Collective Molecular Activities of the Plant: Castanea Mollissima

Plant ID: NPO1191
Plant Latin Name: Castanea Mollissima
Taxonomy Genus: Castanea
Taxonomy Family: Fagaceae
Plant External Links:
NCBI TaxonomyDB:
60419
Plant-of-the-World-Online:
n.a.
Country/Region:
ChinaTraditional Medicine System:
TCMMedicinal Functions:
Antidote; Astringent; Stomachic
China
Overview of Ingredients
45 All known Ingredients in Total
Unique ingredients have been isolated from this plant.Plant-Ingredients Associations were manually curated from publications or collected from other databases.
27 Ingredients with Acceptable Bioavailablity
Unique ingredients exhibit acceptable human oral bioavailablity, according to the criteria of SwissADME [PMID: 28256516] and HobPre [PMID: 34991690]. The criteria details:SwissADME: six descriptors are used by SwissADME to evaluate the oral bioavailability of a natural product:
☑ LIPO(Lipophility): -0.7 < XLOGP3 < +5.0
☑ SIZE: 150g/mol < MW < 500g/mol
☑ POLAR(Polarity): 20Ų < TPSA < 130Ų
☑ INSOLU(Insolubility): -6 < Log S (ESOL) < 0
☑ INSATU(Insaturation): 0.25 < Fraction Csp3 < 1
☑ FLEX(Flexibility): 0 < Num. rotatable bonds < 9
If 6 descriptors of a natural plant satisfy the above rules, it will be labeled high HOB.
HobPre: A natural plant ingredient with HobPre score >0.5 is labeled high human oral availability (HOB)
21 Ingredients with experimental-derived Activity
Unique ingredients have activity data available.Ingredient Structrual Cards
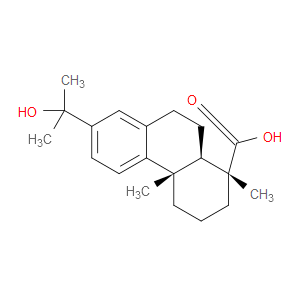
Ingredient ID: NPC95126
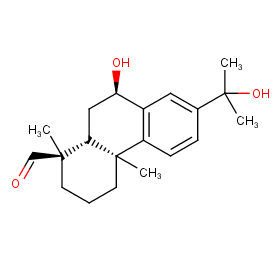
Ingredient ID: NPC94939
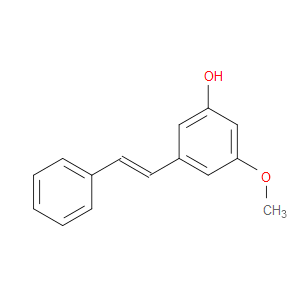
Ingredient ID: NPC94045
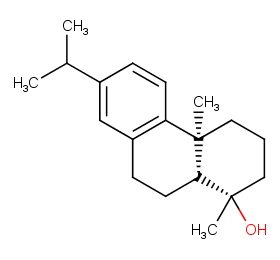
Ingredient ID: NPC93829
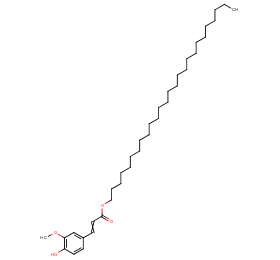
Ingredient ID: NPC9129
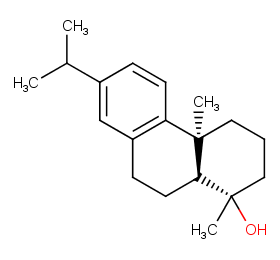
Ingredient ID: NPC9007
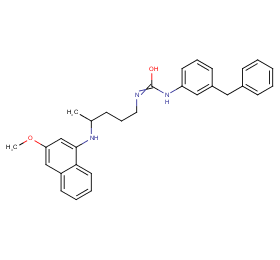
Ingredient ID: NPC84279
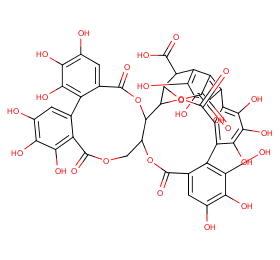
Ingredient ID: NPC64478
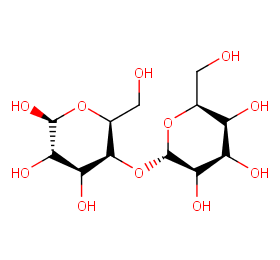
Ingredient ID: NPC50855
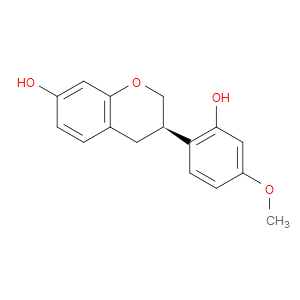
Ingredient ID: NPC50315
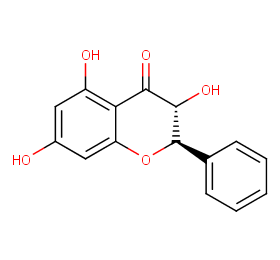
Ingredient ID: NPC4152
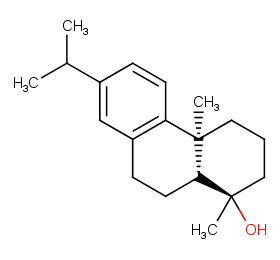
Ingredient ID: NPC38807
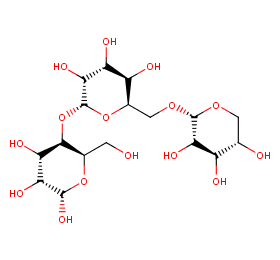
Ingredient ID: NPC37592
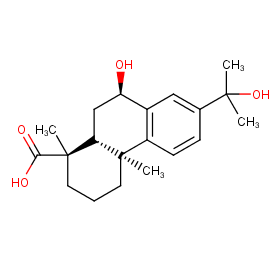
Ingredient ID: NPC369
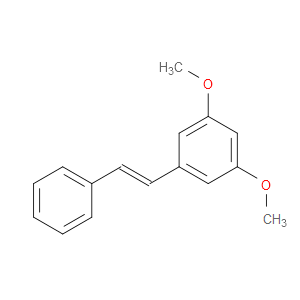
Ingredient ID: NPC35543
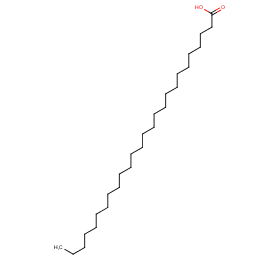
Ingredient ID: NPC32021

Ingredient ID: NPC317671
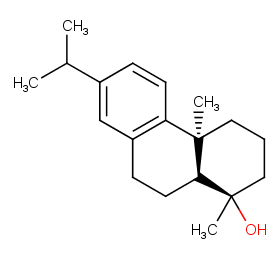
