255,NCT00328562,NCT00588445,NCT00824746,NCT02179671,NCT03333343,NCT03122717,NCT00372515,NCT01544179,NCT01610336,NCT00259064 |
Acute myeloid leukaemiaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A60 |
MAPT,AXL,ERBB4,KDM1A,RELA,PIM1,SRC,TP53,AURKB,FLT3,NFKB1
NCT00130702 |
Malignant neoplasms of adrenal glandDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D11 |
CA12,HMGCR,DPP4,CA4,BLM,NOX4,CYP19A1,CYP3A4,CDK1,MET,AURKB,CA7
|
Ovarian cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C73 |
SRC,KDR,MET,TP53,ERBB2,FLT3
NCT00049556,NCT00023699,NCT00189358,NCT00317772,NCT00181688 |
Adenocarcinoma of bronchus or lungDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25.0 |
CA12,NPSR1,BLM,TSHR,AURKB,XDH,PTK6,CDK1,CHEK2,CA7
NCT00344773 |
HepatoblastomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C12.01 |
CA12,CACNA1C,CA4,BLM,NOX4,CYP19A1,CDK1,MAP4K4,SRC,IGF1R,AURKB
|
Other specified malignant neoplasms of kidney, except renal pelvisDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C90.Y |
ALOX5,CYP19A1,AURKB,XDH,MET,CHEK2,TSHR,ALOX5,CACNA1F,HCK,AURKB
|
Squamous cell carcinoma of bronchus or lungDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25.2 |
BLM,CHEK2,CA7,XDH,ALOX12,CDK1,CA12,TSHR,PTK6,AURKB,NPSR1
|
Prostate cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C82 |
AURKB,ERBB2
NCT00241475,NCT04403568,NCT00483561,NCT00242918,NCT00025116,NCT00265070,NCT00319787,NCT00635856 |
Preterm newborn, unspecifiedDisease Category: 19.Certain conditions originating in the perinatal periodDisease ICD-11 Code: KA21.4Z |
NCT01796522,NCT01429545,NCT01360034,NCT01314859,NCT04846621,NCT04404686,NCT00137501,NCT00486824,NCT02132533,NCT01985594
|
LeishmaniasisDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1F54 |
BLM,LCK,HCK,CCR2,DPP4,NOX4,LYN,BLK,TSHR,FLT3
|
Cytomegaloviral diseaseDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D82 |
ADRA1B,CA2,LYN,CYP1A1,ADRB1,BLK,HCK,LCK,ALOX15,ERBB3
|
Malignant neoplasms of thyroid gland, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D10.Z |
CA12,ADRA1B,CA7,NOX4,ALOX5,CYP1B1,XDH,ERBB3,MET,DPP4
|
InsomniaDisease Category: 07.Sleep-wake disordersDisease ICD-11 Code: 7A00-7A0Z |
CA2
|
Synthesis disorderDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5C52-5C59 |
CES1
|
NeuropathyDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8C0Z |
CYP19A1
|
Hypopharyngeal cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B6D |
EGFR
|
Crohn diseaseDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DD70 |
RORC
|
Insulin-resistance syndromeDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A44 |
CCR2
|
EndometriosisDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GA10 |
CYP19A1
|
Central and peripheral nervous diseaseDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8A04-8E7Z |
ADRB1
|
AbortionDisease Category: 18.Pregnancy, childbirth or the puerperiumDisease ICD-11 Code: JA00 |
DRD1
|
Bone/articular cartilage neoplasmDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2F7B |
FLT3
|
DementiaDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6D80-6D8Z |
DRD1
|
Mineral deficiencyDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5B5K |
XDH
|
Urogenital cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C95-2C9Z |
ERBB2
|
Carcinoid syndromeDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5B10 |
ALOX5
|
Diffuse large B-cell lymphomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A81 |
FLT3
|
Autism spectrum disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6A02 |
DPP4
|
Kidney failureDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GB60-GB6Z |
TP53
|
Intrathoracic organs injuryDisease Category: 22.Injury, poisoning or certain other consequences of external causesDisease ICD-11 Code: NB32 |
MAPK14
|
Lip/oral cavity/pharynx neoplasmDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B6E |
TP53
|
Psychotic disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6A20-6A25 |
DRD1
|
DyslipidemiaDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5C80-5C81 |
HMGCR
|
Unspecified viral infection of unspecified siteDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D9Z |
FLT3
|
Cutaneous lupus erythematosusDisease Category: 14.Diseases of the skinDisease ICD-11 Code: EB50-EB5Z |
MAPK10
|
Inborn purine/pyrimidine/nucleotide metabolism errorDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5C55 |
XDH
|
Mild neurocognitive disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6D71 |
MAPT
|
Duodenal ulcerDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DA63 |
CA2
|
Extraocular muscle disorderDisease Category: 09.Diseases of the visual systemDisease ICD-11 Code: 9C82 |
IGF1R
|
Colon cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B90 |
EGFR
|
Inflammatory arthropathyDisease Category: 15.Diseases of the musculoskeletal system or connective tissueDisease ICD-11 Code: FA2Z |
FLT3
|
Musculoskeletal system/connective tissue diseaseDisease Category: 15.Diseases of the musculoskeletal system or connective tissueDisease ICD-11 Code: FC0Z |
LYN
|
Ventricular tachyarrhythmiaDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BC71 |