Ingredient ID: NPC303032
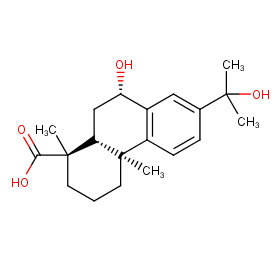
Ingredient ID: NPC293831
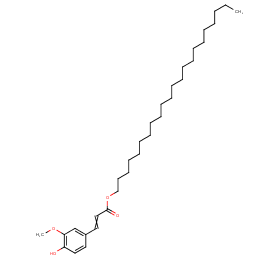
Ingredient ID: NPC283700
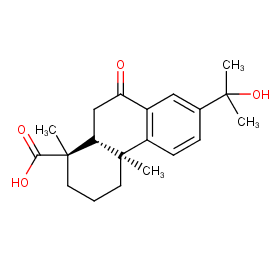
Ingredient ID: NPC269923
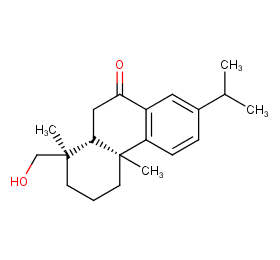
Ingredient ID: NPC265513
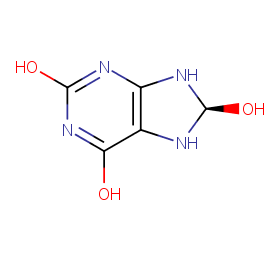
Ingredient ID: NPC263451
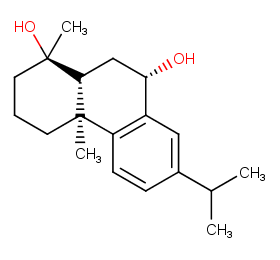
Ingredient ID: NPC24446
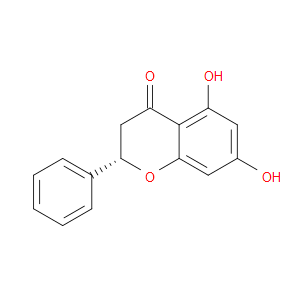
Ingredient ID: NPC243083
Ingredient ID: NPC237224
Ingredient ID: NPC236777
Ingredient ID: NPC234337
Ingredient ID: NPC2290
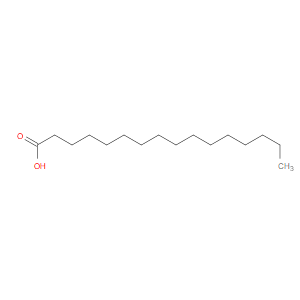
Ingredient ID: NPC216630
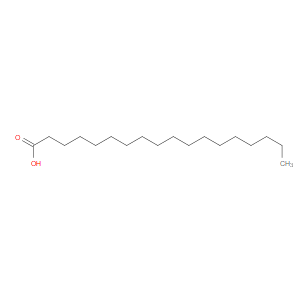
Ingredient ID: NPC209970
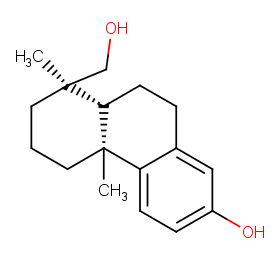
Ingredient ID: NPC189468
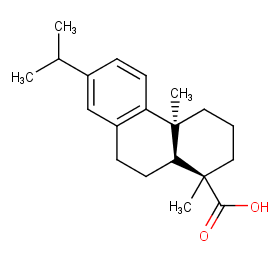
Ingredient ID: NPC189078
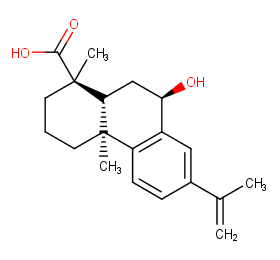
Ingredient ID: NPC183339
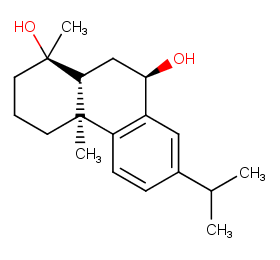
Ingredient ID: NPC181782
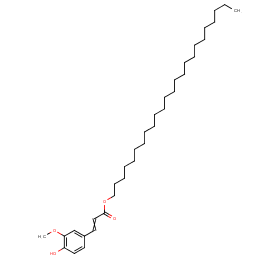
Ingredient ID: NPC176308
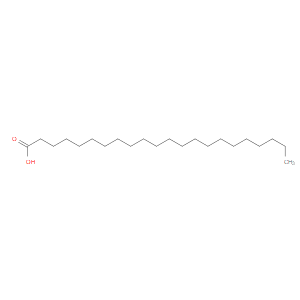
Ingredient ID: NPC171736
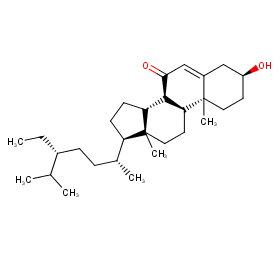
Ingredient ID: NPC15999
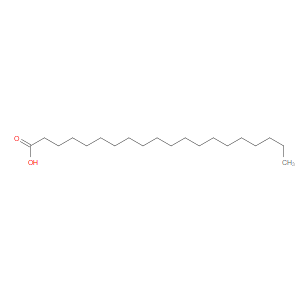
Ingredient ID: NPC149184

Ingredient ID: NPC142330
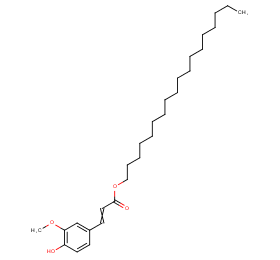
Ingredient ID: NPC141836
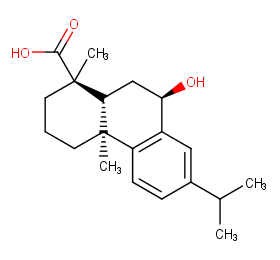
Ingredient ID: NPC133389
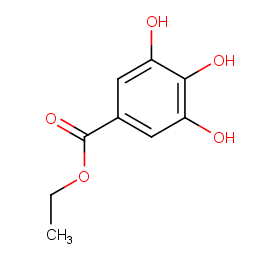
Ingredient ID: NPC1321
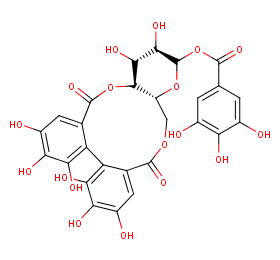
Ingredient ID: NPC121372
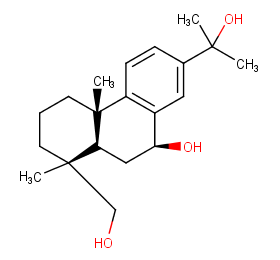
Ingredient ID: NPC104551
Classification of Human Proteins Collectively Targeted by the Plant
Detailed Information of Target Proteins
| Target Type | Protein Class | Gene ID | Protein Name | Uniprot ID | Target ChEMBL ID |
|---|---|---|---|---|---|
| Cytochrome P450 | Cytochrome P450 family 19 | CYP19A1 | Cytochrome P450 19A1 | P11511 | CHEMBL1978 |
| Therapeutic Target | Enzyme | KARS1 | Lysyl-tRNA synthetase | Q15046 | CHEMBL5575 |
| Therapeutic Target | Fatty acid binding protein family | FABP4 | Fatty acid binding protein adipocyte | P15090 | CHEMBL2083 |
| Therapeutic Target | Structural protein | LMNA | Prelamin-A/C | P02545 | CHEMBL1293235 |
Clinical trials associated with plant from natural product (NP) & plant level:
| Clinical trials type | Number of clinical trials | |
|---|---|---|
| 35 | ||
| NCT ID | Title | Condition | Form in clinical use | Associated by plant or compound |
|---|---|---|---|---|
| NCT02059785 | Phase ⅡStudy of Pinocembrin Injection to Treat Ischemic Stroke | Ischemic stroke | Pinocembrin (NPC243083) | |
| NCT03761134 | Efficacy and Safety of Sotagliflozin Versus Placebo in Chinese Patients With Type 2 Diabetes Mellitus Not Adequately Controlled by Metformin With or Without Sulfonylurea | type 2 diabetes mellitus | Urea (NPC142330) | |
| NCT01871558 | Vildagliptin/Metformin in T2DM Patients Starting Basal Insulin | type 2 diabetes mellitus | Urea (NPC142330) | |
| NCT01242215 | A Study to Assess the Efficacy and Safety of ASP1941 in Combination With Sulfonylurea in Type 2 Diabetic Patients | type 2 diabetes mellitus | Urea (NPC142330) | |
| NCT02111096 | A Study of LY2409021 in Participants With Type 2 Diabetes Mellitus | type 2 diabetes mellitus | Urea (NPC142330) | |
| NCT01658501 | Phase 2b Multicenter, Randomized, Double-Blind, Placebo- and Active-Controlled, Parallel-Group Study to Assess the PD Response and Safety of Three Dose Levels of (PB1023) Injection Following 20 Weeks of Weekly SC Dosing in Adults With T2DM | type 2 diabetes mellitus | Urea (NPC142330) | |
| NCT02249884 | Dose-Response Curve: Interventions to Prevent and Treat Radiodermatitis | radiodermatitis | Urea (NPC142330) | |
| NCT02247830 | Management Radiodermatitis in Patients With Breast or Head and Neck Cancer | radiodermatitis | Urea (NPC142330) | |
| NCT00379769 | RECORD: Rosiglitazone Evaluated for Cardiac Outcomes and Regulation of Glycaemia in Diabetes | type 2 diabetes mellitus | Urea (NPC142330) | |
| NCT01777282 | A Study to Determine the Long Term Safety and Efficacy of Albiglutide in Combination With Oral Monotherapy Antihyperglycemic Medications in Japanese Patients With Type 2 Diabetes Mellitus | diabetes mellitus | Urea (NPC142330) | |
| NCT02532855 | A Study to Assess the Addition of Sitagliptin to Metformin Compared With the Addition of Dapagliflozin to Metformin in Participants With Type 2 Diabetes Mellitus (T2DM) and Mild Renal Impairment Who Have Inadequate Glycemic Control on Metformin With or Without a Sulfonylurea (MK-0431-838) | type 2 diabetes mellitus | Urea (NPC142330) | |
| NCT03901144 | A NOVel Moisturiser for Atopic Dermatitis: Effect on the Skin Barrier | atopic eczema | Urea (NPC142330) | |
| NCT03066830 | Efficacy and Safety of Sotagliflozin Versus Placebo in Participants With Type 2 Diabetes Mellitus on Background of Sulfonylurea Alone or With Metformin | type 2 diabetes mellitus | Urea (NPC142330) | |
| NCT03376620 | Role of Topical Putrescine (Fibrostat) for Prevention of Hypertrophic Scars in Mammoplasty Patients | severe cutaneous adverse reaction | Urea (NPC142330) | |