ADRB1
|
Mental/behavioural/neurodevelopmental disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6E20-6E8Z |
ADRA1B
|
Metastatic digestive system neoplasmDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D8Y |
SRC
|
Liver diseaseDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DB90-DB9Z |
MET
|
OsteosarcomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B51 |
SRC
|
Arteries/arterioles disorderDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BD52 |
MAPK1
|
Transplant rejectionDisease Category: 22.Injury, poisoning or certain other consequences of external causesDisease ICD-11 Code: NE84 |
TP53
|
Ureteral cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C92 |
ERBB2
|
OesophagitisDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DA24 |
CYP19A1
|
Attention deficit hyperactivity disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6A05 |
ADRA1B
|
Epidermal dysplasiasDisease Category: 14.Diseases of the skinDisease ICD-11 Code: EK90 |
SRC
|
Phakomatoses/hamartoneoplastic syndromeDisease Category: 20.Developmental anomaliesDisease ICD-11 Code: LD2D |
FLT3
|
Peroxisomal diseaseDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5C57 |
CES1
|
AnxietyDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: MB24.3 |
CYP3A4
|
Cushing syndromeDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A70 |
CYP19A1
|
Viral encephalitisDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1C80 |
CCR2
|
Biliary tract cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C17 |
MET
|
Coronavirus infectionDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D92 |
CSNK2A1
|
FilariasisDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1F66 |
ALOX5
|
Metastatic meninges neoplasmDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D51 |
EPHB4
|
MigraineDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8A80 |
ADRB1
|
Vitamin deficiencyDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5B55-5B5F |
MAPK14
|
Digestive organ benign neoplasmDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2E92 |
KDR
|
ThyrotoxicosisDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A02 |
IGF1R
|
Hypo-thyroidismDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A00 |
TSHR
|
Cardiac arrhythmiaDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BC9Z |
ADRB1
|
Dissociative neurological symptom disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6B60 |
MAPK10
|
Low bone mass disorderDisease Category: 15.Diseases of the musculoskeletal system or connective tissueDisease ICD-11 Code: FB83 |
CA2
|
Autoimmune diseaseDisease Category: 04.Diseases of the immune systemDisease ICD-11 Code: 4A40-4A45 |
RORC
|
SchizophreniaDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6A20 |
DRD1
|
Chronic myelomonocytic leukaemiaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A40 |
FLT3
|
Synovial sarcomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B5A |
SRC
|
GoutDisease Category: 15.Diseases of the musculoskeletal system or connective tissueDisease ICD-11 Code: FA25 |
XDH
|
Circulatory system diseaseDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BE2Z |
ADRB1
|
Intracerebral haemorrhageDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8B00 |
EGFR
|
Ewing sarcomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B52 |
KDM1A
|
LeiomyosarcomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B58 |
SRC
|
Cocaine use disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6C45 |
DRD1
|
SepsisDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1G40-1G41 |
ADRB1
|
Retinal vascular occlusionDisease Category: 09.Diseases of the visual systemDisease ICD-11 Code: 9B74 |
ERBB2
|
MastocytosisDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A21 |
FLT3
|
Conduction disorderDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BC63 |
ADRB1
|
Multiple structural anomalies syndromeDisease Category: 20.Developmental anomaliesDisease ICD-11 Code: LD2F |
IGF1R
|
LiposarcomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B59 |
TP53
|
Blood/blood-forming organ symptonDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: MA14 |
HMGCR
|
Tic disorderDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8A05 |
DRD1
|
EncephalopathyDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8E47 |
XDH
|
Transplanted organ/tissueDisease Category: 24.Factors influencing health status or contact with health servicesDisease ICD-11 Code: QB63 |
FLT3
|
Cystic/dysplastic kidney diseaseDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GB8Y |
EPHB4
|
Metastatic liver/bile duct neoplasmDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D80 |
TP53
|
Skin postprocedural disorderDisease Category: 14.Diseases of the skinDisease ICD-11 Code: EL8Y |
EGFR
|
Supraventricular tachyarrhythmiaDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BC81 |
ADRB1
|
Adrenomedullary hyperfunctionDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A75 |
HIF1A
|
Ovarian dysfunctionDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A80 |
TP53
|
Carcinoma of male breastDisease Category: X.Extension CodesDisease ICD-11 Code: XH5KW8 |