| NCT01340768 | Study to Compare Sitagliptin Versus Sulfonylurea Treatment During Ramadan Fasting in Patients With Type 2 Diabetes (MK-0431-262) | type 2 diabetes mellitus | Urea (NPC142330) | |
| NCT00203905 | A Study of Chemoradiotherapy for Intermediate Stage/Selected Stage IV Cancers of the Head and Neck | head and neck malignant neoplasia | Urea (NPC142330) | |
| NCT02251392 | Evaluation of Therapeutic Efficacy in Use of Chamomilla Gel, Chamomilla Infuse and Urea in Radiodermatitis | radiodermatitis | Urea (NPC142330) | |
| NCT04809220 | A Study of Two Doses of Dulaglutide (LY2189265) in Japanese Patients With Type 2 Diabetes | type 2 diabetes mellitus | Urea (NPC142330) | |
| NCT01137812 | The CANTATA-D2 Trial (CANagliflozin Treatment And Trial Analysis - DPP-4 Inhibitor Second Comparator Trial) | type 2 diabetes mellitus | Urea (NPC142330) | |
| NCT00395746 | Effect of Liraglutide in Combination With Sulfonylurea (SU) on Blood Glucose Control in Subjects With Type 2 Diabetes | type 2 diabetes mellitus | Urea (NPC142330) | |
| NCT01934829 | Urea Cream Treatment Sorafenib-Associated HSFR in HCC | hepatocellular carcinoma | Urea (NPC142330) | |
| NCT01381900 | A Efficacy, Safety, and Tolerability Study of Canagliflozin in Patients With Type 2 Diabetes Mellitus With Inadequate Glycemic Control on Metformin Alone or in Combination With a Sulphonylurea | type 2 diabetes mellitus | Urea (NPC142330) | |
| NCT02670889 | Manipulating the Gut Microbiome Study | argininosuccinic aciduria;carbamoyl phosphate synthetase I deficiency disease;citrullinemia;Ornithine transcarbamylase deficiency | Urea (NPC142330) | |
| NCT00713830 | GLP-1 Receptor Agonist Lixisenatide in Patients With Type 2 Diabetes for Glycemic Control and Safety Evaluation, on Top of Sulfonylurea | type 2 diabetes mellitus | Urea (NPC142330) | |
| NCT05348278 | Urea Cream Prevention for Capecitabine Associated Hand Foot Syndrome | Hand-foot syndrome | Urea (NPC142330) | |
| NCT00174642 | Opposing Step-by-step Insulin Reinforcement to Intensified Strategy | type 2 diabetes mellitus | Urea (NPC142330) | |
| NCT01169779 | Efficacy and Safety of Lixisenatide in Patients With Type 2 Diabetes Mellitus Insufficiently Controlled by Metformin | type 2 diabetes mellitus | Urea (NPC142330) | |
| NCT00667589 | Sorafenib-induced Hand- Foot Skin Reaction Treatment | skin disease | Urea (NPC142330) | |
| NCT04994951 | Pilot Study of Traditional Chinese Medicine (Qing-Re-Liang-Xue Decoction) as Complementary Medicine for Psoriasis Vulgaris of Blood-heat Syndrome. | psoriasis vulgaris | Urea (NPC142330) | |
| NCT00789191 | Effect of Detemir and Sitagliptin on Blood Glucose Control in Type 2 Diabetes | type 2 diabetes mellitus | Urea (NPC142330) | |
| NCT00267111 | Topical Amethocaine Gel for Intramuscular Injection in Term Neonates | pain | Urea (NPC142330) | |
| NCT00576238 | Skin Tolerance Study of Betamethasone Creams in Atopic Eczema and the Preventative Properties of a Moisturiser | atopic eczema | Urea (NPC142330) | |
| NCT00575588 | 52-week add-on to Metformin Comparison of Saxagliptin and Sulphonylurea, With a 52-week Extension Period | type 2 diabetes mellitus | Urea (NPC142330) | |
| NCT00359112 | AVANDAMET Versus Metformin And Sulphonylurea In People With Poorly Controlled Type 2 Diabetes | type 2 diabetes mellitus | Urea (NPC142330) | |
| NCT00866658 | GLP-1 Receptor Agonist Lixisenatide in Patients With Type 2 Diabetes for Glycemic Control and Safety Evaluation, on Top of Basal Insulin +/- Sulfonylurea (GETGOAL-L-ASIA) | type 2 diabetes mellitus | Urea (NPC142330) |
❱❱❱ Associated Human Diseases and Detailed Association Evidence
How do we define the Plant-Targeted Human Disease Association?
Associated human diseases of an individual plant are summurized based on FOUR types of association evidence, these include:
❶ Association by Therapeutic Target: Bioactive protein targets of the plant were defined in "Molecular Targets" section, target-disease associations collected from TTD database were subsequently used to build the associations between the plant and its targeted human diseases.
❷ Association by Disease Gene Reversion: Plant and a specific disease will be associated when >= 1 plant target gene overlaped with disease's DEGs.
❸ Association by Clinical Trials of Plant: Plant and a specific disease will be associated when >= 1 clinical trial (the plant is the intervetion) can be matched in ClinicalTrials.gov database.
❹ Association by Clinical Trials of Plant Ingredients: Plant and a specific disease will be associated when >= 1 clinical trial (the plant ingredient is the intervetion) can be matched in ClinicalTrials.gov database.
Associated Disease of the Plant |
Association Type & Detailed Evidence |
|---|---|
Hepatocellular carcinoma of liverDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C12.02 |
FABP5,FABP4,CYP19A1,OR51E2,CYP19A1
|
HepatoblastomaDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C12.01 |
FABP5,FABP4,CYP19A1,OR51E2
|
Diffuse large B-cell lymphomasDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A81 |
OR51E2,FABP5,FABP4,LMNA
|