NCT00024154
|
OsteoporosisDisease Category: 15.Diseases of the musculoskeletal system or connective tissueDisease ICD-11 Code: FB83.1 |
NCT02467400
|
Post-traumatic stress disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6B40 |
NCT02560805
|
Congenital adrenal hyperplasiaDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A71.01 |
NCT00000102
|
Cannabis use disorderDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6C41 |
NCT05432284
|
Unknown or unspecified psychoactive substance dependence, substance and state of remission unspecifiedDisease Category: 06.Mental, behavioural or neurodevelopmental disordersDisease ICD-11 Code: 6C4G.2Z |
NCT00593463
|
CoughDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: MD12 |
NCT01119534
|
Macular degenerationDisease Category: 09.Diseases of the visual systemDisease ICD-11 Code: 9B75 |
NCT05062486
|
OsteoarthritisDisease Category: 15.Diseases of the musculoskeletal system or connective tissueDisease ICD-11 Code: FA00-FA05 |
NCT05276895
|
Crohn disease of other specified siteDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DD70.Y |
NCT04887818
|
Small cell carcinoma of bronchus or lungDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25.1 |
NCT00298688
|
Hepatocellular carcinoma, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH4W48 |
NCT00071994
|
Ischaemic heart diseases, unspecifiedDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BA6Z |
NCT00000478
|
Chronic hepatitis CDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1E51.1 |
NCT01438320
|
Gastro-oesophageal reflux diseaseDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DA22 |
NCT02226484
|
Cystic fibrosisDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CA25 |
NCT01348204
|
Primary sclerosing cholangitisDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DB96.2 |
NCT03216876
|
Malignant neoplasms of stomach, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B72.Z |
NCT00237900
|
Neuroblastoma, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH85Z0 |
NCT00135135
|
Congenital aplastic anaemiaDisease Category: 03.Diseases of the blood or blood-forming organsDisease ICD-11 Code: 3A70.0 |
NCT01720147
|
Other specified non-alcoholic fatty liver diseaseDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DB92.Y |
NCT04853082
|
ProteinuriaDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: MF96 |
NCT00817037
|
Chronic prostatitisDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GA91.0 |
NCT04552431
|
Neoplasms of unknown behaviour of breastDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2F95 |
NCT00229697
|
Malignant neoplasms of other or unspecified parts of mouthDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B66 |
NCT05456022
|
Malignant neoplasm of pancreas, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C10.Z |
NCT00234416
|
Malignant neoplasms of corpus uteriDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C76 |
NCT00027690
|
Lichen planusDisease Category: 14.Diseases of the skinDisease ICD-11 Code: EA91 |
NCT01375101
|
Chronic kidney disease, stage unspecifiedDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GB61.Z |
NCT00004266
|
Follicular lymphomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A80 |
NCT00455416
|
Mixed tumour, malignant, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH0V86 |
NCT00509002
|
GerminomaDisease Category: X.Extension CodesDisease ICD-11 Code: XH1E13 |
NCT00198159
|
Malignant neoplasms of nasopharynxDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B6B |
NCT00212108
|
Epilepsy/seizureDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8A61-8A6Z |
NCT03949478
|
Primary Raynaud diseaseDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BD42.0 |
NCT00000530
|
XerostomiaDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DA02.1 |
NCT04392622
|
Malignant neoplasms of stomachDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B72 |
NCT03170180
|
Malignant neoplasm metastasis in male genital organsDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2E06 |
NCT00239291
|
Mitral valve stenosisDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BB60 |
NCT01022463
|
Malignant neoplasms of fallopian tube, unspecifiedDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C74.Z |
NCT00189358
|
Cerebrovascular diseaseDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8B22-8B2Z |
NCT00000514
|
Inflammatory bowel diseases, unspecifiedDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DD7Z |
NOX4
|
Zika virus diseaseDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D48 |
AXL
|
KeratoconusDisease Category: 09.Diseases of the visual systemDisease ICD-11 Code: 9A78.50 |
CA4
|
Systemic sclerosisDisease Category: 04.Diseases of the immune systemDisease ICD-11 Code: 4A42 |
NOX4
|
Chronic rhinosinusitisDisease Category: 12.Diseases of the respiratory systemDisease ICD-11 Code: CA0A |
CYP1A1
|
Dilated cardiomyopathyDisease Category: 11.Diseases of the circulatory systemDisease ICD-11 Code: BC43.0 |
ABCG2
|
Lyme borreliosisDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1C1G |
MET
|