Other specified malignant neoplasms of kidney, except renal pelvisDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C90.Y |
FABP5,CYP19A1,FABP5,OR51E2
|
Dermatoses due to ionizing radiation, unspecifiedDisease Category: 14.Diseases of the skinDisease ICD-11 Code: EJ7Z |
NCT02247830,NCT02249884,NCT02251392
|
Malignant neoplasms of biliary tract, distal bile ductDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C15 |
FABP5,LMNA,OR51E2
|
Glioblastoma of brainDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2A00.00 |
FABP5,LMNA,CYP19A1
|
Malignant neoplasms of adrenal glandDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D11 |
FABP5,CYP19A1,OR51E2
|
Type 2 diabetes mellitusDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A11 |
NCT01658501,NCT02532855,NCT03066830,NCT00359112,NCT00395746,NCT01871558,NCT00866658,NCT00575588,NCT01340768,NCT01242215,NCT00174642,NCT02111096,NCT01381900,NCT00789191,NCT01169779,NCT03761134,NCT04809220,NCT00713830,NCT00379769,NCT01137812
|
PsoriasisDisease Category: 14.Diseases of the skinDisease ICD-11 Code: EA90 |
FABP5
NCT04994951 |
Atopic eczemaDisease Category: 14.Diseases of the skinDisease ICD-11 Code: EA80 |
NCT03901144,NCT00576238
|
Carcinosarcoma of uterusDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C76.43 |
FABP5,CYP19A1
|
PheochromocytomaDisease Category: X.Extension CodesDisease ICD-11 Code: XH3854 |
FABP5,OR51E2
|
Adenocarcinoma of pancreasDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C10.0 |
LMNA,OR51E2
|
Mesothelioma of pleuraDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C26.0 |
LMNA,OR51E2
|
Adenocarcinoma of prostateDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C82.0 |
FABP5,OR51E2
|
Chikungunya virus diseaseDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D40 |
LMNA,CYP19A1
|
EndometriosisDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GA10 |
CYP19A1
|
Breast cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C60-2C6Y |
CYP19A1
|
NeuropathyDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8C0Z |
CYP19A1
|
OesophagitisDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DA24 |
CYP19A1
|
Cushing syndromeDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A70 |
CYP19A1
|
Other specified disorders of urea cycle metabolismDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5C50.AY |
NCT02670889
|
Pain, unspecifiedDisease Category: 21.Symptoms, signs or clinical findings, not elsewhere classifiedDisease ICD-11 Code: MG3Z |
NCT00267111
|
Skin diseaseDisease Category: 14.Diseases of the skinDisease ICD-11 Code: EA00-EM0Z |
NCT00667589
|
Hepatocellular carcinoma, NOSDisease Category: X.Extension CodesDisease ICD-11 Code: XH4W48 |
NCT01934829
|
Argininosuccinic aciduriaDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5C50.A0 |
NCT02670889
|
Head and neck cancerDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D42 |
NCT00203905
|
Diabetes mellitus, type unspecifiedDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5A14 |
NCT01777282
|
Disorders of urea cycle metabolismDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5C50.A |
NCT02670889
|
Other specified syndromes with multiple structural anomalies, not of environmental originDisease Category: 20.Developmental anomaliesDisease ICD-11 Code: LD2F.1Y |
NCT05348278
|
CitrullinaemiaDisease Category: 05.Endocrine, nutritional or metabolic diseasesDisease ICD-11 Code: 5C50.A3 |
NCT02670889
|
Cerebral ischaemic strokeDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8B11 |
NCT02059785
|
Unspecified adverse cutaneous reactions to medicationDisease Category: 14.Diseases of the skinDisease ICD-11 Code: EH7Z |
NCT03376620
|
Serous cystadenoma,borderline malignancy of ovaryDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C73.4 |
FABP5
|
Adenocarcinoma of stomachDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B72.0 |
CYP19A1
|
Malignant neoplasm metastasis in lymph nodes of head, face or neckDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2D60.0 |
FABP4
|
Non-alcoholic fatty liver diseaseDisease Category: 13.Diseases of the digestive systemDisease ICD-11 Code: DB92 |
FABP4
|
Malignant neoplasms of bladderDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C94 |
FABP4
|
Squamous cell carcinoma of bronchus or lungDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C25.2 |
OR51E2
|
West Nile virus infectionDisease Category: 01.Certain infectious or parasitic diseasesDisease ICD-11 Code: 1D46 |
CYP19A1
|
Superficial ovarian endometriosisDisease Category: 16.Diseases of the genitourinary systemDisease ICD-11 Code: GA10.B4 |
LMNA
|
Systemic sclerosisDisease Category: 04.Diseases of the immune systemDisease ICD-11 Code: 4A42 |
FABP4
|
Malignant neoplasms of stomachDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2B72 |
FABP4
|
Malignant neoplasms of thymusDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C27 |
FABP5
|
Germ cell tumour of testisDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C80.2 |
FABP5
|
Carpal tunnel syndromeDisease Category: 08.Diseases of the nervous systemDisease ICD-11 Code: 8C10.0 |
OR51E2
|
Malignant neoplasm of pancreasDisease Category: 02.NeoplasmsDisease ICD-11 Code: 2C10 |
FABP4
|

